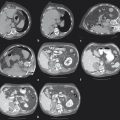
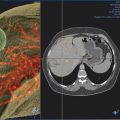
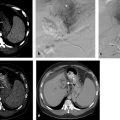
1 Percutaneous Ablation of Renal Cell Carcinoma
1.1 Introduction
Renal cell carcinoma (RCC) accounts for approximately 4% of all adult malignancies, with more than 65,000 estimated new cases projected in the United States in 2015, resulting in nearly 14,000 deaths during the same period of time. 1 The disease is more common in men than in women. 1 Risk factors include hypertension, smoking, obesity, and hemodialysis in patients with end-stage renal disease. Patients with certain genetic syndromes, such as von Hippel–Lindau (VHL) disease, have a high incidence of multiple and recurrent tumors. The classic presentation is the triad of flank pain, hematuria, and palpable abdominal mass. Other symptoms include loss of appetite, weight loss, anemia, and fatigue. Four dominant RCC subtypes have been identified, with the clear cell variety representing about 75% of cases. 2
Renal malignancies are staged using the TNM staging system ( Table 1.1 ). 3 Approximately one-fifth of patients present with advanced disease and have associated poor 5-year survival. 1 The increased use of cross-sectional imaging, however, has led to earlier detection. Currently, more than half of all renal masses are discovered incidentally, 4 with as many as 80% of patients presenting with localized, low-stage disease. 1 Early detection of small, organ-confined tumors has led to the development of nephron-sparing treatment options. Partial nephrectomy has shown comparable long-term survival when compared to radical nephrectomy in the treatment of organ-confined, stage I and II tumors, 5 , 6 , 7 and has thus become the treatment of choice for these patients. Image-guided percutaneous ablation is also fast becoming an accepted treatment option for select patients with small tumors. 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 These methods offer a number of advantages over surgery. First, percutaneous ablation has been shown to be associated with fewer complications and faster recovery compared to surgery. 8 The procedures can also be repeated when not completely successful or in syndromic patients with recurrent tumors. Importantly, there is some evidence to suggest that renal function in patients with solitary kidneys is better preserved following ablation as compared to partial nephrectomy. 20 Finally, percutaneous ablation may be the only local treatment option in patients who are poor surgical candidates.
Primary tumor (T) | |
TX | Primary tumor cannot be assessed |
T0 | No evidence of primary tumor |
T1 | Tumor ≤ 7 cm in greatest dimension, limited to the kidney |
T1a | Tumor ≤ 4 cm in greatest dimension, limited to the kidney |
T1b | Tumor > 4 cm but ≤ 7 cm in greatest dimension, limited to the kidney |
T2 | Tumor > 7 cm in greatest dimension, limited to the kidney |
T2a | Tumor > 7 cm but ≤ 10 cm in greatest dimension |
T2b | Tumor > 10 cm, limited to the kidney |
T3 | Tumor extends into major veins or perinephric tissues but not into the ipsilateral adrenal gland and not beyond Gerota’s fascia |
T3a | Tumor grossly extends into the renal vein or its segmental (muscle-containing) branches, or tumor invades perirenal and/or renal sinus fat but not beyond Gerota’s fascia |
T3b | Tumor grossly extends into the vena cava below the diaphragm |
T3c | Tumor grossly extends into the vena cava above the diaphragm or invades the wall of the vena cava |
T4 | Tumor invades beyond Gerota’s fascia (including contiguous extension into the ipsilateral adrenal gland) |
Regional lymph nodes (N) | |
NX | Regional lymph nodes cannot be assessed |
N0 | No regional lymph node metastasis |
N1 | Metastasis to regional lymph node(s) |
Distant metastasis (M) | |
M0 | No distant metastasis |
M1 | Distant metastasis |
Several percutaneous ablative methods have been described, including radiofrequency ablation (RFA), cryoablation, microwave ablation, and high-intensity, focused ultrasound ablation. The largest experience to date has been with RFA and cryoablation, with hundreds of cases reported in the literature. 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 , 27 , 28 , 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39 , 40 , 41 , 42 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 61 , 62 , 63 , 64 , 65 , 66 , 67 , 68 There are comparatively limited data on the effectiveness of percutaneous microwave ablation and high-intensity focused ultrasound. 16 , 69 , 70 , 71 , 72 , 73 , 74 , 75 , 76 Several investigators have reported the short-term and mid-term effectiveness of both RFA and cryoablation of small renal masses with favorable results. 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 , 27 , 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39 , 40 , 41 , 42 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 62 , 63 , 65 , 66 , 67 , 77 , 78 Long-term survival data (> 5 years) following RCC ablation is sparse; however, early studies have shown promise. 13 , 28 , 61 , 64 , 77 To date, there is no compelling evidence to suggest superiority of one ablative method over another. A recent meta-analysis comparing RFA and cryoablation demonstrated similar clinical efficacy and complication rates. 79 As such, the choice between RFA or cryoablation is often driven by physician preference and local practice patterns.
1.2 Indications
Surgical resection remains the standard of care for RCC treatment given the lack of robust, long-term survival and disease-free survival data with percutaneous ablation procedures. Percutaneous ablation is thus limited to patients who are not ideal surgical candidates. Formal indications for the use of percutaneous ablation in the treatment of RCC have yet to be validated with prospective scientific studies. 33 Percutaneous ablation therapies are, however, considered appropriate for use in elderly patients, in patients with comorbid conditions that preclude surgery, in individuals with a solitary kidney or renal insufficiency, and in patients with multiple or recurrent tumors as with VHL disease. Additionally, ablation is usually performed in patients with a > 1-year life expectancy, because a small RCC is unlikely to cause clinically significant morbidity before 1 year. 14
In addition to the patient-specific criteria already listed, there are tumor-specific criteria, such as lesion size and location, that are also important in determining the feasibility of percutaneous intervention. Multiple studies have shown decreased effectiveness of RFA with increasing tumor size. 14 , 15 , 16 , 17 , 19 , 20 , 21 , 23 , 34 , 35 , 36 , 37 , 53 , 55 Zagoria et al 17 reported a technical success rate with RFA ablation of 100% in tumors < 3 cm as compared to 69% with tumors > 3 cm. In a retrospective study of 100 tumors treated with RFA, Gervais et al 14 , 15 found that tumor size was an independent predictor of RFA success. Ninety-two percent of tumors < 3 cm were successfully ablated in a single session, whereas 44% of tumors measuring 3 to 5 cm required more than one ablative session for complete eradication. Tumors > 5 cm invariably required more than one session.
Tumor location is also important in determining the success of an ablative procedure. Gervais et al showed that a noncentral location is an independent predictor of complete tumor necrosis. 14 , 15 This is believed to be due in part to insulation of the exophytic tumor by surrounding perirenal fat allowing generation of higher ablative temperatures. Poor treatment outcomes with centrally located tumors in turn may be the result of a “heat sink” effect of blood flow in hilar vessels causing perfusion-mediated tumor cooling. 80 A retrospective study of 115 renal tumors treated with cryoablation reported similar results with regard to tumor size and position. 39 In the study, Atwell et al reported technical success in 112 of 115 tumors (97%). The three technical failures occurred in central tumors, two of which were > 4 cm. Other considerations include the proximity of central tumors to the collecting system, ureter, and central renal vasculature, which increases the risk of complication, including significant hemorrhage and ureteral injury. On the other hand, injury to adjacent organs or bowel is a concern with some exophytic masses.
1.3 Contraindications
Absolute contraindications to percutaneous renal ablation include uncorrected coagulopathy and acute illness. 33 Serious comorbidities (e.g., congestive heart failure, pulmonary hypertension, cardiac arrhythmias) can be considered a relative contraindication necessitating assessment on a case by case basis. High-risk patients may benefit from close monitoring by anesthesiology personnel. Patients with large tumors, as already discussed, may be better served by surgical resection. By the same token, local tumor spread beyond Gerota’s fascia or distant metastatic spread may preclude curative intent. Ablation may still be indicated with advanced disease, with the purpose of debulking or for palliation to control pain or hematuria. 81
1.4 Patient Selection and Preprocedure Workup
All potential ablation patients are evaluated in the interventional radiology clinic in the weeks or months leading up to the procedure. The clinic visit is an opportunity for the physician to develop a rapport with the patient as well as the patient’s family. The proposed procedure, including the risks, benefits, and alternatives, is discussed in detail with the patient. A detailed medical history is obtained and a full physical exam performed. Additionally, every patient should undergo contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI) for tumor staging, and appropriate laboratory tests should be ordered.
1.4.1 Laboratory Studies
Appropriate blood tests include a complete blood count (CBC), coagulation studies, and renal function tests. Anticoagulants and antiplatelet agents are discontinued far enough in advance of the procedure for coagulation parameters and function to return to normal. 32 Given the elective nature of the procedure and the hypervascularity of RCCs, it is reasonable to postpone the procedure in the setting of markedly abnormal laboratory findings, such as an international normalized ratio (INR) > 1.5 or a platelet count < 50 × 103/μL. If necessary, fresh frozen plasma and platelets can be administered prior to or during the procedure.
Patients being considered for percutaneous ablation often have abnormal renal function. It is important to assess renal function prior to the procedure with a serum creatinine and estimated glomerular filtration rate (GFR) because this will help to guide the procedural approach and imaging follow-up. Iodinated contrast may be used during CT-guided ablation to define the tumor margins before applicator placement or during the procedure to assess whether the tumor has been successfully ablated. In the case of cryoablation, contrast is not used as frequently because the ice ball is well seen with both unenhanced CT and MRI. 9 , 11 , 12 , 41 , 42 , 43 , 44 , 45 , 46 , 47 , 48 , 49 , 50 , 51 , 82
1.4.2 Preprocedure Biopsy
There is some controversy regarding the need for a biopsy prior to percutaneous ablation. However, given that a substantial fraction of small, solid masses are benign, 83 it seems prudent to obtain a tissue diagnosis in most cases. This not only has the advantage of decreasing unnecessary treatment in patients with benign neoplasms, such as angiomyolipoma or oncocytoma, it also allows tumor subtyping and grading in the case of malignancy, information that can be used to tailor future treatment and surveillance. 84 The timing of the biopsy is also somewhat controversial; some advocate tissue sampling prior to ablation, 85 whereas others favor performing both procedures on the same day. 86 In most circumstances, it seems reasonable to obtain a biopsy in advance of the ablation because this provides the pathologist more time to analyze the specimen with special staining, and, in the case of a benign diagnosis, it obviates the need for an additional clinic visit and further workup. 87 On the other hand, if the probability of renal cancer is extremely high, as in the case of a suspicious mass in a patient with VHL disease, same-day biopsy may be a reasonable approach .
1.4.3 Prophylactic Antibiotics
Periprocedural administration of prophylactic antibiotics is another topic of debate. 32 Some practitioners favor empirical antibiotic use in all patients, whereas others reserve prophylaxis for select, high-risk cases. 88 , 89 At our institution, antibiotics are given only to patients who are felt to be at increased risk of infection. This includes patients with a history of ileal conduit and ureteral stent placement. Some have advocated that patients with diabetes and those with a history of chronic immunosuppression also warrant consideration for extended antibiotic coverage tailored toward gram-negative enterics. 54
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree