Introduction
Thyroid disease can present with overt symptoms, insidiously, or with isolated thyromegaly. Thyroid disease in children can encompass isolated biochemical abnormalities that have little or no physiological consequence, or with overt clinical symptoms. Clinically, hypothyroidism occurs more commonly than hyperthyroidism. Thyroid nodules and masses occur much less commonly than functional disorders but can portend the presence of thyroid cancer. Box 13.1 provides a classification of thyroid disorders in children. This chapter focuses on the most common conditions that affect the thyroid gland of children and adolescents.
Autoimmune thyroid disease
- •
Hashimoto thyroiditis, juvenile acquired hypothyroidism
- •
Stimulating antibody, Graves disease
- •
Blocking antibody, hypothyroidism
Infectious thyroiditis
- •
Suppurative thyroiditis
- •
Subacute thyroiditis
Binding protein abnormalities
- •
Complete TBG deficiency
- •
Partial TBG deficiency
- •
TBG excess
- •
Transthyretin variants
TSH Receptor mutations
- •
Loss-of-function hypothyroidism
- •
Gain-of-function hyperthyroidism
Thyroid hormone resistance syndromes
- •
Thyroid hormone beta receptor (TRβj) mutations
- •
Peripheral tissue resistance syndrome
- •
Pituitary resistance syndrome
- •
Thyroid hormone membrane transport defects
Iodine deficiency syndromes
- •
Goiter
- •
Mental impairment
- •
Cretinism
Diffuse nontoxic goiter
- •
Nonthyroidal illness
- •
Thyroid neoplasia
- •
Adenoma
- •
Nonfunctional
- •
Functional
- •
Papillary-follicular carcinoma
- •
Medullary carcinoma
- •
MEN2A, 2B, Ret mutations
- •
Sporadic
- •
Undifferentiated
- •
Metastatic
Thyroid Hormones and Their Action
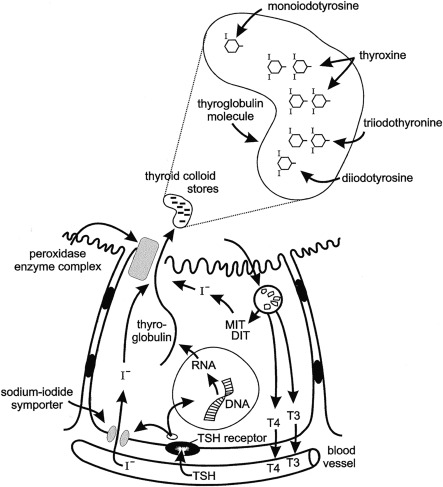
Few hormones exert as profound and essential role in human physiology as thyroid hormones. The major hormones released by the thyroid gland include tetraiodothyronine, or thyroxine (T4), and triiodothyronine (T3). The production of these hormones involves several discrete biochemical steps that are shown in Fig. 13.1 . Of these hormones, T3 plays the pivotal role in affecting physiology, being the molecule that principally binds to the thyroid hormone receptor (TRs). The thyroid hormone nuclear receptor belongs to the steroid hormone–retinoic acid receptor superfamily and is a regulator of deoxyribonucleic acid (DNA) transcription.
Two genes encode the TR; one on chromosome 17 designated alpha ( TRa ) and one on chromosome 3 designated beta ( TRb ). The TRs can exist as monomers or homodimers, and they can dimerize with other members of the family of nuclear receptors. After T3 binding to the TR, gene transcription is regulated in many tissues.
T4 is the predominant hormone released from thyroid follicular cells. After release, it circulates in protein-bound and free states at a ratio of about 1000 to 1. Thyroid hormone-binding proteins in the blood include thyroxine-binding globulin (TBG), prealbumin or transthyretin, and albumin. TBG is the predominant carrier protein for T4; TBG and albumin also carry T3. In the euthyroid study, the circulating concentration of free T4 (FT4) and free T3 are about 0.03% and 0.30%, respectively, of total hormone concentrations.
It is important to recognize that circulating levels of thyroid hormones and carrier proteins change with age ( Tables 13.1 and 13.2 ). Absolute mean free T4 and free T3 concentrations are about 10 and 4 pg/mL, respectively, and differ according to age. In adolescents and adults, the plasma concentrations of the several binding proteins are 1 to 3 mg/dL for TBG, 20 to 30 mg/dL for thyroxine-binding prealbumin, and 2 to 5 g/dL for albumin. TBG concentrations are greater in children than in adults, and they decline to adult levels during adolescence. Because the thyroid hormone-binding proteins are produced in the liver, they are acute phase reactants, with concentrations increasing during acute illness, they also increase in response to estrogen exposure.
| Age | TSH a (μU/mL) | T4 b (μg/dL) | TBG b (mg/dL) | Tg b (ng/mL) |
|---|---|---|---|---|
| Cord blood | 1–20 | 6.6–15 | 0.8–5.2 | 15–101 |
| 1–7 days | 1–39 | 11–22 | 0.8–5.2 | 1–110 |
| 1–4 weeks | 0.5–6.5 | 8.2–17 | 0.6–5 | 11–92 |
| 1–12 months | 0.5–6.5 | 5.9–16 | 1.6–3.6 | 12–113 |
| 1–5 years | 0.6–8 | 7.3–15 | 1.4–2.8 | 5–72 |
| 6–10 years | 0.6–8 | 6.4–13 | 1.4–2.8 | 3–40 |
| 11–15 years | 0.6–8 | 5.5–12 | 1.4–2.8 | 3–40 |
| 16–20 years | 0.5–6 | 4.2–12 | 1.4–2.8 | 2– 36 |
| 21–50 years | 0.5–6 | 4.3 –12 | 1.2–2.6 | 2–35 |
| T3 a (ng/dL) | rT3 a (ng/dL) | Free T4 b (ng/dL) | Free T3 b (pg/mL) | |
|---|---|---|---|---|
| Cord blood | 14–86 | 100–501 | 1.2–2.2 | — |
| 4–7 days | 36–316 | 34–258 | 2.2–5.3 | 1.3–6.1 |
| 1–4 weeks | 105–345 | 26–290 | 0.9–2.3 | 2.2–8 |
| 1–12 months | 105–245 | 11–129 | 0.8–2.1 | 2.5–7 |
| 1–5 years | 105–269 | 15–71 | 0.8–2 | 2.8–5.2 |
| 6–10 years | 94–241 | 17–79 | 0.8–2 | 2.8–5.2 |
| 11–15 years | 83–213 | 19–88 | 0.8–2 | 2.9–5.6 |
| 16–20 years | 80–210 | 25–80 | 0.8–2 | 2.4–5 |
| 21–50 years | 70–204 | 30–80 | 0.9–2.5 | 2.4– 4.4 |
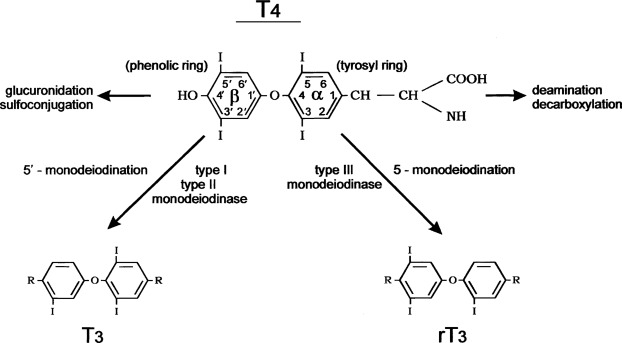
Conversion of T4 to T3 involves the deiodination of T4 ( Fig. 13.2 ). Monodeiodination of the beta or outer ring by monodeiodinase (MD) type II produces T3. Monodeiodination of the alpha or inner ring produces reverse T3 (rT3), which is inactive metabolically. Under normal circumstances, T3 and rT3 are produced at similar rates. About 70% to 90% of circulating T3 is derived from peripheral conversion of T4, and 10% to 30% of circulating T3 is from the thyroid gland. Reflecting the age-related changes in the hormones that regulate T4 stability, the clearance of T4 generally decreases from infancy to adulthood ( Table 13.3 ).
| Thyroxine Kinetic Parameter | Children (3–9 years) | Adolescents (10–16 years) | Adults (23–26 years) |
|---|---|---|---|
| Half-life (d) | 5 (0.13) | 6 (0.35) | 6.7 (0.30) |
| Fractional clearance b | 0.14 (0.005) | 0.12 (0.008) | 0.11 (0.004) |
| Distribution volume (L/kg) | 0.16 | 0.16 | 0.12 |
| (0.008) | (0.014) | (0.005) | |
| Thyroxine turnover (μg/kg/day) | 1.9 (0.09) | 1.5 (0.07) | 1.1 (0.06) |
Regulation of Thyroid Function
The production of T4 and T3 within the thyroid gland is regulated by the thyroid-stimulating hormone ([TSH]; also called thyrotropin ), which is released from the anterior pituitary gland ( Fig. 13.3 ). TSH receptors are present on thyroid follicular cells and are G protein–coupled receptors with a large extracellular amino terminus. Mutations of the TSH receptor can result in constitutive activation of the receptor with severe hyperthyroidism, whereas inactivating mutations result in TSH unresponsiveness and hence hypothyroidism.
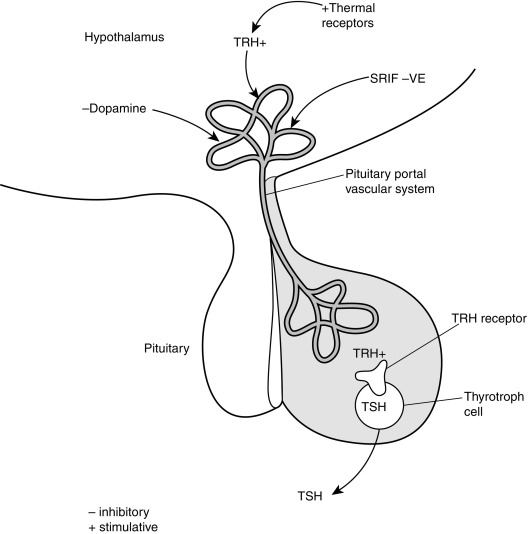
TSH receptor activation stimulates adenylate cyclase accumulation within follicular cells, which in turn causes accumulation of cyclic adenosine monophosphate (cAMP). Increased cellular concentrations of cAMP promote iodide trapping, iodotyrosine synthesis, thyroglobulin (TG) synthesis, and hormone release.
TSH release is regulated by the hypothalamic hormone thyrotropin-releasing hormone (TRH) (see Fig. 13.3 ). This peptide hormone is produced in medial neurons of the paraventricular nucleus of the hypothalamus and is released into the portal circulation of the pituitary gland. Several different neurotransmitters have been observed to influence TRH release.
In addition to normal regulation of TSH receptor activity by TSH, thyroid function can be adversely affected by antibodies that can either stimulate or block TSH action. TSH receptor–stimulating immunoglobulin antibodies (TSIs), or thyroid receptor antibodies (TRAbs), are present in the circulation of individuals with Graves disease (GD) and activate TSH receptors. Conversely, TSH receptor–blocking immunoglobulins (TBIs) antagonize TSH action and can lead to hypothyroidism.
Clinical and biochemical assessment of thyroid status
Clinical Evaluation of the Thyroid
Thyroid disease can present with overt symptoms, insidiously, or with isolated thyromegaly. Thus evaluation of the thyroid gland should be included in the routine examination of children. The thyroid gland can be visualized by having the patient look to the ceiling and swallow. As the thyroid moves, the margins of the gland are viewed to estimate size and symmetry. The thyroid should be palpated to assess size, consistency, and symmetry. This can be performed with the clinician standing behind the patient and palpating the neck with the fingertips. The texture of the thyroid can be assessed to determine if it is smooth or irregular and if nodules are present, which may feel firm or soft. If any asymmetry or abnormal thyroid fullness is noted, ultrasonographic evaluation is recommended because pathological thyroid nodules may feel like normal tissue.
To assess gland size, one may estimate the size of each thyroid lobe relative to that of a teaspoon (5 g) or a tablespoon (15 g). In general, until the end of puberty, gland size (in grams) approximates the patient’s age in years times 0.5 to 0.7. Thus each thyroid lobe of a 10-year-old child is approximately one-half of a teaspoon for a total gland size of 5 to 7 g. For teens and adults, each lobe of the thyroid may reach one teaspoon in size for an approximate total gland size of about 10 g.
In newborns and young infants, the thyroid can be examined by placing the infant supine on the parent’s lap, with the head toward the parent’s knees. The head can then be gently lowered backward to expose the neck, which facilitates thyroid palpation. If the examiner can palpate each ring of the trachea from the sternal notch to above the larynx, then the absence of pretracheal thyroid tissue is suggested, which is observed. Such an absence occurs in cases of failure of thyroid formation or migration. Failure to detect pretracheal thyroid tissue in older children warrants visual examination of the base of the tongue for ectopic thyroid tissue.
When a sublingual thyroid gland is discovered late in childhood or in adolescence, the tissue should be palpated with a gloved finger, during regular office visits, because nodules and malignancies may develop in ectopic thyroid glands. In contrast, when an ectopic thyroid is detected in infancy and replacement therapy is started, the residual thyroid tissue becomes atrophic and does not present long-term problems.
Biochemical Evaluation of Thyroid Function
Approximately 97% of the thyroid hormone released from the thyroid gland is T4. After its release, less than 1% of T4 remains free. The rest of the thyroid hormone circulates bound to the proteins thyroglobulin (TBG; 70%), prealbumin (transthyretin; 10%), and albumin (15% to 20%) . T3 is also released from the thyroid and is generated peripherally. Although T4 constitutes the bulk of circulating thyroid hormone, T3 has far greater affinity for nuclear TRs and exerts most of the potent cellular effects of thyroid hormone action.
Thyroid function can be assessed by measurement of total T4 and total T3 levels, along with indices that reflect thyroid hormone-binding proteins (T3 or T4 resin uptake). The levels of estimated free (unbound) T4 (FT4) are measured to assess thyroid hormone status, without the confounding influences of carrier proteins.
Several conditions are seen in which thyroid hormone levels are abnormal, yet the individual is euthyroid. Because of their confusing nature, these conditions may result in the patient being erroneously diagnosed or treated for hypothyroidism or hyperthyroidism.
When FT4 values are normal, yet total T4 values are high, familial dysalbuminemic hyperthyroxinemia needs to be considered. This autosomal dominant disorder is most commonly seen in Hispanic individuals and can be diagnosed by thyroid hormone-binding protein electrophoresis. If FT4 values are normal but total T4 values are low, the possibility of TBG deficiency must be entertained. TBG deficiency is an X-linked disorder that may be associated with color blindness. In these and other conditions affecting thyroid hormone binding, treatment is not needed and the patient should be educated about the condition to avoid treatment by unsuspecting practitioners.
T4 is much more abundant in the circulation and T3 is the more metabolically active thyroid hormone. T3 is produced peripherally from T4 and is also secreted by the thyroid. A metabolically inactive form of T3′, reverse T3′, is also produced, and its level is elevated in conditions, such as euthyroid sick syndrome, or nonthyroidal illness syndrome (see Fig. 13.2 ).
It is important to consider that estimated FT4 values may not be accurate in infants because of the fact that circulating levels of TBG are elevated, confounding biochemical assessment in this age group. FT4 levels determined by equilibrium dialysis assay, though, are accurate.
Ultrasensitive thyrotropin or TSH assays have been developed, and assessment of TSH has greatly improved the evaluation of thyroid status. TSH levels help to distinguish many thyroid disorders that present with either low or high T4 levels in most cases. TSH values within the normal range for the assay are indicative of a euthyroid state if the hypothalamic pituitary axis is intact. Elevations of TSH generally indicate primary thyroid dysfunction; suppressed TSH values indicate hyperthyroidism. When both FT4 and TSH levels are elevated, TSH-producing pituitary adenomas and thyroid hormone resistance need to be considered.
Hyperthyroidism is distinguished from subclinical hyperthyroidism, a condition in which levels of T4, FT4, and T3 are normal, but TSH levels are suppressed in the absence of thyroid overt disease. The causes of subclinical hyperthyroidism are similar to those of overt hyperthyroidism. Thus it is important to reevaluate individuals with isolated suppression of TSH levels every 3 to 6 months, until the clinical situation declares itself.
Critical in the interpretation of TSH levels in children is recognition that the normative range differs from that of adults (see Box 13.1 ), which is defined by an upper limit values of about 4 μU/mL or less. In comprehensive studies of this issue, the upper limit of TSH values in healthy children and adolescents without thyroid disease is about 7 μU/mL. The application of adult reference range to children thus results in the erroneous diagnosis of subclinical hypothyroidism and results in the unnecessary referral of children for subspecialty care by primary care providers.
Hypothyroidism
Disorders of the thyroid lead to hypothyroidism much more commonly than to hyperthyroidism. Hypothyroidism may be present at birth, acquired during childhood or adolescence, present with or without symptoms, or present gradually or acutely.
It is commonly believed by the public and many practitioners that hypothyroidism is associated with and is a cause of obesity, yet there is little support for the notion that the hypothyroid state contributes to obesity. It is also important to note that TSH levels are slightly higher in obese individuals than nonobese individuals. With weight loss, TSH levels normalize in these children. Thus slight elevations in TSH in obese individuals are physiological and do not warrant therapy.
Hypothyroidism may be elusive, with symptoms elicited only in retrospect. In the extreme, hypothyroidism can be associated with cold intolerance, bradycardia, carotenemia, coarse and brittle hair, dry skin, pallor, and myxedema. These symptoms may not be distressing, which allows prolonged hypothyroidism to escape detection.
The most common causes of hypothyroidism in children are autoimmune processes resulting in Hashimoto thyroiditis. Autoimmune thyroiditis also leads to juvenile acquired hypothyroidism that can present with growth failure when chronically present. Hypothyroidism in children can be caused by iodine exposure or hypothalamic pituitary dysfunction. Other causes of hypothyroidism include exogenous goitrogens, cystinosis, acute thyroiditis, and thyroid irradiation during cancer treatment. Hypothyroidism in the newborn is a serious health concern and is detected by newborn screening programs.
Hashimoto or Autoimmune Thyroiditis
Autoimmune thyroiditis with thyroid enlargement is one of the most common presentations of childhood thyroid disease. It is associated with antibodies against thyroglobulin and thyroperoxidase and is characterized by lymphocytic infiltration of the thyroid gland, which results in thyromegaly. Depending on the nature of the antithyroid antibodies, Hashimoto disease may be associated with a euthyroid state, hypothyroidism, or transient hyperthyroidism.
Hashimoto thyroiditis may rarely occur in very young infants, and typically presents in adolescents, affecting females more than males. The thyroid gland is usually diffusely enlarged and may have an irregular, cobblestone feel. Asymmetric thyroid enlargement, mimicking a thyroid nodule, may be noted. The presence of antithyroid antibodies and the absence of nodules on ultrasonography can distinguish inflammation from other pathologic processes.
Importantly, the presence of antithyroid antibodies does not portend the development of complete or partial thyroid failure that will warrant therapy. In the healthy adult population, up to 5% of individuals had circulating antithyroid antibodies that are present. Less than 10% of these individuals will develop hypothyroidism, with those having elevated antithyroperoxidase (anti-TPO) antibodies being much more at risk than those having anti-TG antibodies.
In children, the incidence of antithyroid antibodies at a population level is not generally known. Of those children with antithyroid antibodies, about 20% are reported to develop hypothyroidism needing therapy. These children often have very high antithyroid antibody titers. Thus if a child is found to have low levels of antithyroid antibodies, it is reasonable to assess thyroid indices every 6 to 12 months and initiate therapy when the TSH rises above the upper limit of normal for children. However, if high titers are present at presentation, it is reasonable to initiate therapy at that point.
Untreated in some children, Hashimoto thyroiditis can result in progressive thyromegaly and hypothyroidism. Treatment with levothyroxine prevents hypothyroidism and the TSH elevations that stimulate gland enlargement. When T4 levels are modestly depressed (> 5 μ/dL) or normal, treatment can be initiated with 1 to 2 mcg/kg/day of levothyroxine. If profound hypothyroidism is present, pseudotumor cerebri may develop when children are treated with conventional doses. Thus treatment is often initiated with one-third to one-half of the usual dose of levothyroxine. After 2 to 4 weeks, the patient can be advanced to conventional doses. However, children with profound hypothyroidism can develop pseudo tumor cerebri, even if treatment is initiated with low doses of levothyroxine.
Interestingly, it has been recently reported that severe hypothyroidism in children with TSH elevations > 500 μU/mL, experience complete resolution of the hypothyroid state. Based on the experience of others, this happenstance is very uncommon.
Although it has been reported that there are some differences in the oral bioavailability of different levothyroxine preparations, from a practical vantage, these differences are small. Thus the routine use of less expensive generic compounds versus more expensive brand-name products is justified.
The timing of levothyroxine ingestion has been the subject of study. Taking the medication at bedtime is associated with higher T4 levels and lower TSH levels over the course of the day. This is believed to be related to better gastrointestinal absorption in the evening than day.
The suggestion has also been made that hypothyroidism in adolescents can be treated with a single dose given weekly. This approach is not recommended, however, because thyroid hormone levels are high, shortly after the dose is administered, and are low by the week’s end. Treatment of congenital hypothyroidism with weekly doses of levothyroxine also can result in mental retardation. It is also recognized that excessive soy intake can interfere with the absorption of levothyroxine.
Of note, other potential therapies that may theoretically alter the autoimmune process have been tested. No proven benefit has been observed in patients who have taken selenium.
Hashitoxicosis
Uncommonly, patients may present with Hashitoxicosis, in which immunologic destruction of thyroid tissue results in the release of preformed thyroid hormone, which leads to elevated T4 levels. In contrast to GD, hyperthyroidism is transient, eye findings are absent, radionuclide uptake is low, and elevated levels of thyroid-stimulating immunoglobulins are not present.
Hashimoto thyroiditis may be associated with other autoimmune diseases, including diabetes mellitus, adrenal insufficiency, vitiligo, and hypoparathyroidism. Autoimmune thyroiditis is also seen in patients with inflammatory bowel disease and juvenile arthritis. Annual surveillance of thyroid gland size and TSH levels should thus be considered for children with other autoimmune problems, and clinicians should be vigilant for signs of hyperthyroidism or hypothyroidism. Conversely, children with autoimmune thyroiditis should be observed for signs of diabetes mellitus and Addison disease.
The incidence of celiac disease in the setting of Hashimoto thyroiditis is about 1%. If patients manifest abdominal discomfort, weight loss, or gastrointestinal symptoms, Celiac disease screening should be performed, but it does not need to be done routinely in children with thyroid disease. We have found a 1% incidence of autoimmune liver disease in children with autoimmune thyroid disease. Because such liver disease can be occult, we annually assess circulating transaminase levels, and if levels are elevated, evaluation of possible liver disease is initiated.
Several groups of children are at risk for autoimmune thyroiditis. Because girls with Turner syndrome are predisposed to autoimmune thyroiditis, TSH levels should be assessed annually. Turner syndrome should also be considered in girls with hypothyroidism, especially if the child is prepubertal at presentations. Children with Down syndrome also warrant annual screening for hypothyroidism.
Subclinical Hypothyroidism
Subclinical hypothyroidism refers to a condition in which circulating T4 and T3 levels are normal, but TSH levels are elevated. As noted earlier, many children are erroneously diagnosed with this condition when TSH levels are found to be elevated relative to adult reference range values. However, if proper pediatric-based TSH levels are applied, the vast majority of children so diagnosed will not have hypothyroidism. Thus some experts have questioned if subclinical hypothyroidism is a real entity in children.
Studies of children with mild TSH elevations (5–10 μU/mL) reveal that only a small fraction will progress to TSH elevations over 10 μU/mL. Data also show that treatment of children with TSH levels 5 to 10 μU/mL does not exhibit somatic or other benefits when treated with levothyroxine. Thus treatment of children with TSH levels lower than 10 μU/mL is not needed. For children with TSH levels over 10 μU/mL, treatment with low doses of levothyroxine is indicated.
Juvenile Acquired Hypothyroidism
When autoimmune thyroiditis occurs during childhood, it is referred to as juvenile acquired hypothyroidism. In children, severe hypothyroidism can be well tolerated. Thus prolonged hypothyroidism may not be detected until growth failure occurs.
Because untreated infantile hypothyroidism is associated with mental retardation, the assumption is often made that juvenile hypothyroidism is associated with learning problems and poor academic performance. This notion is not correct, as children with juvenile hypothyroidism can be successful academically and do not manifest overt learning problems related to the hypothyroid state, nor cognitive impairment.
Children with severe hypothyroidism may manifest cold intolerance, decreased frequency of bowel movements, and decreased physical activity. Bradycardia, facial puffiness, delayed reflexes, and carotenemia may be present. In comparison with Hashimoto thyroiditis, the thyroid gland is either small or only modestly enlarged. Antithyroid antibodies are usually present. These patients are generally not obese, and body mass index values are similar before and after treatment. The development of slipped capital femoral epiphyses may antedate the detection of hypothyroidism.
Some children with juvenile hypothyroidism may present with signs of puberty without pubic hair. Boys may present with testicular enlargement and girls may present with menarche, with or without breast development. With treatment of the hypothyroid state, these characteristics may regress. Available evidence suggests that the hypothyroid state leads to increased gonadotropin secretion, which triggers gonadal activity. In some children, puberty may develop within a year or two of treatment onset, which may limit catch-up growth.
Juvenile hypothyroidism may not be recognized until a sizable statural deficit is present, and the lost height is usually not completely recovered. Children with juvenile hypothyroidism who present with growth failure manifest very low T4 values that are often less than 2 mcg/dL, and profoundly elevated TSH levels that are higher than 250 μU/mL. Hypercholesterolemia and anemia may be present.
The magnitude of the height deficit is proportional to the duration of hypothyroidism, which can be estimated as the difference between the chronologic and bone ages. When the individual is treated with conventional doses of levothyroxine, accelerated skeletal maturation is observed, with the skeletal age advancing disproportionately faster than gains in height. Thus predicted heights fall, and generic growth potential is not achieved.
Because of the poor outcomes of patients with hypothyroidism, we have treated these patients with low doses of levothyroxine (0.25–0.5 mcg/kg/day; e.g., 50 mcg for a 10-year-old). Interestingly, we find that, with low-dose levothyroxine therapy, T4 values normalize (6–7 mcg/dL) within 2 months, and TSH levels normalize or remain only modestly elevated. When serial bone age determinations have been made, we have not observed the disproportionate advancement of skeletal age seen with conventional therapy. However, we do not know if this approach leads to more favorable height outcomes. Some have also suggested that treating these children with gonadotropin-releasing hormone analogs will lead to improved long-term growth. Yet, we have found that catch-up growth slows markedly in some hypothyroid children receiving gonadotropin-releasing hormone analog therapy and predicted adult heights fall and others have not observed added benefit. Because the loss in adult height is proportional to the duration of hypothyroidism, early detection of this disorder is the best intervention for preventing statural deficits.
Iodine-Induced Hypothyroidism
Iodine is a trace clement that is essential for thyroid hormone formation (see Fig. 13.1 ). Recommended dietary iodine intake is approximately 8 pg/kg, or 100 to 150 pg/day, for adolescents and adults. Although modest iodine intake is essential for thyroid function, high-level iodine exposure results in an acute block in the release of preformed thyroid hormone and impaired thyroid hormone synthesis, a phenomenon referred to as the Wolff-Chaikoff effect . When iodine-induced hypothyroidism is suspected, it can be diagnosed by the detection of high iodine levels in urine samples.
In children, iodine can be absorbed through the skin, and iodine-induced hypothyroidism has been observed after cutaneous iodine or betadine use. We have also observed iodine-induced suppression of thyroid hormone production in children with central intravenous lines, when regular cleansing of the insertion site with iodine was included in central line care. Neonatal hypothyroidism has also been associated with maternal povidone iodine exposure at the time of delivery.
In preterm infants, iodine-induced hypothyroidism warrants special attention, because the suggestion has been made that cutaneous iodine exposure is a major cause of hypothyroidism in premature infants. Fortunately, studies show that iodine-induced hypothyroidism is uncommon in the United States.
Significant iodine exposure also occurs from amiodarone, an antiarrhythmic drug that contains 37% iodine. Hypothyroidism occurs in 10% of individuals treated with this compound. Amiodarone can also reach the fetus by transplacental passage and induce fetal hypothyroidism.
In addition to iodine excess, iodine deficiency also leads to hypothyroidism. Estimates are that more than 1 billion people worldwide are at risk for iodine deficiency. Clinically, iodine deficiency is associated with goiter, hypothyroidism, and endemic cretinism.
In the United States, geographic areas of iodine deficiency exist. With the prevalent use of iodized salt, however, the incidence of iodine deficiency has been markedly reduced, and hypothyroidism and goiter caused by iodine deficiency are rare. Of note, the iodine intake in the United States has declined over the past decade, an issue that may have future clinical implications. In Australia, reduction in iodine intake has been recently observed, with potential implications for pregnant and lactating women. The exclusive use of deiodized salt, which includes sea salt, is thus not recommended.
Hypothalamic-Pituitary Dysfunction
Central hypothyroidism should be considered in children with a history of head trauma, brain tumors, meningitis, central nervous system irradiation, or congenital nervous system malformations. Central hypothyroidism has also been associated with the use of retinoid X receptor-selective ligands in the treatment of lymphomas.
In contrast to primary hypothyroidism, the diagnosis of hypothyroidism, secondary to hypothalamic-pituitary dysfunction, may be difficult to establish. Often, circulating levels of T4 are in the low-normal range, and TSH levels may be low, normal, or elevated. FT4 values, however, are usually low.
Whereas congenital central hypothyroidism will be diagnosed in states that perform T4 screening of newborns, neonatal screening programs that rely on TSH determinations will not detect this condition. Central hypothyroidism should therefore be suspected in infants with cholestasis, poor growth, hypoglycemia, structural nervous system problems, or pituitary insufficiency. When neonatal T4 values are interpreted, care should also be taken to use infant thyroid hormone values for comparison, because infantile T4 levels are higher than those seen in adults (see Tables 13.1 and 13.2 ).
Importantly, up to 30% of children who will develop central hypothyroidism may have normal T4 and TSH levels at birth. Thus all children with evidence of hypopituitarism should be regularly monitored for the onset of central hypothyroidism.
When central hypothyroidism is suspected, the TRH test helps distinguish pituitary (secondary) and hypothalamic (tertiary) hypothyroidism. Central nervous system imaging should also be performed to look for congenital malformations or hypothalamic-pituitary lesions. Care should be taken to search for other pituitary hormone deficiencies, especially abnormalities of the hypothalamic-pituitary adrenal and growth hormone axes.
Treatment consists of replacement therapy with levothyroxine. Interestingly, some children with central hypothyroidism require doses lower than those used to treat primary hypothyroidism. Because TSH values are not helpful in guiding treatment, measurement of FT4 levels is recommended. One recommendation is to use a dose 1.6 mcg/kg body weight. This dose was found to be associated with improved markers commonly associated with central hypothyroidism and suggests that levothyroxine dosage, based on body weight, and aiming for a FT4 level in the upper reference range, is superior to titration of levothyroxine dose, aiming at middle normal FT4 concentrations in these patients.
It is important to consider that children with central hypothyroidism may also have other endocrine deficiencies. These individuals have been shown to be at increased risk for sudden death.
Giant Hemangiomas
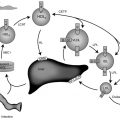
Hypothyroidism has been associated with giant hemangiomas. In some infantile hemangiomas, the endothelium of these vascular structures produces type 3 iodothyronine deiodinase, which degrades circulating T4 (see Fig. 13.2 ).Treatment of hypothyroidism (consumptive) in this setting requires high doses of levothyroxine.
Hypothyroidism in Cancer Survivors
It is well recognized that children who are cancer survivors, who have had head and neck irradiation, are at increased risk for differentiated thyroid cancer. More common, though, is the development of mild hypothyroidism. Up to 30% of children who have had head and neck irradiation will develop primary hypothyroidism. Thus annual TSH screening is suggested. In addition, ultrasound studies are recommended, beginning 5 years after radiation exposure. Practitioners who argue that palpation alone is sufficient for the follow-up of individuals who have had head and neck irradiation need to recognize that ultrasonography will detect thyroid nodules well before palpation.
Thyroid hormone resistance
Thyroid hormones exert their effects by binding to specific nuclear receptors to regulate cellular gene expression. When the TR is mutated, impaired tissue responsiveness results, leading to thyroid enlargement, elevated levels of T4 and T3, tachycardia, and behavioral problems. Unlike GD, TSH levels are normal or slightly elevated.
The most common forms of thyroid hormone resistance are caused by mutations of the TR beta gene. More than 100 mutations have been identified that result in impaired affinity for T3. Mutant TRs also block the function of normal TRs. Thus thyroid hormone resistance is a dominant-negative mutation, and inheritance is autosomal dominant. Detection of thyroid hormone resistance in the index case may therefore lead to diagnosis of the condition in other family members. In up to 50% of children with thyroid hormone resistance, the mutations are spontaneous.
Most individuals with resistance to thyroid hormone have generalized thyroid hormone resistance. These individuals are eumetabolic and asymptomatic, with TSH levels in the normal range.
In contrast, some individuals have isolated pituitary thyroid hormone resistance. These individuals have symptoms of hyperthyroidism, because they are sensitive to the effects of increased thyroid hormone levels. Resistance to thyroid hormone can also be associated with central nervous system problems. Approximately 50% of individuals with resistance to thyroid hormone have attention deficit hyperactivity disorder, and a minority have mental retardation.
Because individuals compensate for thyroid hormone resistance by secreting more thyroid hormone, treatment is generally not necessary. However, patients with thyroid hormone resistance may be improperly diagnosed as having GD and undergo ablation of the thyroid. In this situation, replacement therapy with large doses of exogenous thyroid hormone is needed. With the earlier recognition of resistance to thyroid hormone because of newborn screening, the issue of whether children with resistance to thyroid hormone should be treated prenatally or during infancy has been raised. Treatment is generally reserved for infants who show elevated TSH levels, growth failure, seizures, and developmental delay.
In some cases, TSH secretion may be profound, leading to massive thyromegaly, which may adversely impact upper airway function. These cases are generally associated with severe loss of function mutations. Treatment with high doses of T3 every other day has been shown to be somewhat effective in this setting. In other cases, thyroidectomy is needed to prevent airway compromise.
Hyperthyroidism
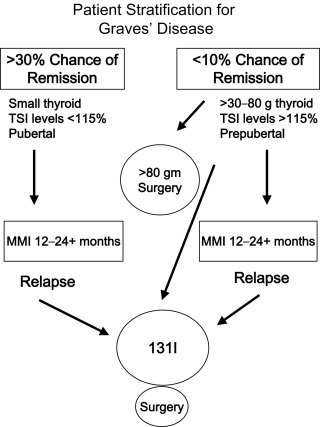
Hyperthyroidism has profound influences on the fetus, neonate, growing child, and adolescent, including physical and behavioral effects. In children, thyrotoxicosis is often present for extended periods before recognition, contributing to significant health problems. A number of conditions result in hyperthyroidism in childhood (see Table 13.1 ), yet GD is the most common cause, and thus will be the major focus of this chapter with treatment approaches stratified ( Fig. 13.4 ).
Other causes of hyperthyroidism in children include autonomously functioning thyroid nodules, Hashitoxicosis, neonatal thyrotoxicosis, and infections of the thyroid. Hyperthyroidism also results from thyroid hormone ingestion, McCune–Albright syndrome, struma ovarii, and TSH-producing pituitary adenomas. Epidemic hyperthyroidism has also been seen when thyroid tissue has been inadvertently included in meat products. Several conditions can masquerade as hyperthyroidism, including thyroid hormone resistance and thyroid hormone-binding protein disorders, yet are best left untreated.
Clinical Evaluation of the Hyperthyroid State
Pediatric thyroid disease can present with overt symptoms, silently, or with isolated thyromegaly. When hyperthyroidism is suspected during childhood, the practitioner should look for somatic signs (see Table 13.2 ). One of the universal features of hyperthyroidism is tachycardia. In the absence of such, it is unlikely that hyperthyroidism is present. Other clinical features can include a prominent stare and proptosis, although eye findings occur less commonly in children and adults. It is the common perception that children presenting with hyperthyroidism present thin with weight loss, yet this is often not the situation.
Graves Disease
GD is the most common cause of hyperthyroidism in children and affects 1 in 10,000 children. Interestingly, the incidence of GD in children may be increasing. An autoimmune disorder, GD is caused by thyroid gland stimulation by TRAbs or TSI, and involves genetic factors.
Hyperthyroidism can exert profound adverse effects on children, including excessive physical activity, tremor, tachycardia, flushing, palpitations, weight loss, accelerated linear growth, reduced bone mineralization, and poor school performance. In comparison with adults, eye disease occurs in the minority of pediatric patients with GD, and when it occurs, is usually mild.
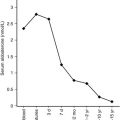
Over the past several years, additional outcome data have become available to complement older studies looking at spontaneous remission rates of children with GD. Collectively, these studies show that the majority of pediatric patients with GD will not undergo spontaneous remission, even after many years of antithyroid drug (ATD) therapy (see Table 13.3 ). Thus most pediatric patients will require either radioactive iodine (RAI, 131 I) or surgery.
Antithyroid Drug Therapy
ATDs act by inhibiting oxidation and organic binding of thyroid iodide to impair thyroid hormone production and include methimazole (MMI) and propylthiouracil (PTU). MMI is 10- to 20-fold more potent than PTU and has a longer half-life. Importantly, these medications do not cure the hyperthyroid state, rather they palliate the condition, until spontaneous remission occurs or definitive therapy is rendered.
Each of these medications is associated with adverse events that must be considered when prescribed. Before the initiation of drug therapy, a back-up plan that takes into account the patient’s age and treatment risks must be developed at therapy onset in the event that a toxic reaction occurs. Failure to initially consider alternative treatments can result in a crisis when adverse effects occur.
Because it takes 1 or 2 months until biochemical hyperthyroidism resolves on drug therapy, treatment with beta-blockers (propranolol, atenolol, or metoprolol), can be used to control GD symptoms. Focusing on symptom control with beta-blockers also alleviates the false perceived need for initial high-dose ATD therapy. Because atenolol does not cross the blood barrier as much as propranolol, patients will not feel as tired on this medication and thus is preferred. For individuals who have reactive airway disease, the cardiac selective beta-blocker metoprolol may be used.
In 2008 Rivkees brought to public attention a number of serious complications associated with PTU therapy in children. PTU-induced liver injury occurs in up to 1 in 2000 children, occurs rapidly, and is often irreversible. Serial monitoring of transaminase levels in a child on PTU is not viewed as useful in reducing the hepatotoxicity risk. Thus the only way to reduce the risks of PTU-related hepatotoxicity is to avoid the use of this medication.
PTU, though, may be needed in special circumstances. These conditions include situations when neither prompt 131 I or surgical treatment are options in a patient who has had a toxic reaction to MMI and ATD medication is necessary. In this setting, PTU use should be short term.
If PTU is prescribed, patients and guardians must be informed of the risk of liver failure and to be alert for signs and symptoms of liver abnormalities, including pruritus, jaundice, anorexia, light colored stools, dark urine, and abdominal pain. If these symptoms develop, the patient should immediately stop the medication, a physician contacted, and laboratory tests obtained to evaluate hepatic function and transaminase levels.
MMI is now the drug choice for hyperthyroidism. The MMI doses described in published reports range from 0.1 to 1.0 mg/kg/day. However, one does not need to use high doses at treatment onset, as MMI side effects are in part dose related. The response to ATDs influencing circulating thyroid hormone levels is not instantaneous, and several months are needed for thyroid hormone levels to normalize. Thyroid function tests should be obtained monthly after therapy onset. After T4 levels become normal, in most cases the MMI dose can be cut by half to maintain euthyroidism. Although MMI is often prescribed in divided doses over the day, once-a-day dosing is sufficient, and is associated with better compliance than multiple daily doses.
MMI is available in 5, 10, and 20 mg tablets. When used in children, the following doses that are fractions of tablets can be used: infants, 1.25 mg/day; 1 to 5 years, 2.5 to 5.0 mg/day; 5 to 10 years, 5 to10 mg/day; and 10 to 18 years, 10 to 20 mg/day. Because the hyperthyroid state can be associated with low white cell counts, and patients will be treated with a medication that can depress neutrophil levels, one should obtain a complete blood count at therapy onset.
MMI therapy is not without risks. Minor side effects may affect up to 20% of children, and major side effects may occur in 1% of children. The most common minor adverse side effects related to MMI are hives, arthralgia, and neutropenia. Children may also develop major side effects, including agranulocytosis, Stevens-Johnson syndrome, and vasculitis. MMI adverse events most commonly occur within 6 months of therapy onset. Yet, children may develop adverse events more than 12 months after treatment onset.
Agranulocytosis is a potential serious ATD adverse event and occurs in 0.3% of adults taking PTU or MMI. The agranulocytosis risk is dose dependent and is rare. If an individual receiving MMI feels ill, becomes febrile, or develops pharyngitis, MMI should be stopped immediately, a practitioner contacted, and a complete blood cell count obtained.
Note that in the MMI-only era that we are now in, reports of MMI-associated agranulocytosis have recently been published. Similar to the monitoring of adverse events associated with PTU, it is important that adverse events related to MMI be monitored and brought to the attention of the US Food and Drug Administration by practitioners via the MedWatch program ( https://www.fda.gov/Safety/MedWatch/ ).
Antithyroid Drug Therapy Duration and Prediction of Remission
Based on considerable evidence, prolonged ATD therapy will not result in an increased chance of remission for most children, whereas in a minority of children it may. As such, with the initial evaluation of a child with GD, practitioners should attempt to stratify children into two groups: children with a 30% to 40% chance of remission with prolonged ATD therapy, and children with a slim chance of spontaneous remission.
Data shows that one can distinguish patients with a greater likelihood of remission from those with a much lower chance, following prolonged ATD therapy. The chance of remission after years of ATDs will be low if the thyroid gland is large (> 2 times normal size for age), the child is young (< 12 years), not Caucasian, serum TRAb/TSI levels are elevated, or the patient presents with profound hyperthyroidism at presentation (FT 4 > 4 ng/dL).
Prospective studies in adults show that if remission does not occur after 12 to 18 months of ATD therapy, it is unlikely remission will occur with prolonged therapy. In the pediatric population, published data show that when ATDs are used for 1 to 2 years, remission rates are 15% to 30%, and possibly up to 40% in some children. Please note that remission is defined as being either euthyroid or hypothyroid for 1 year or more after cessation of therapy.
In adults, assessment of TRAb or TSI levels is useful in determining disease course and remission likelihood. This issue has been less studied in children. Consistent with the notion that GD will remit in only a small proportion of children, TRAb levels normalize after 24 months in only 18% of pediatric patients on ATDs. TRAb levels thus persist longer in children than in adults. There are no data showing normalization of TRAb levels in pediatric patients on ATDs for longer.
For children with unfavorable risk factors for spontaneous remission at treatment onset, it is reasonable to treat children for up to 2 years with MMI and see if spontaneous remission occurs. At that point, if there is no remission, it is appropriate to move on to definitive therapy if desired by the family. Alternatively, treatment for longer periods can be considered, as long as side effects to medication do not occur. This approach may be especially useful if the child is considered too young for surgery or RAI. For the child with favorable risk factors for remission, if spontaneous remission has not occurred after the 2 years of ATDs, continuation of antithyroid medication for prolonged periods is also acceptable, yet one needs to be attentive to adverse effects.
Strategies for the Child With an Adverse Event to Methimazole
With MMI being the drug of choice for treatment, and the risk of hepatotoxicity being present with the use of PTU, practitioners may face a clinical dilemma of what to do when a patient taking MMI develops an adverse reaction. There are few published reports addressing this issue in children.
For children who develop allergic symptomatology, antihistamine medications may be attempted. However, this approach is generally not effective.
If an individual develops neutropenia, agranulocytosis, or hepatotoxicity, the antithyroid medication must be discontinued immediately. Because the hyperthyroid state will resume quickly, and it is best not to treat patients with RAI or surgery, while profoundly hyperthyroid, therapy to control the hyperthyroid state, until definitive therapy must be initiated within days. If a patient will be treated with RAI, beta blockade should be initiated and treatment rendered within the week the ATD is stopped to prevent recurrence of profound hyperthyroidism. This approach requires a coordinated effort with nuclear medicine experts, as soon as the decision is made to discontinue the ATD.
If the individual is viewed as being too hyperthyroid for treatment with RAI, practitioners may consider the short-term use of PTU to control the hyperthyroid state, until the patient is viewed as stable enough for treatment with RAI. Because PTU may also be associated with agranulocytosis, if PTU is used, white blood counts need to be very closely followed.
If the plan is surgical intervention, iodine drops may be started immediately after MMI is discontinued and continued for at least 2 weeks. This period will allow the hyperthyroid state to resolve and allow adequate time for surgical planning.
Radioactive Iodine Therapy
The goal for 131 I therapy for GD is to induce hypothyroidism. There are several important caveats related to the use of RAI in children. RAI should not be given to cause euthyroidism in children, as this results in partially irradiated residual thyroid tissue that may be associated with a higher risk of thyroid neoplasm than the normal population.
It has been suggested that dosages delivering 30,000 to 40,000 centiGray (cGy) (rad) to the thyroid are necessary to ablate the thyroid gland. But, dosages delivering 10,000 to 20,000 cGy to the thyroid are more often used and result in partial or complete destruction of the thyroid. Typically, administered thyroid doses of 150 μCi/g (5.5 megaBecquerel [MBq]/g) generate radiation doses of 12,000 cGy to the thyroid.
Dosing is based on the Quimby-Marinelli equation: Dose (β + γ radiation; in Gy) = 90 × [oral 131 I dose (microcurie [μCi]) × oral 24-hour uptake (%)/g × 100%]. For example, if the desired dosage is 300 μCi/g, and the thyroid is 30 g with an uptake of 75% (0.75), the desired administered dosage will be 12 mCi. (Dosage in mCi = 300 μCi/g × 30 g/0.75 uptake = 12,000 μCi or 12 mCi).
Some centers give a fixed administered dosage of 10 or 15 mCi 131 I to all children, rather than individually calculated administered activation. There are no studies comparing outcomes of fixed doses versus calculated doses in children. In adults, the two different approaches lead to similar outcomes; however, in children, a potential advantage of calculated versus fixed dosing, is that it is possible to use lower dosages of 131 I if the administered dose is calculated.
When children are to be treated with 131 I, ATDs should be stopped 3 to 5 days before treatment. Patients are placed on beta-blockers until T4 and/or FT4 levels normalize posttherapy. Whereas some clinicians restart ATDs after treatment with 131 I, this is rarely required in children. Thyroid hormone levels begin to decrease about 7 days after radioiodine therapy in children. Continued ATD use can make it difficult to assess if posttreatment hypothyroidism is the result of 131 I or the ATD.
Side effects of 131 I therapy are unusual. Less than 10% of children will complain of mild tenderness over the thyroid in the first week after 131 I therapy. This problem can be treated with either acetaminophen or nonsteroidal antiinflammatory agents for 24 to 48 hours
There are rare reports of children with severe hyperthyroidism developing thyroid storm after 131 I. In general, these children were severely hyperthyroid when 131 I was rendered. Thus if T4 levels are over 20 mcg/dL or FT4 levels are greater than 5 ng/dL, children should be treated with MMI until T4 and/or free T4 levels normalize, before proceeding with 131 I therapy. Importantly, most children with GD have been hyperthyroid for months before diagnosis; there is no need to rush to 131 I therapy.
It usually takes 6 to 12 weeks after 131 I treatment for the patient to become biochemically euthyroid or hypothyroid. Until then, symptoms of hyperthyroidism can be controlled using beta-blockers. The use of potassium iodide or Lugol solution, 1 week after 131 I, will also quickly attenuate biochemical hyperthyroidism, without adversely affecting the outcome of radioiodine therapy.
Several studies have reported the details of 131 I therapy for childhood GD. Children as young as 1 years old have been treated with 131 I with excellent results. But, treatment of such young children is not common, nor is now recommended. 131 I dosages in children and teenagers have ranged from 100 to 400 μCi/g of thyroid tissue. Similar to that found in adults, responses to 131 I therapy are related to gland size and dose. Some 25% to 40% of children treated with 50 to 100 μCi of 131 I per gram of thyroid tissue are hyperthyroid several years after therapy. In children treated with 150 to 200 μCi of 131 I per gram of thyroid, hyperthyroidism remains in 5% to 20%, and 60% to 90% become hypothyroid.
The development of progression of ophthalmopathy following 131 I in adults has been reported. However, children rarely develop severe ophthalmopathy and proptosis is mild. Studies show that eye disease worsens in only a small percentage of children with GD, irrespective of therapy type.
In adults, it has been shown that progression of ophthalmopathy can be prevented by treatment with prednisone for 3 months following 131 I therapy. Adjunctive prednisone therapy is not routinely recommended for the majority of children, as most do not have significant eye disease. The prolonged administration of prednisone is also associated with growth failure, weight gain, and immune suppression. Nevertheless, prednisone (0.5 mg/kg × 4–6 weeks) may be useful for the child who has moderate or severe eye disease and will be treated with 131 I.
Risks of Radioactive Iodine in Children Treated for Graves Disease
There is no evidence showing adverse effects to offspring of children treated with 131 I. Birth defects are not higher in offspring born to individuals treated with 131 I for hyperthyroidism during childhood or adolescence. In addition, the rates of birth defects are not higher in children treated with 80 to 700 mCi of 131 I for thyroid cancer, which are dosages that are much higher than those used for GD.
The thyroid gland is unique in its developmental sensitivity to malignancy after low-level radiation exposure. There is an increased risk of thyroid cancer in individuals less than 20 years of age at the time of low-level thyroid irradiation. In contrast, individuals who are older than 20 years of age do not exhibit an increased risk of thyroid cancer when exposed to low-level thyroid irradiation.
The risk of thyroid neoplasms in children is greatest with exposure to low-level external radiation (0.1–25 Gy; ~ 0.09–30 μCi/g), and not with the higher dosages used to treat GD. At present, we are not aware of any cases of thyroid cancer that developed in pediatric patients treated with more than 150 μCi of 131 I per gram of thyroid tissue for childhood GD that can be attributed to 131 I therapy.
Important in considering RAI use in children, is the potential influences of 131 I therapy on other cancers, as 131 I therapy results in low-level, whole-body radiation exposure. Several studies in adults have examined potential risks of 131 I therapy for GD on cancers. These studies have not revealed increased mortality or increased rates of cancer following 131 I for GD.
In comparison with studies in adults, few studies have focused on outcomes of 131 I therapy for childhood GD. The most extensive study of pediatric patients involved 36-year outcomes of patients who were less than 20 years old when treated with 131 I therapy for GD. There was no evidence for increase cancer risk in this population. Yet, this sample size was modest.
The total-body radiation dose after 131 I varies with age, and the same absolute dose of 131 I will result in more radiation exposure in a young child than in an adolescent or adult. Currently, we do not have dosimetry data on 131 I use in pediatric patients with GD to assess total-body exposure in pediatric patients. Based on phantom modeling, it is estimated that at 0, 1, 5, 10, 15 years, and adulthood, respective total-body radiation doses will be 11.1, 4.6, 2.4 1.45, 0.90, and 0.85 Roentgen equivalent man (rem) (0.01 Sievert [Sv]) per millicurie of 131 I administered. Based on the Biologic Effects of Ionizing Radiation Committee V (BEIR VII) analysis of low-level, acute exposure to radiation, theoretical lifetime attributable risk of cancer mortality and all cancer incidence can be projected. Based on these theoretical calculations, we feel that it is prudent to avoid RAI therapy in children under 5 years of age and to avoid more than 10 mCi in patients less than 10 years old.
Thyroidectomy and Risks
Surgery is an effective form of therapy for GD if an expert surgeon can perform it, and in some situations it is preferable to RAI. When surgery is performed, near-total or total thyroidectomy is indicated, as subtotal thyroidectomy is associated with a higher relapse rate. Hypothyroidism is nearly universal in children and adults who undergo total thyroidectomy. In comparison, after subtotal thyroidectomy, hyperthyroidism recurs in 10% to 15% of patients.
Surgery is preferred in children younger than 5 years when definitive therapy is needed and can be performed by a skilled thyroid surgeon. In individuals who have large thyroid glands (> 80 g), the response to 131 I is poor, and surgery is recommended for these patients.
In preparation for surgery, the patient should be rendered euthyroid. Typically, this is done by continuing MMI until T4 levels normalize. A week before surgery, iodine drops are started (1–3 drops, three times per day), which inhibits thyroid hormone production and causes the gland to become firm and less vascular.
Postoperatively, younger pediatric patients are at a higher risk for transient hypoparathyroidism than adolescents or adults. To mitigate postoperative hypocalcemia, we treat children with 0.5 mcg of calcitriol, twice a day, for 3 days before surgery. Postoperatively, the calcitriol is weaned over 15 days (0.5 mcg twice a day × 5 days; 0.5 mcg every day × 5 days; 0.5 mcg every other day × 5 days). Using this approach, only 5% of patients require postoperative calcium infusions versus 40% of patients without preoperative treatment.
Acute complications that follow thyroidectomy include hemorrhage, hypocalcemia, and recurrent laryngeal nerve paresis. In children, rates from 0 to 6 years were 22%; from 7 to 12 years, 11%; and from 13 to 17 years, 11%. These rates are higher than those observed in adults.
Complication rates are related to the expertise of the surgeon. Considering these data, if local pediatric thyroid surgery expertise is unavailable, referral of a child with GD to a high-volume, thyroid surgery center with pediatric experience should be considered. Very low complication rates for children undergoing the thyroidectomies for GD have been reported with this type of multidisciplinary model.
Following our model, there is the recent trend in the development of pediatric thyroid centers across the United States. These centers often have experts in pediatric thyroid surgery.
Somatic Complications of Graves Disease
In addition to focusing on the thyroid-related aspects of GD, somatic considerations are important as well. Individuals with GD have an increased basal metabolic rate and increased caloric needs. This may result in altered dietary patterns, resulting in increased caloric intake. After correction of the hyperthyroid state, it is recognized that body mass index will increase, which can be dramatic in some individuals. As such, anticipatory guidance and monitoring of body weight after implementation of antithyroid therapy is important.
GD is also associated with increased bone turnover, which can lead to osteopenia in affected individuals. GD has thus been associated with an increased fracture risk.
Thyrotoxicosis can trigger thyrotoxic periodic paralysis, which is a life-threatening condition associated with profound hypokalemia. This condition is most frequently observed in Asian men and is associated with hypokalemia, which affects the lower extremities and is secondary to thyrotoxicosis. The condition results from an intracellular shift of potassium induced by the thyroid hormone sensitization of Na + /K + -adenosine triphosphatase and is associated with mutations of the inwardly rectifying potassium channel Kir2.6.
Another potential concern related to the resolution of GD is the development of acute myopathy. This has been sporadically reported in children following correction of the hyperthyroid state, resulting in large increases in circulating muscle enzymes as hyperthyroid state transitions to hypothyroidism. Following treatment, individuals presenting with muscle cramps should be evaluated for this possibility through the monitoring of creatine phosphokinase levels.
Autoimmune thyroiditis may be associated with other autoimmune diseases, including diabetes mellitus, adrenal insufficiency, vitiligo, and hypoparathyroidism. Autoimmune thyroiditis is also seen in patients with inflammatory bowel disease and juvenile arthritis. Annual surveillance of thyroid gland size and TSH levels should thus be considered for children with other autoimmune problems, and clinicians should be vigilant for signs of hyperthyroidism or hypothyroidism. Conversely, children with autoimmune thyroiditis should be observed for signs of diabetes mellitus and Addison disease.
The incidence of celiac disease in the setting of autoimmune thyroiditis is about 1%. If patients manifest abdominal discomfort, weight loss, or gastrointestinal symptoms, Celiac disease screening should be performed, but it does not need to be done routinely in children with thyroid disease. We have found a 1% incidence of autoimmune liver disease in children with autoimmune thyroid disease. Because such liver disease can be occult, we annually assess circulating transaminase levels, and if levels are elevated, evaluation of possible liver disease is initiated.
Several groups of children are at risk for autoimmune thyroiditis and GD. Because girls with Turner syndrome are predisposed to autoimmune thyroiditis, TSH levels should be assessed annually. Turner syndrome should also be considered in girls with hypothyroidism, especially if the child is prepubertal at presentation. Children with Down syndrome also warrant annual screening for thyroid dysfunction.
Neonatal thyrotoxicosis
Thyrotoxicosis in the neonate is a severe and life-threatening condition that can be associated with lasting neurologic problems. Neonatal thyrotoxicosis most commonly occurs in the setting of active or past maternal GD. If a mother has GD, the chance is 1 in 80 that TRAbs will be transferred to the fetus, resulting in intrauterine or neonatal hyperthyroidism. Rarely neonatal thyrotoxicosis will persist, like the GD disease seen in older children. In other rare cases, persistent neonatal thyrotoxicosis is caused by activation of the TSH receptor.
The fetal thyroid gland is responsive to maternal TRAbs, which, if present at elevated levels, may result in hyperthyroidism. Fetal hyperthyroidism manifests during the second half of gestation, as transfer of TRAbs from the mother to the fetus increases with progression of pregnancy.
The risk of fetal hyperthyroidism and neonatal GD is proportional to the magnitude of elevation of TRAb levels. Fetal hyperthyroidism is generally associated with levels of TRAbs more than 2- to 4-fold greater than the upper limit of normal for assay. Because the fetus is at risk for hyperthyroidism when there is active or past maternal GD, fetal growth and heart rate should be regularly assessed from mid-pregnancy onward. Excessive fetal heart rate (> 160 beats per minute after 20 weeks) and the presence of a fetal goiter suggest hyperthyroidism in the fetus. In addition, accelerated maturation of the femoral ossification center is seen with fetal hyperthyroidism.
If a mother with GD is taking antithyroid medications during pregnancy, fetal thyroid hormone synthesis will be inhibited, which will prevent the development of intrauterine hyperthyroidism. However, the infant may be born with a goiter and hypothyroidism. At birth, circulating levels of T4 may be low and TSH levels elevated. In most cases, the effects of ATDs wane, and thyroid function normalizes within a week. If significant transplacental passage of TRAbs has occurred, however, thyrotoxicosis will develop.
If a mother with a history of GD is not taking ATDs during pregnancy, the fetus may develop intrauterine hyperthyroidism. If the condition is not recognized, it may result in profound intrauterine thyrotoxicosis and growth retardation. Such infants have prematurely fused cranial sutures, advanced skeletal age, long-term learning problems, and mental retardation. If fetal hyperthyroidism is recognized prenatally by the presence of fetal tachycardia (heart rate > 160 beats per minute after 22 weeks), treatment of the mother with ATDs will reduce intrauterine thyrotoxicosis.
Treatment of thyrotoxic infants consists of administration of MMI (1.25 mg per day) and beta-blockers (propranolol 1 mg per kg per day). Lugol solution or saturated potassium iodide may be given (1–2 drops every 8 hours) for 7 to 10 days to more rapidly control biochemical hyperthyroidism. After approximately 2 weeks of ATD therapy, thyroid hormone levels will decline. When thyroid hormone levels fall below normal, supplementary levothyroxine (37.5 mcg/day for full-term infants) is added to prevent hypothyroidism. As TRAbs are cleared from the infant’s circulation, spontaneous recovery begins within 3 months and is usually complete by 6 months. Thus the infant can be weaned from treatment after 3 months. Monitoring of the infant’s TRAb levels is also a useful predictor of when antithyroid medication can be tapered.
Other causes of hyperthryoidism
Activating Mutations of the Thyroid-Stimulating Hormone Receptor
Several mutations of the TSH receptor have been reported that resulted in ligand-independent receptor activation and hyperthyroidism. Affected individuals present with hyperthyroidism in the neonatal period with potential serious adverse effects on the child including advanced skeletal maturation and craniosynostosis, impaired growth, and neurologic impairment. This diagnosis should be considered in the young child presenting with hyperthyroidism and bone age advancement, in the absence of TSI elevation. The hyperthyroid state may be controlled with ATDs, yet, either RAI if surgery is needed. Because hyperthyroidism in the neonatal and early childhood can be associated with abnormal growth and maturation and neurologic abnormalities, it is essential that the hyperthyroid state be exquisitely controlled in young children.
McCune–Albright Syndrome
McCune–Albright syndrome is an inherited disorder characterized by spontaneous activation of the G protein alpha subunit. Because the TSH receptor is G protein–coupled, somatic rotations of the G protein alpha subunit within the thyroid gland may result in hyperthyroidism. Children with McCune–Albright syndrome may be recognized with a distinctive pattern of café au lait spots with a ragged border, and/or history of polyostotic or monostotic fibrous dysplasia. Because there is clonal activation in the thyroid gland, which results in areas of increased thyroid proliferation and activity, and other regions that will not be affected, similar to the treatment of isolated hyperfunctioning thyroid nodules, definitive treatment by surgery is recommended.
Thyroid-Stimulating Hormone Producing Adenomas
Thyrotropinomas are rare in adults and exceedingly rare in children. Such tumors are typically macroadenomas presenting with the signs of hyperthyroidism and occasional central nervous system symptoms that may include visual field loss and headaches. Such individuals will have elevated levels of circulating thyroid hormones, but without a suppressed TSH.
Transsphenoidal surgery is the preferred treatment and adjunctive radiotherapy may be needed. In adults, successful treatment with octreotide has been reported. It is important to distinguish this condition from isolated central thyroid hormone resistance, which can present with similar biochemical features. Distinguishing features include the presence of a pituitary mass with TSH-producing tumors, the lack of response to TRH stimulation, and elevated levels of the a-glycoprotein subunit, in pituitary adenomas.
Infectious Bacterial Thyroiditis
Occasionally, a child presents with hyperthyroidism, tenderness over the thyroid gland, and fever caused by bacterial infection of the thyroid, a condition called acute thyroiditis . Acute thyroiditis can be associated with the presence of a fistula connecting the piriform sinus on the left side of the pharynx to the thyroid. Fevers can be high, and erythrocyte sedimentation rates and white counts elevated. Ultrasonography may reveal a local abscess. In contrast to GD, uptake of technetium 99-pertechnetate or radioiodine is reduced when thyroid scanning is performed.
The offending bacteria include Haemophilus influenza and group A streptococci. Thus treatment with an antibiotic resistant to disruption by β-lactamase is recommended. In severe cases, hospitalization and intravenous antibiotic administration is indicated, because lymphatic drainage into the mediastinal region may occur. Surgical drainage is needed if a localized abscess develops and the response to antibiotics is poor.
Because the infectious process results in destruction of thyroid tissue, release of preformed thyroid hormone and hyperthyroidism may occur during infection. The hyperthyroid state is usually transient, and treatment with ATDs is not indicated. If the patient becomes symptomatic, beta-blockers may be used.
After the child has recovered, pharyngography is indicated to test for a patent piriform sinus tract. Occasionally, the tract may close as the result of the infection. If the tract persists, however, and acute thyroiditis recurs, resection is needed.
Subacute Thyroiditis
Viral infections of the thyroid may occur and result in subacute thyroiditis. In comparison with acute thyroiditis, subacute thyroiditis may be less severe. Fever, thyroid tenderness, and hyperthyroidism may be present and may last for several weeks. Because clinically distinguishing between bacterial and viral thyroid infections is difficult, antibiotic treatment is indicated when infectious thyroiditis is suspected.
Hyperfunctioning Nodules
Warm or hot nodules lead to excessive production of thyroid hormone and can be associated with clinical and biochemical hyperthyroidism. Interestingly, activating mutations of the TSH receptor and G s have been discovered in hyperfunctioning nodules. Although hyperfunctioning nodules may be ablated with radioiodine, surgical excision of hyperfunctioning nodules is recommended in children and adolescents, because radiation-exposed normal thyroid tissue will remain after the hyperfunctioning nodule is ablated. Although the risk of malignancy in hyperfunctioning nodules is low, thyroid cancers have been described in warm nodules.
Toxic Multinodular Goiters
Multinodular goiters are uncommon in children, but patients with this condition can develop thyrotoxicosis, which is usually related to the time the goiter has been present and goiter size. In this setting, hyperthyroidism develops when a single nodule in the thyroid, becomes overly active, and functions autonomously. Some 46% of patients may have T3 thyrotoxicosis, and nodules are 3 cm or more in diameter.
In adults, 131 I is routinely used in the treatment of isolated toxic adenomas and toxic multinodular goiters. The use of radioiodine to treat these conditions in children is uncommon, and few follow-up data are available. Although strong justification exists for the use of radioiodine in the treatment of childhood GD, especially when appropriate doses are used, we recommend that radioiodine be avoided in children with toxic adenomas or multinodular goiters. When a toxic nodule is present, either as an isolated nodule or in the setting of a multinodular goiter, thyroid function is suppressed in the nontoxic regions. When radioiodine is given, uptake will be limited to the autonomously functioning tissue, and if large doses are administered, the remaining thyroid tissue will receive external irradiation. Because the risk of thyroid cancer following external radiation is very low after 20 years of age, the use of radioiodine for toxic nodule ablation in adults is not associated with increased thyroid cancer risks. In the child or adolescent treated with 131 I for toxic nodules, however, low-level irradiation to the remaining thyroid tissue will occur with an increased thyroid cancer risk.
Amiodarone
Amiodarone is a class III antiarrhythmic medication that is associated with both hyper- and hypothyroidism caused by amiodarone’s high iodine content and direct toxic effect on the thyroid. Two types of amiodarone-induced thyrotoxicosis (AIT) have been described. In type I AIT, excess iodine results in enhanced thyroid hormone production. Type II AIT, is more common than type I and is a destructive thyroiditis that results in direct toxic effect of amiodarone on thyroid follicular epithelial cells in excess release of T4 and T3, without increased hormone synthesis. The treatment of this condition involves glucocorticoids.
Excessive Iodine Intake
Iodine is a micronutrient and typical intake is 150 mg/day for adults. In general, excessive iodine intake does not result in thyroid disease. However, in some individuals with iodine deficiency who are predisposed to getting GD, excess iodine can trigger hyperthyroidism, a condition referred to as the Jod–Basedow effect .
Thyroid nodules and thyroid cancer
Background
Thyroid nodules in children and adolescents are less common when compared with adults; however, the risk of malignancy for a nodule diagnosed in a pediatric patient is approximately twofold higher. Over the past 2 decades, there has been a 2% to 4% per year increase in the number of patients diagnosed with thyroid nodules and differentiated thyroid cancer (DTC), with the largest increase noted in females between 15 and 19 years of age, where thyroid cancer accounts for 8% of all cancers. Similar to adults, over 85% of pediatric thyroid cancers are papillary thyroid cancer (PTC), with the remainder divided between follicular thyroid carcinoma (FTC) and medullary thyroid cancer (MTC), the latter most commonly associated with multiple endocrine neoplasia type 2 (MEN2).
Thyroid nodules may be detected by physical examination or by incidental discovery during unrelated head and neck radiologic imaging. When a thyroid nodule is detected, serum TSH and a thyroid and neck ultrasound (US) should be obtained to determine the best plan for management. If the TSH is suppressed, a radionuclide scan may be performed to identify a hyperfunctioning nodule, a lesion associated with a lower risk for malignancy. Serum calcitonin levels are not routinely assessed; however, calcitonin immunohistochemistry or a calcitonin washout from the fine needle aspiration (FNA) needle should be considered based on a family history of MEN2, physical examination concerns for MEN2B, or cytologic findings consistent with MTC.
Evaluation of a Thyroid Nodule
US is the most efficient and accurate method for determining if a thyroid nodule may be surveilled or should undergo further evaluation with FNA. US characteristics of nodules that should be assessed, included in the radiology report, and used to stratify nodules for FNA per the Thyroid Imaging and Reporting Data System (TI-RADS) include composition (cystic, spongiform, mixed or solid), echogenicity, (anechoic, hyperechoic, isoechoic, or hypoechoic), shape on transverse imaging (taller than wide or wider than tall), margin (smooth, ill-defined, lobulated, or with extrathyroidal extension), and echogenic foci (none, comet-tail, macrocalcifications, rim calcifications, or punctate calcifications) ( Fig. 13.5 ). Cystic or mixed composition, with a greater than 75% cystic component, is the single, most reliable feature associated with a lower risk of malignancy. Features associated with a higher risk of malignancy include solid composition, hypoechogenicity, taller than wide shape on transverse imaging, lobulated or irregular margin, and punctate echogenic foci. US assessment of the lateral neck must be performed on all patients found to have an indeterminate or suspicious thyroid nodule to determine if there is lymphadenopathy concerning thyroid cancer metastasis. US features that are most predictive of an abnormal lymph node include rounded shape, punctate echogenic foci, and the presence of peripheral vascularity. Size is generally not a predictive feature for malignancy, and lymph nodes in the upper neck (level II) are typically larger than lymph nodes in the lower (levels III and IV) or posterior (level V) regions of the neck (see Fig. 13.6 ).