76.1
Introduction
Teriparatide (TPTD), the aminoterminal (1-34)parathyroid hormone (PTH) peptide, is a proremodeling anabolic agent. When administered by daily subcutaneous injection, it first stimulates bone formation (primarily modeling based) and then bone remodeling, with a net increase in bone formation throughout administration . As a result, TPTD produces substantial increments in skeletal mass and the growth of new bone tissue permits restoration of bone microarchitecture, including improved cancellous architecture and cortical thickness . Abaloparatide (ABL), approved for osteoporosis treatment in the United States in April 2017, also delivered by daily subcutaneous injection, is a synthetic analog of PTH-related peptide (PTHrP). Like TPTD, ABL acts through the PTH1 receptor (PTH1R), however, the two agents have differential affinities for different conformations of PTH1R that provides an underlying scientific basis for some of the clinical differences between these agents. Preclinical data suggest that ABL, such as TPTD, improves both bone mass and microscopic bone structure . Both TPTD and ABL provide rapid protection against vertebral and nonvertebral fractures within 18–19 months and improve spine and hip bone mineral density (BMD), though increments in the spine are much larger . These agents are thus well suited to those with severe osteoporosis at high risk of fracture, particularly those at high imminent risk . This chapter highlights the most important recent clinical studies with TPTD, overviews clinical data with ABL, and provides a rationale for early proactive use of anabolic treatment for osteoporosis in patients at high risk of fracture.
76.2
Teriparatide monotherapy in postmenopausal women with osteoporosis
The pivotal trial that investigated the fracture outcomes of TPTD enrolled 1637 women with severe osteoporosis, all of whom had a prevalent vertebral fractures (mean >2 fractures per patient) and mean spine T -score −2.6. Participants were randomized to either 20 or 40 µg TPTD or placebo daily . Although BMD increased more with the 40 µg dose, fracture outcomes were not superior and hypercalcemia was more common with 40 µg daily; therefore the 20 µg TPTD dose was Food and Drug Administration (FDA) approved for osteoporosis treatment (in 2002). After a median treatment period of 19 months, TPTD 20 µg had increased BMD of the spine by 9.7%, femoral neck (FN) by 2.8% and total hip (TH) by 2.6% (all P <.001) but decreased radius BMD [−2.1%, not significant (NS)]. Vertebral fracture risk reduction with TPTD was 65% [95% confidence interval (CI) relative risk 0.22–0.55], and height loss was reduced in patients with incident vertebral fractures. Incident nonvertebral fractures were reduced by 35% for all, and by 50% for those defined as fragility fractures (relative hazard 95% CI 0.25–0.88). Despite the decline in radius BMD, there were fewer wrist fractures in TPTD-treated women (for all wrist fractures, 13 with placebo and 7 with TPTD and for those considered fragility wrist fractures, 7 with placebo and 2 with TPTD). There were four hip fractures in the placebo group and two in the TPTD group (too few to evaluate statistically). Longer duration of TPTD treatment (≥14 months) was associated with greater reduction in nonvertebral fracture incidence and reduced back pain . TPTD was associated with a 20% decline in median serum levels of 25-hydroxyvitamin D and a 20% increase in serum levels of 1,25-dihydroxyvitamin D .
There was a low incidence of hypercalcemia, and a small increase in mean 24-hour urine calcium and serum uric acid, without clinical sequelae. Adverse events included limb pain, dizziness, and leg cramps. Overall new cancer diagnoses were less frequent with TPTD versus placebo . In rodents, long-term high-dose TPTD produced osteogenic sarcoma, dependent on dose, and duration of administration . In over 15 years of postmarketing experience and several long-term surveillance studies in the United States and Europe, there has been no evidence of an association between TPTD exposure and osteosarcoma .
76.2.1
Trials comparing teriparatide with antiresorptive agents
TPTD has been compared to several antiresorptive agents, including risedronate, alendronate, zoledronic acid, and denosumab, with study sizes large enough to observe fracture outcomes in several studies highlighted here. TPTD was compared to active treatment with risedronate (blinded and randomized trial) over 18 months in 710 postmenopausal women with back pain likely due to osteoporosis-related vertebral fractures (mean baseline duration 7–8 months; mean age 71, mean spine T -score −2.7; mean FN T -score −2.4) . Although there was no difference in the primary outcome (proportion of women who experienced reduced back pain at 6 months), the incidence of new vertebral fractures was reduced by >50% in women on TPTD (incidence 4% with TPTD vs 9% with risedronate, P <.01); but there was no difference in nonvertebral fracture incidence. Spine, TH, and FN BMD increased significantly more with TPTD compared with risedronate.
TPTD was compared in a blinded randomized trial to alendronate in 428 women and men with glucocorticoid-induced osteoporosis (mean age 56, 80% women and of those, 80% postmenopausal; mean spine T -score −2.6, mean TH T -score −2) . At 18 months, spine BMD increased 7% with TPTD versus 3% with alendronate, while TH BMD increased about 4% with TPTD versus 2% with alendronate (group differences significant at both skeletal sites). Vertebral fracture incidence was reduced substantially with TPTD (0.6% incidence) compared with alendronate (6.1% incidence; P <.05), but there was no difference in the incidence of nonvertebral fractures.
The VERtebral fracture treatment comparisons in Osteoporotic women (VERO) trial was the first to compare fracture outcomes as a primary end point in patients treated with anabolic compared with antiresorptive treatment . In this trial, 1360 postmenopausal women with severe osteoporosis (mean age 72; mean spine T -score −2.3, mean FN T -score −2.3) were randomized to receive TPTD versus risedronate for 24 months. All women had at least one severe or two moderate vertebral fractures, similar to the criteria used to enroll in the original placebo-controlled trial . Group difference in new vertebral fracture incidence was the primary outcome. Consistent with recruitment criteria, the mean number of a prevalent vertebral fractures was 2.7 per patient; about 90% of patients had at least one severe vertebral fracture, 36% had a clinical vertebral fracture in the year prior to the study, and 44% had a history of nonvertebral fracture. New vertebral fractures occurred in 5% of subjects on TPTD and 12% on risedronate (risk ratio 0.44; 95% CI 0.29–0.68; P <.0001). TPTD produced similar benefits over risedronate for new and worsening vertebral fractures, multiple vertebral fractures and moderate and severe vertebral fractures. Clinical fractures (clinical spine and nonvertebral fractures) were also reduced to 4.8% with TPTD versus 9.8% with risedronate [hazard ratio (HR) 0.48, P <.0009]. Trends were seen for reductions in subject incidence of nonvertebral fractures alone (4% TPTD vs 6% risedronate; HR 0.66, 95% CI 0.39–1.10; P =.1) and major nonvertebral fractures (3% TPTD vs 5% risedronate, HR 0.58, 95% CI 0.32–1.05; P =.06). As a greater number of patients on risedronate had more than one nonvertebral fracture ( n =10) versus those randomized to TPTD ( n =2), the rate ratio for all nonvertebral fractures was significantly reduced with TPTD (HR 0.56, 95% CI 0.35–0.9; P <.02). The numbers of both wrist and hip fractures were both lower with TPTD. This study convincingly demonstrates the greater potential of TPTD (compared with risedronate) to strengthen bone and reduce the occurrence of osteoporosis-related fractures across the skeleton in women with severe osteoporosis. Subgroup analyses showed that superior efficacy with TPTD versus risedronate was independent of age, baseline BMD, severity, and number of prior vertebral fractures, nonvertebral fracture history and glucocorticoid use . Effects were also independent of whether patients had taken prior osteoporosis therapy, including “recent use”; however, “recent use” was defined very liberally as any use >6 months within the preceding 3 years. Therefore this did not separate the true recent users from more remote users of bisphosphonates. Although serum levels of 25OHD declined in the TPTD group and were stable in the risedronate group, the difference in 25OHD levels did not affect relative risk reductions for fractures with TPTD versus risedronate .
A recent metaanalysis assessed limb fracture incidence from 23 randomized clinical trials in which the approved daily TPTD dose (20 µg) was used for at least 6 months (median treatment period 18 months). The control group included both placebo and active comparators . Results showed that TPTD reduced the incidence of hip fracture by about 50% compared to the control group, though there was no difference in the incidence of wrist, forearm, or humerus fractures.
76.2.2
Histologic evaluation of teriparatide effect and comparison with antiresorptive agents
In 51 paired iliac crest biopsy specimens obtained before and after TPTD (including 19 placebo and 32 TPTD patients ), cancellous bone volume, trabecular connectivity, and cortical thickness significantly increased, providing the histologic foundation for the improvement in biomechanical skeletal competence that reduces fracture incidence. A longitudinal biopsy study of men and women receiving TPTD with continued hormone therapy produced similar findings . A recent study of TPTD versus placebo in patients undergoing TH replacement for osteoarthritis showed that TPTD rapidly stimulated bone formation in the FN of the human femur ( Fig. 76.1 ), suggesting a mechanism for improvement in hip strength with TPTD .
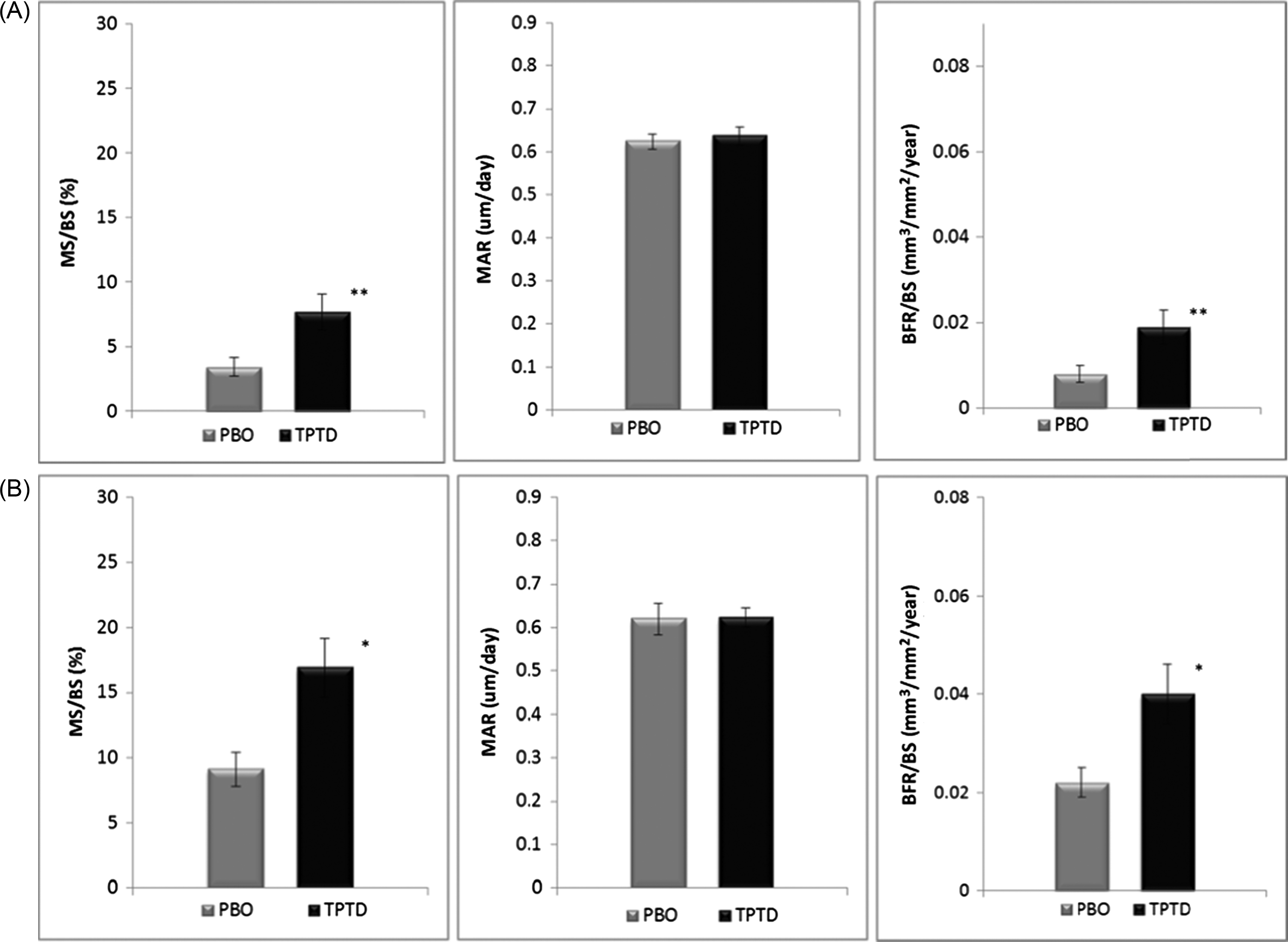
In a series of bone biopsy studies comparing TPTD with antiresorptive medications, bone formation indices were consistently significantly greater with TPTD than alendronate, zoledronic acid, and denosumab during treatment periods from 3 to 24 months . Bone formation was stimulated in all four bone envelopes (cancellous, endocortical, intracortical, and periosteal, though bone formation was much lower on the periosteal surface) , and increased bone formation rates were seen as early as 7 weeks after starting TPTD in cancellous and endocortical envelopes .
Cancellous microstructure was improved in patients who received 24 months of TPTD, regardless of whether they had previously received alendronate therapy or were treatment naïve, and the magnitude of the microstructural improvement was associated with the early increase in serum Propeptide of Type I Procollagen (PINP) level . TPTD administration for 2 years also increased bone formation indices in cortical bone, both in previously untreated women and in women with prior alendronate exposure, but increases in cortical width and thickness were accompanied by increases in cortical porosity in women who were switched from alendronate to TPTD .
76.2.3
Sequential and combination treatment: teriparatide and antiresorptive therapy
Observational studies suggest that BMD is lost in individuals who do not take antiresorptive agents after cessation of TPTD treatment, whereas potent antiresorptives can maintain or induce further BMD gains . After 2 years of TPTD, subsequent treatment with denosumab was more effective than bisphosphonates at improving BMD further, particularly in the spine . In the denosumab and teriparatide administration trial (DATA)-Switch study, patients who received 2 years of TPTD, followed by 2 years of denosumab, had dramatic BMD improvements totaling 18% in the spine and almost 7% in the TH over the 4-year sequential study . BMD increments were also accompanied by increases in spine microstructure, as reflected by trabecular bone score (TBS) increments of about 5% in patients who received TPTD for 2 years followed by denosumab for 2 years . In the observational extension to this study , in women who started antiresorptive therapy (either bisphosphonates or denosumab) promptly (after completing the 4-year study on TPTD or denosumab), BMD gains were maintained. In contrast, women who did not take antiresorptive treatment had rapid BMD decline at all sites; hip BMD losses were most prominent in those who ended the study on denosumab compared with those who ended the study on TPTD.
Although the combination of TPTD and antiresorptive agents could theoretically produce additive effects on bone strength, studies have shown different outcomes based on skeletal site (spine vs hip), type of measurement [Dual X-ray Absorptiometry (DXA) vs quantitative computed tomography (QCT)], specific antiresorptive used, and whether patients are treatment-naïve or treatment-experienced when TPTD was started. Furthermore, in treatment-experienced patients, there are differences in outcome based on whether the prior antiresorptive was continued or stopped when TPTD was initiated . In previously untreated patients the combination of TPTD and zoledronic acid over 1 year increased BMD faster and to a greater extent in the TH compared with TPTD alone (2.3% with combination vs 1.1% with TPTD alone, P <.02), while the effect of spine BMD was similar to that of TPTD alone (7.5% with combination vs 7% with TPTD; NS) . In the DATA trial, women who were randomized to receive the combination of TPTD with denosumab, compared to either agent alone, had both TH and spine BMD gains that were larger than those seen with either agent alone .
Several recent metaanalyses have compared TPTD monotherapy to combination therapy . These metaanalyses are difficult because the protocols, antiresorptive medications used, TPTD doses, and study lengths are heterogeneous. Overall, the findings corroborate the concept that combination TPTD with antiresorptive therapy has a benefit to both TH and FN BMD at least through the first year, with waning effects thereafter .
In general, there are few patients in whom de novo treatment with combined therapy is indicated, because the sequence of TPTD or ABL (see next) followed by potent antiresorptive treatment is highly effective at rapidly reducing fracture rates and building skeletal mass. The most important population in whom to consider combination therapy is in those who have already been on a potent antiresorptive agent. With remote exposure (several years since prior treatment), there is probably not an impaired BMD response to TPTD, while in those who are currently on bisphosphonate or denosumab treatment when TPTD is indicated, the effect on hip BMD is suboptimal; it consistently declines for at least 1 year during TPTD treatment . Although there are no fracture studies large enough to determine if this effect is associated with an increase in the risk of fracture, a surrogate outcome of bone strength (finite element modeling based on QCT data) showed no benefit on hip strength in patients who switched from alendronate to TPTD over 18 months . In contrast, hip strength did increase in patients randomized to add TPTD to ongoing alendronate.
Similarly, but even more dramatically, hip BMD declined precipitously in patients switched from denosumab to TPTD and remained below baseline for the entire 2 years of subsequent TPTD treatment . This may be related to increased cortical remodeling upon cessation of denosumab treatment and further remodeling stimulation with TPTD administration. There has not yet been a specific study evaluating the combination of ongoing denosumab with the addition of TPTD versus switch from denosumab to TPTD. By analogy with the de novo combination TPTD plus denosumab study , as well as observations in women who added TPTD to ongoing alendronate , it is possible that adding TPTD would be a more effective strategy to increase BMD and skeletal strength in patients who are having a suboptimal response to denosumab (such as intercurrent major fracture or decline in BMD).
76.2.4
Rechallenge with teriparatide
Women who received TPTD in addition to ongoing alendronate were followed for a year on continued alendronate alone, during which BMD remained stable. Rechallenge with TPTD produced similar biochemical and BMD changes to those seen during the first TPTD course . In women and men who completed a 2 year TPTD course and 1 year follow-up off therapy, rechallenge with TPTD produced biochemical and BMD responses, but they were of lesser magnitude than those seen with the first TPTD course .
76.2.5
Alternative treatment regimens: weekly and cyclic treatment
Once-weekly TPTD 28.2 µg reduced the risk of incident radiographic vertebral fractures by 68% versus placebo over approximately 1.5 years . Although nonvertebral fracture incidence was also about 50% lower with this low dose once-weekly TPTD, the group difference was not statistically significant. Once-weekly TPTD 56.5 µg over 18 months reduced vertebral fracture incidence by 80% in a population of 578 postmenopausal women and men with a prevalent vertebral fracture . Although nonvertebral fracture incidence was also lower with this higher once-weekly dose TPTD (vs placebo), the reduction did not meet statistical significance.
A recent study compared 3-month TPTD cycles to standard daily TPTD over 24 months in previously untreated women and in those who continued alendronate. In the latter women, results of cyclic treatment were similar to those of daily therapy, despite administration of only half of the cumulative TPTD dose. However, in the women on no antiresorptive therapy, cyclic TPTD treatment did not increase BMD beyond that expected for the cumulative TPTD dose given; in the cyclic group the BMD effect was half that of the daily group, consistent with the half cumulative TPTD dose . Moreover, in an extension to this study, the same cumulative dose of TPTD given cyclically over 4 years did not increase BMD more than standard daily treatment over 2 years in either previously untreated or alendronate experienced women .
Attempts to deliver TPTD by transdermal patch have been largely unsuccessful, with the exception of one formulation. In this one study, BMD gains were similar or better than seen with subcutaneous administration over 6 months, but there have been no longer term or confirmatory data .
76.2.6
Use of teriparatide beyond osteoporosis
Although TPTD is approved only for the treatment of patients with osteoporosis, a large body of animal and clinical data suggests that TPTD could have orthopedic applications beyond the prevention of fractures . Clinical benefits of TPTD have been most clearly demonstrated with spine fusion surgeries but also in some studies of fracture healing (e.g., pelvic fractures), arthroplasty integration, and osteonecrosis of the jaw.
76.3
Abaloparatide treatment for osteoporosis
PTHrP, originally discovered to be the factor responsible for humoral hypercalcemia, was subsequently found to be an important paracrine and autocrine factor produced by multiple tissues. One of its most important physiologic roles is in maternal–fetal development, where it regulates placental calcium transport, mammary gland development and lactation, cartilage and endochondral bone formation, keratinocyte differentiation, vascular smooth muscle cell function, tooth eruption, and pancreatic beta islet function, among other effects. In adults, PTHrP is not a systemic hormone, but is produced by cells of osteoblast lineage, and has an ongoing local role in the regulation of bone remodeling and fracture repair .
Exogenously administered PTHrP produces a potent anabolic effect on bone through binding to the PTH1R on osteoblasts and osteoblast precursors, which stimulates formation, activity, and life span of osteoblasts . Because of these positive preclinical findings, PTHrP was tested in a short-term clinical randomized trial of postmenopausal women with osteoporosis . The effects of two doses of daily PTHrP(1–36) were compared with daily PTH(1–34) over 3 months. The high dose produced substantial hypercalcemia, but the lower PTHrP(1–36) dose resulted in improved BMD, similar to the effect seen with PTH(1–34). Continued pharmacologic development of the native PTHrP peptide was halted in part by concerns about hypercalcemia and peripheral vasodilatory effects.
ABL is a synthetic PTHrP analog, specifically chosen for clinical development among a large number of PTH and PTHrP analogs, for what appeared to be an optimized anabolic profile . With substitutions in the C-terminal region of the molecule, it retains 75% homology with PTHrP(1–34) and 40% homology with PTH1–34 . The amino acid substitutions produce particularly high affinity for the G-protein dependent (RG) subtype of PTH1R but with low affinity for the G-protein independent (R 0 ) conformation (with ABL, affinity for RG is 1600-fold greater than affinity for R 0 , whereas differential affinity for RG versus R 0 with PTHrP is 110-fold greater and with PTH1–34 is 12-fold greater) . These differences appear to result in a similar bone formation stimulus but lesser bone resorption stimulus compared with both PTH and PTHrP.
Consistent with the in vitro observations, in preclinical rodent models, ABL produced a dose-dependent increase in biochemical and histomorphometric markers of bone formation without stimulation of bone resorption and increased trabecular and cortical BMD with improvements in bone strength . In cynomolgus monkeys, ABL treatment increased bone formation, bone density, and bone strength with no apparent effects on bone resorption or serum calcium .
76.3.1
Early human studies with abaloparatide
In the phase 2 study, 222 postmenopausal women with osteoporosis were randomized to receive 20, 40, or 80 µg daily ABL, 20 µg daily TPTD or placebo (Pbo) for 24 weeks . Spine BMD increased with TPTD and dose dependently with ABL. TH BMD increments were greater with the 40 and 80 µg ABL doses (2% and 2.6%, respectively) compared with TPTD (0.5% increase). Bone formation markers increased similarly with ABL and TPTD, whereas the bone resorption marker increased significantly more with TPTD.
In 138 women with evaluable TBS results from this study , TBS was unchanged in the Pbo group (1.1%) but increased significantly with TPTD (2.2%) and all three ABL doses, in a dose/response fashion (2.3%, 3.1%, and 4.2%, respectively). The mean TBS increment with the 80 µg ABL dose (4.2%) was significantly higher than the TBS increment seen with TPTD (2.2%).
76.3.2
Abaloparatide pivotal clinical trial (Abaloparatide Comparator Trial in Vertebral Endpoints)
In the pivotal Abaloparatide Comparator Trial in Vertebral Endpoints (ACTIVE) study, 2463 postmenopausal women with osteoporosis, between the ages of 49 and 86, were randomized to blinded daily subcutaneous ABL 80 µg versus Pbo or open-label TPTD 20 µg for 18 months . Study inclusion for women <65 years of age was based on T -score ≤−2.5 with a prevalent vertebral fracture or history of recent nonvertebral fracture (within the preceding 5 years). Women ≥65 years of age were included with T -score ≤−3, even in the absence of fracture, or T -score up to −2 with prevalent vertebral or recent nonvertebral fracture. At baseline the groups were well matched with mean age 69 years and mean spine, TH, and FN T -scores were −2.9, −1.9, and −2.1, respectively. The population included 24% with a radiographically confirmed vertebral fracture, 31% with a prior nonvertebral fracture and 37% with no prior fracture.
76.3.3
Abaloparatide Comparator Trial in Vertebral Endpoints biochemical marker and BMD results
The marker of bone formation (PINP) increased by 93% at 1 month and remained elevated by approximately 45% above baseline at 18 months. The bone resorption marker Crosslinked C-Telopeptide (CTX) increased more gradually, and the relative increment for CTX was of lower magnitude than seen for PINP (43% increase CTX at 3 months and 20% increase at 18 months compared with baseline levels). The increase in PINP was similar in ABL and TPTD groups at 1 month, but thereafter the PINP increase was larger with TPTD. Increments in CTX were numerically, but not significantly, larger at all-time points with TPTD versus ABL.
At 18 months, spine BMD increments were similar with ABL (11.2%) and TPTD [10.5%; no significant difference (NSD)]. In contrast, BMD increments at the TH and FN were faster and significantly larger with ABL versus TPTD at 6 months (for TH, ABL 2.3% vs TPTD 1.4% and for FN, 1.7% ABL vs 0.9% TPTD). Beyond the first 6 months, TH and FN BMD increased in parallel with both anabolic agents such that BMD remained higher with ABL throughout the drug treatment period. At 18 months, TH BMD had increased 4.2% with ABL versus 3.3% with TPTD ( P <.001), and FN BMD increased 3.6% with ABL versus 2.7% with TPTD ( P <.001) .
76.3.4
Abaloparatide Comparator Trial in Vertebral Endpoints fracture effects
New vertebral fracture incidence (the primary end point) was 4.2% with Pbo, 0.6% with ABL and 0.8% with TPTD, with relative risk reductions of 86% for ABL and 80% for TPTD versus Pbo (both P <.001). Time to first nonvertebral fracture revealed an early separation between ABL and both Pbo and TPTD groups. Over 18 months, nonvertebral fractures occurred in 4.7% of Pbo patients, 2.7% of ABL patients, and 3.3% of TPTD patients. At 18 months, risk reductions were 43% with ABL versus Pbo (relative hazard 95% CI 0.32–1.0; P =.049) and 28% with TPTD versus Pbo (relative hazard 95% CI 0.42–1.22; P =.22). Major osteoporotic fractures were reduced by 70% with ABL (relative hazard 95% CI 0.15–0.61; P <.001) and 33% with TPTD (relative hazard 95% CI 0.39–1.14; P =.14); major osteoporotic fracture incidence was 55% lower with ABL than TPTD (relative hazard 95% CI 0.21–0.95; P =.03) .
Numbers needed to treat were calculated from the pivotal trial for both ABL and TPTD (as the reciprocal of the absolute risk reductions seen with each agent vs Pbo); . To prevent one new vertebral fracture, 28 women would need to be treated with ABL and 30 treated with TPTD. To prevent one new nonvertebral fracture, 55 women would need to be treated with ABL and 92 treated with TPTD.
A responder analysis of BMD outcomes suggested that more women on ABL increased BMD >3% above baseline in spine, TH, and FN compared with either Pbo or TPTD. At 18 months, 45% of ABL, 2% of PBO, and 33% of TPTD-treated participants increased BMD to this extent at all three skeletal sites .
76.3.5
Subgroups and special populations from Abaloparatide Comparator Trial in Vertebral Endpoints
Prespecified evaluations of treatment effects on both fracture and BMD by subgroups of age, baseline BMD, prevalent spine fracture, or prior nonvertebral fracture history revealed no significant qualitative or quantitative interactions . ABL provided protection against fractures throughout the skeleton across a wide variety of ages and baseline risks, suggesting potential utility in a broad group of postmenopausal women with osteoporosis. The effects of ABL on fracture risk reduction were similar across a broad range of baseline fracture risk assessment tool (FRAX) scores, with no interactions between baseline FRAX score and treatment effect . The effects of ABL have been evaluated from the ACTIVE trial in various special populations. In elderly women ≥80 years of age , BMD increments were similar compared to those of the full population. There were similar conclusions with respect to ABL effects in patients with chronic kidney disease , type 2 diabetes mellitus , and in women from different geographical regions .
76.3.6
Effects on the wrist
The effects of ABL and TPTD from ACTIVE on the wrist have been evaluated in detail . The incidence of wrist fracture in the Pbo group was 1.8% (15 women with 17 fractures) and with TPTD was 2.1% (17 women with 17 fractures), compared with 0.8% (7 women with 7 fractures) in the ABL group (risk reduction 51%, P =.11 vs Pbo and 57%, P =.052 vs TPTD). A higher incidence of wrist fracture was seen in all three groups in those women who had a wrist fracture prior to entering the study (compared to those with no wrist fracture history), but relative risk reductions were similar to ABL versus Pbo and TPTD in this group of patients. Consistent with the clinical data in the wrist, by 18 months, ultradistal BMD of the radius had increased with ABL and decreased with both Pbo and TPTD ( P <.01 ABL vs both other groups). In the proximal one-third radius site, BMD declined slightly with both ABL and Pbo (NSD between ABL and Pbo) but declined significantly more with TPTD ( P <.01 vs both other groups).
76.3.7
Abaloparatide Comparator Trial in Vertebral Endpoints transiliac crest bone biopsies
Transiliac crest bone biopsies were obtained between 12 and 18 months in 105 women from the ACTIVE trial ( n =35, 36, and 34 in the Pbo, ABL, and TPTD groups, respectively) . There were surprisingly few differences in structural or dynamic variables among the three groups, including cortical width, wall thickness, cancellous bone volume, trabecular number, width or separation, or bone formation rate. However, the dynamic analyses were limited to cancellous bone, with no dynamic cortical assessments. Furthermore, the biopsies were not paired from participants before and after treatment, limiting the conclusions that could be drawn. In terms of static histomorphometric parameters, eroded surface was lower in the ABL-treated group, and the mineral apposition rate was higher in the TPTD-treated group than in Pbo-treated women. Cortical porosity was higher with both ABL and TPTD than with Pbo. Additional bone biopsy studies are underway to provide further insight on the effects of ABL on cellular activity and skeletal microstructure.
76.3.8
Safety of abaloparatide in Abaloparatide Comparator Trial in Vertebral Endpoints
The numbers of deaths, serious adverse events, and serious cardiovascular events were all similar across groups. Hypercalcemia incidence was 3.4% with ABL, 6.4% with TPTD, and 0.4% with Pbo. Hypercalciuria occurred in 11.3% on ABL, 12.5% on TPTD, and 9% with Pbo. There were more discontinuations (10%) due to adverse events in the ABL group (compared with 6% with Pbo and 7% with TPTD) . Adverse events of dizziness, nausea, headache, and palpitations were all more common with ABL but were usually mild and self-remitting. Some, but not all of these symptoms, were associated with orthostatic hypotension. A blood pressure decline ≥20 mmHg systolic or ≥10 mm diastolic 1 hour after the first dose was reported in 4% of ABL-treated women and 3% of Pbo-treated women . This was generally resolved within 4 hours of the injection. A blood pressure decline of the same magnitude at any point over the course of the ACTIVE study was observed in 17.4% with ABL, 16.4% with Pbo, and 15.5% with TPTD . At 1 hour postdose, heart rate increased on average 7 mmHg in the ABL group, 5.5 mmHg in the TPTD group, and 1.5 mmHg in the Pbo group. The increase in heart rate was generally resolved by 6 hours postdose.
76.4
Abaloparatide Comparator Trial in Vertebral Endpoints extension study
Women in the ABL and Pbo groups who completed the ACTIVE study were offered participation in an extension where all women transitioned to alendronate (Aln; 70 mg orally once weekly) within 1 month of completing ACTIVE and continued Aln for 2 subsequent years. Ninety-two percent of eligible participants enrolled ( n =1139 women of 1243 who completed ACTIVE). The primary end point was the group difference in new vertebral fracture incidence over the cumulative 25 months of ABL or Pbo in ACTIVE and 6 months of Aln in ACTIVE extension (ACTIVExtend) study . Women remained blinded to the original treatment assignment during the first 6 months of ACTIVExtend. During the first 6 months of the ACTIVExtend study, there were seven new vertebral fractures in the group that transitioned from Pbo to Aln (Pbo/Aln) and zero in the group that transitioned from ABL to Aln (ABL/Aln). The overall cumulative rate of vertebral fractures at this primary end point (25 months) was 4.4% for Pbo/Aln and 0.6% for ABL/Aln (87% relative risk reduction, group difference P <.001). Similarly, for nonvertebral fractures, during the first 6 months of Aln in the extension study, there were seven nonvertebral fractures in the Pbo/Aln group and three in the ABL/Aln group, resulting in 25-month cumulative incidence rates of 5.6% versus 2.7%, respectively (52% relative risk reduction, group difference P =.02). BMD increments at 25 months for the spine, TH, and FN were 3.5%, 1.4%, and 0.5% with Pbo/Aln and 12.8%, 5.5%, and 4.5%, respectively, with ABL/Aln (all group differences P <.001). Biochemical marker levels decreased similarly in both groups upon transition to Aln.
Secondary endpoints in ACTIVExtend were determined for the 1005 women who completed the full 43 months of the extension study . At 43 months, in the Pbo/Aln group, the new vertebral fracture rate was 5.6% (32 women had fractures), whereas in the ABL/Aln group, the corresponding cumulative 43 months, new vertebral rate was 0.9% (only 5 women had fractures), resulting in a final 84% relative risk reduction for new vertebral fracture over 43 months (group difference P <.001). Also at 43 months, there was a 39% relative risk reduction for nonvertebral fractures in the ABL/Aln group versus Pbo/Aln ( P <.05). In a combined intention-to-treat analysis of all individuals who enrolled in the ACTIVE and ACTIVExtend studies, there were five hip fractures in those who started with Pbo and zero hip fractures in those who started on ABL, resulting in a significant reduction in hip fracture occurrence ( P <.027).
76.5
Clinical issues
Although the ACTIVE study excluded patients who had exposure to bisphosphonates within the preceding 5 years, many patients who might benefit from ABL are those who are currently on osteoporosis medication. For TPTD, BMD and strength effects are different in patients who transition from bisphosphonates to TPTD versus those who take TPTD as initial therapy . A key question is whether this same difference in response will be seen in patients who transition from a bisphosphonate to ABL. Of even greater importance is to determine the optimal strategy for utilizing ABL in women who are currently on denosumab and who remain at high risk (including those with incident fractures on treatment, actively declining BMD, or persistently very low BMD). The effects of ABL upon transition from denosumab might be more positive than those seen after transition from denosumab to TPTD because ABL is somewhat less proresorptive. However, it is not at all clear whether there will be any major benefit, especially to the cortical predominant regions of the skeleton, with this transition. The possible use of ABL combined with continued antiresorptive treatment should be explored in these patients. Although the ACTIVExtend trial utilized Aln after ABL treatment, there is no rationale for limiting antiresorptive therapy after ABL to Aln. The antiresorptive agent used after ABL should be based on individual patient factors and a reassessment of patient risk.
One practical advantage of ABL is its stability at room temperature; the delivery pen contains 1 month of medication and does not require refrigeration after beginning use. The effectiveness of ABL treatment in men is currently being studied. In addition, the study of a transdermal delivery system is underway and may extend the use of this compound to a much larger target population compared with subcutaneous daily delivery.
76.6
Which patients are appropriate for teriparatide and abaloparatide treatment?
A prior history of fracture is the most important determinant of future risk of fracture; however, imminent risk is particularly high in patients with recent fracture. Patients who have suffered a recent clinical fracture have a 10% risk of sustaining another fracture in the very next year and an 18% risk over the ensuing 2 years . For women who sustain an incident radiographic vertebral fracture, subsequent vertebral fractures occur in 20% in the very next year . Patients with a history of multiple fractures are also at particularly high risk of fracture . In order to comprehensively evaluate an individual’s future fracture risk, vertebral imaging must be performed to diagnose vertebral fractures since most of these fractures occur initially without prominent symptoms. In a recent National Health and Nutrition Examination Survey (NHANES) study, using the strictest criteria for diagnosis of vertebral fracture through dual-energy X-ray absorptiometry based vertebral fracture assessment, the prevalence of vertebral fracture was approximately 5% in the 60s, 10% in the 70s, and 20% in the 80s . Fewer than 10% of those diagnosed with vertebral fracture had a known prior history. These findings provide the rationale for recommending targeted screening of appropriate individuals as part of a comprehensive evaluation of future fracture risk .
In addition to prior recent fracture, multiple fractures and prevalent vertebral fracture, an individual with very low BMD ( T -score <−3), even in the absence of fractures, are a candidate for PTH receptor agonist therapy. Long-term risk of fracture is extremely high in these patients (though imminent risk might not be high, particularly if young and otherwise healthy). This recommendation is based on multiple studies that have shown that the sequence of PTH treatment first, followed by potent antiresorptive therapy, is most effective at improving BMD, particularly for the hip region . Nevertheless, anabolic therapy should also be considered for individuals who incur incident fractures or active bone loss while on other therapies or who have persistent severe osteoporosis despite prior therapy. In patients on current bisphosphonates, BMD declines in the hip for 12–24 months upon switching to TPTD . The hip BMD decline is quite a bit more prominent in patients on denosumab who switch to TPTD. In denosumab-treated patients, hip BMD remains below the postdenosumab baseline level throughout an entire 2 years of TPTD administration. In bisphosphonate and denosumab-treated patients, it might be preferable to consider using TPTD in combination with ongoing bisphosphonate or denosumab. Although a direct comparison between adding TPTD to ongoing denosumab compared with switching to TPTD and stopping denosumab has not been done, the addition of TPTD is likely to be favorable for BMD (compared with switching to TPTD), based on analogy from de novo coadministration of TPTD and denosumab in the first 2 years of the DATA trial . Additional supportive data are from the study of TPTD with zoledronic acid where hip BMD changes were superior with combination therapy compared to TPTD alone . Furthermore, in patients on long-term alendronate, adding TPTD to ongoing alendronate produced a superior effect on hip BMD compared with stopping alendronate and switching to TPTD .
Like TPTD, ABL has been associated with an increased risk of osteosarcoma in rodents . Therefore neither ABL nor TPTD is recommended for individuals who have had osteosarcoma or who are at elevated risk (family history of osteosarcoma, skeletal radiation, Paget’s disease). In addition, neither agent is recommended for those who have metastatic bone cancer, myeloma, hyperparathyroidism, or hypercalcemia. The course of TPTD or ABL is 18–24 months . Treatment should be followed by a potent antiresorptive therapy, usually either denosumab or a potent bisphosphonate .
76.7
Conclusion
Because of the underlying effects, these agents produce on microarchitecture and mass of bone, as well as cellular renewal, TPTD and ABL may be able to ensure greater strength and more long-term protection against fracture than antiresorptive agents alone. Antiresorptive agents after TPTD and ABL are able to sustain benefits on both BMD and fracture risk reduction. For patients with severe osteoporosis, early anabolic therapy may help achieve therapeutic goals (fracture free intervals and BMD T -scores significantly above osteoporosis range) faster than antiresorptive agents alone and may permit a shorter duration of overall therapy, hopefully minimizing the risk of long-term adverse consequences.
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








