31 Gastric Pull-Up
Abstract
The gastric pull-up procedure for reconstruction of circumferential pharyngoesophageal defects was introduced in the 1960s. The procedure was popularized and refined in the 1970s and 1980s. It was during this period that large case series demonstrating the procedure as safe and reliable were published. Classic indications for the procedure are for reconstruction of circumferential pharyngeal defects after total laryngopharyngectomy and/or cervical esophagectomy. The procedure is completed in three steps. First, the larynx and the pharyngoesophageal region and upper thoracic esophagus are mobilized through the neck. Second, the stomach and lower esophagus are mobilized in and through the abdominal cavity. The last step involves transposition of the stomach into the neck, followed by pharyngogastric anastomosis to restore continuity of the alimentary tract. The advantage of this procedure is the immediate reconstruction of a mucosal lined alimentary tract with only a single anastomosis required. The major disadvantage is the morbidity of operating in three separate visceral spaces. Recently, the popularity of gastric pull-up has decreased with the advent of microvascular free flap reconstruction. However, for reconstruction after total laryngopharyngoesophagectomy and upper thoracic esophagectomy, gastric pull-up remains a good option.
31.1 History
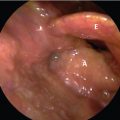
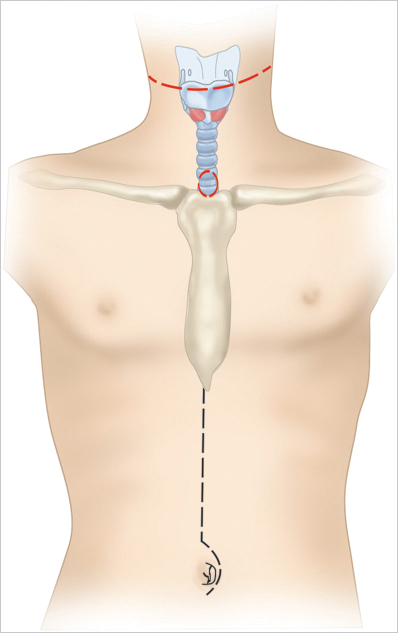
Reconstruction of the upper alimentary tract following circumferential resection of cancer of the hypopharynx and cervical esophagus has long been a challenge. In the first half of the 20th century, reconstruction was mainly performed by using local flaps as described by Trotter (1912) and Wookey (1940). 1 Multistaged reconstruction with tubed pedicled flaps was the next advancement in pharyngeal reconstruction, which took up to 6 to 8 months to complete the reconstruction. Some patients already had recurrence by the time reconstruction was complete. The multiple stages required prolonged hospitalization, and during which time, the patient was dependent upon nasogastric tube feedings. The deltopectoral flap, popularized by Bakamjian et al, was a major advancement and took only two procedures to complete the reconstruction. 2 In 1960, Ong and Lee reported their experience with pharyngogastric anastomosis following pharyngolaryngoesophagectomy.s. Literatur Le Quesne and Ranger, and later Harrison and Thompson, refined the technique in favor of trans-hiatal pull-through, without opening the chest, and thus the gastric transposition or gastric pull-up operation was born. 4 , 5 , 6 , 7 This operation accomplished reconstruction of the alimentary tract in a single stage, reducing the prolonged morbidity of multistage procedures. Patients are able to swallow a normal diet in less than 2 weeks postoperatively ( Fig. 31‑1). In 1981, Spiro and his colleagues at the Memorial Hospital published a series of 120 patients who were reconstructed with the gastric transposition following cervical esophagectomy or circumferential pharyngectomy. 8 , 9 This series established that the procedure was safe and highly reliable in carefully selected patients. However, the postoperative morbidity for this procedure was high. Refinements in surgical techniques using laparoscopic and thoracoscopic esophagogastric approaches to mobilize the stomach and esophagus have not reduced the morbidity. 10

31.2 Indications
The classic indications for the gastric pull-up procedure are for reconstruction of circumferential pharyngeal defects after total laryngopharyngectomy and/or cervical esophagectomy. 11 , 12 However, the usefulness of this procedure has been further modified to reconstruct the hypopharynx and cervical esophagus in non-oncologic disease processes in both adults and children. These indications include severe stricture or fistula after caustic ingestion, infection, and in the treatment of esophageal atresia. The advantage of the gastric pull-up is the immediate restoration of a mucosal lined alimentary tract with tissue from elsewhere in the alimentary tract with only a single mucosal anastomosis required. In recent years, however, the gastric pull-up procedure has fallen out of favor because of significant morbidity of dissection in three separate visceral spaces and the availability and reliability of free tissue transfer for reconstruction. 13 , 14 , 15 However, for reconstruction after total laryngopharyngoesophagectomy and upper thoracic esophagectomy, gastric pull-up remains the best option. 11 , 12
31.3 Contraindications
Cancers involving the upper thoracic esophagus and perforating through the membranous trachea are not suitable for the gastric pull-up procedure. Similarly, radiologically demonstrated mediastinal lymph nodes are a contraindication. Previous surgery on the stomach, such as partial gastrectomy or the presence of other pathology in the stomach or esophagus such as esophageal varices, a peptic ulcer, or gastric tumor makes the stomach unsuitable for transposition. Patients with previous mediastinal surgery are not ideal candidates for safe dissection and mobilization of upper thoracic esophagus.
31.4 Preoperative Preparation
The preoperative evaluation should include imaging of the head, neck, and mediastinum using CT scan with contrast or MRI. In addition, an upper gastrointestinal (GI) series with barium esophagogram should be included to evaluate the suitability of the stomach for transposition. Any abnormalities should be further investigated, and alternative methods of reconstruction should be pursued if the stomach is not suitable for transposition. Esophagoscopy, tracheobronchoscopy, and upper GI endoscopy are essential, and a biopsy is required for confirmation of tissue diagnosis. Preoperative consultation with a thoracic surgeon is required for participation in the surgery.
A thorough bowel preparation prior to the operation and admission to the hospital a day prior to surgery is desirable for IV hydration. A detailed informed consent from the patient must be obtained after discussing risk, benefits, alternatives, and possible complications and sequela of the operation. These include complications of surgery and anesthesia, including the risk of fistula, mediastinitis, and pneumothorax. The patient should be informed of the postoperative sequela of dumping syndrome, gastric outlet obstruction, and gastric reflux. The likelihood of postoperative hypothyroidism and hypoparathyroidism should be discussed, and the patient should be made aware of the need for thyroid and calcium replacement. In the past, the risk of intraoperative and postoperative mortality was high, and the patient should be advised of the small but finite risk of death.
31.5 Operative Steps
The patient is positioned on the operating table in the supine position with the neck extended. The procedure is started with both the head and neck and thoracic surgery teams working simultaneously. This allows the procedure to proceed efficiently and can minimize the total operative time. However, if there is any question about the resectability of the primary tumor, that is, involvement of the prevertebral fascia, carotid artery, or invasion of thoracic trachea, then the abdominal and mediastinal components of the procedure should be delayed until the head and neck surgeon can make such a determination. Excessive crowding around the operating table can be avoided by having the two surgeons stand on opposite sides of the table. The head and neck team can use the area around the head of the bed for any assistant surgeons. The skin of the patient is prepped with Betadine solution from the upper lip, to the pubis, and separate drapes are applied to isolate the fields for dissection in the neck and laparotomy. 16 , 17 , 18 , 19 , 20
31.5.1 Cervicothoracic Dissection
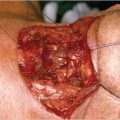
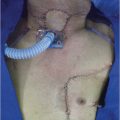
A transverse incision in the midcervical region is preferred, leaving a distance of at least 3 cm, above the upper border of the permanent tracheostome ( Fig. 31‑2). Alternatively, a U-shaped curvilinear incision can be employed along the anterior border of the sternocleidomastoid muscles, ending at the lower end with the circular incision for the tracheostome. The permanent tracheostome is ideally placed in the suprasternal notch. If required, the incision can be extended below the tracheostome in the midline of the anterior chest wall overlying the manubrium sterni. This extension facilitates excision of the manubrium in patients with distal tracheal invasion requiring substernal tracheal resection. Resection of the manubrium can also be used to facilitate transposition of the stomach through the thoracic inlet in obese patients, in whom the normal thoracic inlet is not sufficient to deliver the stomach in to the neck.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree