White Cell Disorders: Introduction
White blood cells (WBC) provide major host defense mechanisms against invading pathogens through phagocytosis and the immune response. In addition, lymphocytes provide immune surveillance against the development of cancer and are important mediators of disorders of the immune system. White cell disorders are usually the result of overproduction or underproduction of one or more of the WBC series, which include the granulocytes—neutrophils, basophils, and eosinophils—and/or mononuclear cells—lymphocytes and monocytes. Less frequently, WBC disorders result from WBC dysfunction despite normal number of cells. The acuity and severity of these disorders are related to the number of white cells, the subset(s) of WBC involved, their degree of maturity, and their functional capacity.
Initial clinical manifestations of WBC disorders can vary widely but often include signs and symptoms of infection. Disorders of WBC, benign or malignant, frequently also involve abnormalities of the red blood cells (RBCs) and platelets. In these situations, the signs and symptoms of disease at presentation may include those related to anemia (i.e., weakness and easy fatigability) and/or thrombocytopenia (i.e., easy bruisability, mucosal or gastrointestinal bleeding, and hematuria). A careful history and physical examination with evaluation of the peripheral blood, and bone marrow when needed, will establish a diagnosis in many cases.
Aging is not associated with significant changes in the peripheral WBC count or WBC differential (Table 103-1). Therefore, abnormalities in white cell numbers should be evaluated as a probable sign of an active disease process. Most white cell disorders are more common with increased age and the most common WBC disorders in the elderly are neoplastic diseases. Table 103-2 lists the spectrum of white cell disorders encountered in clinical practice. Full descriptions of these processes are available in textbooks of hematology and oncology. This chapter focuses on these disorders as related to geriatric medicine.
|
The most common assessment of white cells is quantitative evaluation of their numbers. Careful attention should also be paid to the distribution of the different types of white cells (WBC differential). Equally important is the microscopic examination of the peripheral blood smear (and bone marrow when needed) for morphologic changes in the WBC as well as the red cells and platelets. Specific quantitation of WBC subsets by immunophenotyping techniques can aid in the diagnosis of many neoplastic WBC disorders. Qualitative or functional defects are more difficult to establish with laboratory techniques and are usually strongly suggested by the medical history.
Decreased White Blood Cells
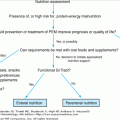
Leukopenia may occur with a broad array of medical problems. Leukopenia can range from a mild suppression of the WBC to clinically significant neutropenia (Table 103-3). Most intrinsic (hereditary/familial) disorders and syndromes are detected during infancy or childhood. Leukopenia in older adults is generally the result of acquired or secondary disorders, either reactive (drugs, nutritional, infections, immune) or malignant. Table 103-4 presents key components in the clinical workup of neutropenia.
1. Clinical history | Duration (acute vs. chronic) |
History of infection | |
History of bleeding/bruising/fatigue | |
Medication history | |
Presence of comorbidities (rheumatologic disease, cirrhosis, malabsorption) | |
2. Examination | Evaluate for signs of infection including careful skin and mucosal examination |
Evidence of bleeding/bruising | |
Splenomegaly | |
3. Complete blood count | Severity of neutropenia |
Presence of anemia or thrombocytopenia | |
Macrocytosis | |
Immature granulocytes on differential | |
4. Peripheral smear | Toxic granulation/vacuolation of neutrophils |
Presence of dysplastic features | |
Immature granulocytes | |
Nucleated red cells | |
Megaloblastic changes (hypersegmentation of neutrophils) | |
5. Bone marrow biopsy | Indicated for severe neutropenia, pancytopenia, evidence of dysplasia or granulocytic immaturity on peripheral smear |
Elderly patients commonly consume many prescribed and over-the-counter medications and are at higher risk for developing drug-related neutropenia or pancytopenia. The bone marrow is one of the more rapidly proliferating organs of the body; therefore, it is not surprising that exposure to noxious agents may temporarily or permanently inhibit production of one or more elements of the blood. The list of possible offenders is extensive and constantly evolving with new drug development. Table 103-5 lists some of the more commonly reported offenders. In a situation of neutropenia, leukopenia, or pancytopenia, the physician should thoroughly review the patient’s medications and the duration of the medication exposure. In the absence of alternative explanations, medications administered within 4 weeks of onset of neutropenia should be evaluated. Short duration of exposure does not remove the possibility of a drug-induced cytopenia; it could be an idiosyncratic reaction. Diagnosis involves identification of a possible offending agent and exclusion of alternative etiologies for the leukopenia. A bone marrow biopsy is often indicated as part of the workup if the leukopenia is severe.
1. Cancer chemotherapeutic agents |
2. Psychotropic drugs: |
| Clozapine |
| Risperidone |
| Olanzapine |
| Haloperidol |
| Phenothiazine |
| Tricyclic antidepressants |
| Meprobamate |
3. Anti-inflammatory/antiarthritic drugs: |
| Sulfasalazine |
| Nonsteroidal anti-inflammatory agents |
| Colchicine |
| Immunosuppressive agents |
| Penicillamine |
| Gold salts |
| Allopurinol |
| Immunosuppressants |
4. Thyroid suppressants: |
| Propylthiouracil |
5. Cardiovascular drugs: |
| Antiarrhythmic agents (procainamide, quinidine, amiodarone) |
| ACE inhibitors (captopril, enalapril) |
| Nifedipine |
| Hydralazine |
| Propranolol |
| Methyldopa |
| Ticlopidine |
| Dipyridamole |
| Digoxin |
6. Antibiotics: |
| Sulfa drugs |
| Semisynthetic penicillins |
| Cephalosporins |
| Macrolides |
| Vancomycin |
| Rifampin |
| Clindamycin |
| Gentamicin |
| Linezolid |
7. Anticonvulsants: |
| Phenytoin and derivatives |
| Carbamazepine |
8. Thiazide diuretics |
9. Antihistamines- H2 blockers |
10. Oral hypoglycemia drugs |
Drug effects on the bone marrow range from mild neutropenias (more common) to agranulocytosis; lymphocytopenia occurs less frequently. The RBC and platelets may or may not be affected. Drug-induced reduction of the granulocytic series may be a direct toxic effect, as with many cancer chemotherapeutic drugs or may be the result of an immunologic phenomenon wherein a drug–antibody complex reacts with mature neutrophils and/or their precursors in the peripheral blood or bone marrow. Treatment usually consists of supportive care, with administration of antibiotics for febrile patients. Upon withdrawal of the offending agent, there may be a relatively brisk marrow recovery within 14 to 21 days. A bone marrow examination during this recovery period may reveal an increased number of immature elements, which can be confused with a malignant process such as an acute leukemia or myelodysplasia. If this is a possibility, close follow-up for an additional 2 to 3 weeks will usually provide the answer. The recovering marrow will go on to differentiate, but the malignant marrow will either stay the same or worsen. Other marrow toxins include a variety of household and industrial chemicals, especially organic solvents, naphthalenes, insecticides, and herbicides. Inquiries about chemical exposure from hobbies or occupations must therefore be part of history taking. While the hematologic effects of drug and chemical exposure are frequently reversible, some may result in myelodysplastic syndromes (MDS) or aplastic anemia which, in turn, may evolve into acute myelogenous leukemia (AML).
Mild neutropenia may be associated with nutritional anemias secondary to folate or B-12 deficiency. Older individuals, especially those living alone, may not be attentive to their diet for various socioeconomic, psychological, or medical reasons. The main sources of folates are fresh green vegetables, many fruits, and beans. Cooking and canning destroys folates. The body’s folate stores can be depleted after 4 to 5 months of poor dietary intake. Thus, a dietary history may provide an important clue to the diagnosis of the hematologic problem. Dietary folate deficiency may be aggravated by alcoholism and chronic hemolysis. Additional hematologic findings of folate deficiency include macrocytic anemia and hypersegmented neutrophils. Potential folate deficiency is best evaluated by measuring RBC folate.
In contrast, body stores of vitamin B-12 are not readily depleted by poor dietary habits alone. It takes 3 to 5 years to deplete the body stores of vitamin B-12. However, gastric atrophy is more common with increasing age, and this may lead to failure of gastric secretion of intrinsic factor, which binds to dietary cobalamin—a necessary step in the absorption of vitamin B-12 in the ileum. Other conditions that cause a B-12 deficiency include gastrectomy or subtotal small-bowel (ileal) resection. It should be noted that neurologic signs and symptoms of B-12 deficiency may be confused with other neurologic problems in the elderly patient. These include peripheral paresthesias (peripheral neuritis), loss of balance (posterior column damage), spasticity (lateral column damage), and impaired cognitive function. Any of these neurological signs and symptoms may be mistakenly attributed to “old age,” but if caused by B-12 deficiency may be corrected with vitamin B-12 replacement therapy. Low serum cobalamin levels (<200 ng/L) are diagnostic of deficiency and will be found in the majority of patients who are symptomatic. Subclinical cobalamin deficiency can be detected by measuring serum methylmalonic acid. Subclinical cobalamin deficiency is characterized by low normal serum cobalamin level (200–350 ng/L) and an elevated methylmalonic acid level. It should be noted that homocysteine will be elevated in both folate and cobalamin deficiency.
Copper deficiency is a more recently reported but uncommon cause of neutropenia. Neutropenia is typically associated with anemia and may be confused with early MDS, particularly in an older adult. Copper deficiency is most commonly associated with malabsorption, malnutrition, use of total parenteral nutrition, or excess oral zinc supplementation. The diagnosis is suggested by the appropriate clinical setting and a low serum copper level. Hematological changes are potentially reversible with oral copper supplementation.
A number of infections can be associated with neutropenia and/or leukopenia in older adults. These infections include viral (e.g., influenza, varicella, hepatitis A, B, or C, cytomegalovirus, human immunodeficiency virus, parvovirus B19), and bacterial (e.g., Staphylococcus aureus, brucellosis, tularemia, rickettsia, and tuberculosis). Neutropenia associated with most viral illnesses is self-limited and of minimal clinical consequence. A few viruses (hepatitis B, Epstein–Barr, and human immunodeficiency virus) can cause protracted, clinically significant neutropenia. Bacterial septicemia, particularly with Gram-negative organisms, is a serious cause of acquired neutropenia and confers a poor prognosis. The mechanism of leukopenia is usually bone marrow suppression, but in the case of overwhelming bacterial sepsis, can result from exhaustion of bone marrow reserves. Treatment focuses on supportive care including use of antibiotics for infection; the role of myeloid growth factors in this setting is unproven.
Severe and prolonged bone marrow failure may occur in association with drugs, toxins, or infections, but is frequently idiopathic. Aplastic anemia is characterized by severe pancytopenia and bone marrow hypoplasia. Clinical signs and symptoms reflect the cytopenias: infection, bleeding, and/or the signs/symptoms of anemia. Pure white cell aplasia occurs rarely. Treatments for this life-threatening disease include supportive care (transfusion, antibiotics), immunosuppression (antithymocyte globulin, cyclosporine, steroids), and stem cell transplant (limited application in the elderly population).
Immune neutropenia may be associated with various rheumatologic conditions such as systemic lupus erythematosus, rheumatoid arthritis, polyserositis, and lymphoproliferative malignancies. In these situations, neutropenia is caused by the elimination of immunoglobulin-coated granulocytes by the reticuloendothelial system. Immune-mediated neutropenia is often clinically significant with absolute neutrophil count (ANC) <500 cells/mm3, predisposing to increased infection risk. In general, the bone marrow is hypercellular or normocellular and lacking in mature neutrophils. Neutropenia of Felty’s syndrome (rheumatoid arthritis, splenomegaly, and neutropenia) has a complex etiology that includes immune destruction and suppression as well as splenic sequestration. These neutropenias frequently respond to granulocyte colony-stimulating factors (G-CSF) or granulocyte-macrophage colony-stimulating factors (GM-CSF).
Any condition causing splenomegaly (e.g., hepatic cirrhosis) may lead to sequestration of sufficient neutrophils to cause mild-to-moderate neutropenia as a component of the pancytopenia of hypersplenism. The mild-to-moderate neutropenia usually does not predispose to increased infections and often occurs in association with anemia and/or thrombocytopenia.
Increased WBC
The most common causes of sustained increases in WBC are neoplastic diseases. However, there are a number of nonneoplastic etiologies for increases in one or more white cell forms. The most common among these is a leukocytosis, specifically neutrophilia, in response to infection. This is associated with a “left shift” in the differential, with increases in bands and other less-mature granulocytes, including metamyelocytes and myelocytes. Such a normal granulocytosis can also be seen in response to an exogenous steroid. Increases in specific subsets of white cells (e.g., eosinophils or basophils) can occur in response to a number of exposures or illnesses. Hereditary and familial (e.g., Down syndrome) causes of leukocytosis are rare and usually detected early in life.
Infection stimulates the acute release of neutrophils from the marginated storage pools of the bone marrow. Neutrophilia commonly occurs with acute bacterial infections, and is less predictably seen with viruses. An increase in band neutrophils and metamyelocytes is most common. Toxic granules in those cells are frequently present on review of the peripheral blood smear. A dramatic “leukemoid reaction,” characterized by a WBC >50,000/μL, a marked left shift with increased immature granulocytes including myelocytes, promyelocytes, and even blasts, may be confused with malignant disorders, especially chronic myelogenous leukemia (CML). Leukemoid reactions are characterized by a high-leukocyte alkaline phosphatase (LAP [low in CML]) and absence of the Philadelphia chromosome. Leukemoid reactions are uncommon and the probability of an underlying bone marrow disorder should always be suspected in this setting.
Corticosteroids are the most common medications associated with increases in WBC, primarily neutrophilia. The mechanism includes reduced neutrophil adhesion and release from marrow stores. The hematopoietic colony-stimulating factors (e.g., G-CSF, GM-CSF) are designed to stimulate neutrophilia, and lithium can induce neutrophilia through similar CSF pathways. Beta-adrenergic agonists also frequently stimulate acute release of neutrophils from marginated pools.
Mild neutrophilia (WBC 12,000–20,000/μL) can occur after a stress, resulting from release of neutrophils from marginated pools into the bloodstream. Stressful stimuli include exercise, seizures, anesthesia, and surgery. Acute inflammation and tissue necrosis are also associated with neutrophilia. Examples include burns, electric shock, trauma, and infarction. Asplenia can be associated with moderate leukocytosis. Chronic inflammatory conditions such as colitis, rheumatoid arthritis, and vasculitis may stimulate a modest increase in bone marrow production and release of neutrophils. Mild to moderate leukocytosis (WBC <30,000/μL) may also occur in association with a variety of malignancies without bone marrow involvement.
Isolated increases in WBC subsets occur with or without increase in total WBC. Monocytosis may be associated with inflammatory diseases, specifically infections, autoimmune and granulomatous diseases, and numerous malignancies. Eosinophilia (>600/μL) usually represents a reaction to drugs, infection (e.g., fungi, parasites), allergic disorders, vasculitis, or malignancies. Lymphocytosis, though frequently representing malignancy, may occur in response to infection.
Malignant Disease of the Hematopoietic System
Hematologic malignancies, although most frequently characterized by elevated WBC, can be associated with low, normal, or high WBC. The WBC differential, as well as the red cell and platelet counts, are usually abnormal.
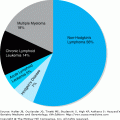
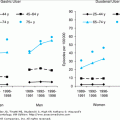
Malignant diseases of the WBCs include the four major subtypes of leukemia—AML, acute lymphocytic leukemia (ALL), CML, and chronic lymphocytic leukemia (CLL); as well as the MDS (preleukemias); the myeloproliferative disorders other than CML; hairy cell leukemia; large granular lymphocytic leukemia; metastatic cancers to the bone marrow; and circulating non-Hodgkin’s lymphoma cells. Older adults make up a large proportion of incident and prevalent cases of these diseases. In fact, the age-specific incidence rates of all leukemias increase in the elderly population (Figure 103-1). As the population ages, the burden of these diseases is nearly certain to rise, with the elderly population being most affected.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree