1. Temperature greater than 38.8°C 2. Systolic blood pressure ≤90 mm Hg for adults, less than the 5th percentile for children, or greater than 15 mm Hg orthostatic drop in diastolic blood pressure or orthostatic dizziness/syncope 3. Diffuse macular rash with subsequent desquamation 4. Three of the following organ systems involved: Liver: bilirubin, AST, ALT more than twice the upper normal limit Blood: platelets <100 000/mm3 Renal: BUN or creatinine more than twice the upper normal limit or pyuria without urinary tract infection Mucous membranes: hyperemia of the vagina, oropharynx, or conjunctivae Gastrointestinal: diarrhea or vomiting Muscular: myalgias or CPK more than twice the normal upper limit Central nervous system: disorientation or lowered level of consciousness in the absence of hypotension, fever, or focal neurologic deficits 5. Negative serologies for measles, leptospirosis, and Rocky Mountain spotted fever; blood or CSF cultures negative for organisms other than Staphylococcus aureus |
Abbreviations: AST = aspartate transaminase; ALT = alanine aminotransferase; BUN = blood urea nitrogen; CPK = creatine phosphokinase; CSF = cerebrospinal fluid.
| 1. Isolation of group A streptococci: From a sterile site for a definite case From a nonsterile site for a probable case | |
| 2. Clinical criteria: Hypotension and two of the following: Renal dysfunction Liver involvement Erythematous macular rash | Coagulopathy ARDS Soft-tissue necrosis |
Abbreviations: ARDS = adult respiratory distress syndrome.
Menstrual TSS (Figure 18.1), which occurs within a day or two of or during menstruation, primarily has been associated with use of certain tampons, notably those of high absorbency, and is associated with production of TSS toxin-1 (TSST-1) by the causative bacterium. When initially described, menstrual TSS occurred primarily in women of ages 15 to 25. Presently, menstrual TSS occurs more often in younger females, particularly ages 12 to 15; the reason for this shift in menstrual TSS to the younger age group is unclear. There are two major mechanisms that explain the association of tampons with menstrual TSS: (1) tampons introduce oxygen, which is absolutely required for production of TSST-1, into the otherwise anaerobic vagina; and (2) Pluronic L-92, a surfactant present in the TSS-associated Rely tampon from the 1980s and no longer used in tampons, amplifies TSST-1 production.
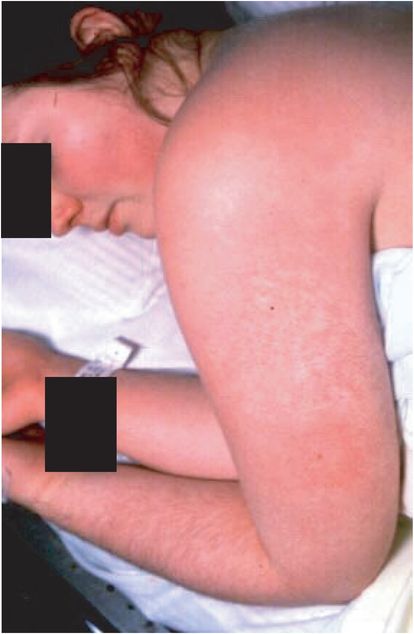
Figure 18.1 Menstrual toxic shock syndrome (vaginal colonization): diffuse blanching erythema. (Courtesy of David Schlossberg, MD.)
Nonmenstrual TSS occurs in both males and females, adults and children, and is associated with S. aureus strains that produce TSST-1 or staphylococcal enterotoxins, notably enterotoxin serotypes B and C. These three SAgs are produced in amounts nearly 105 times higher than most other SAgs. Nonmenstrual TSS occurs in association with nearly any kind of staphylococcal infection, but major forms have been identified: postsurgical, upper respiratory virus (influenza)-associated, RED syndrome (see below), enterocolitis-associated, and occasionally with use of contraceptive diaphragms. Postsurgical TSS is often associated with S. aureus infections that do not result in pyogenic responses, and thus the infection source may be difficult to find. Influenza TSS may occur as a consequence of influenza or parainfluenza damage to the respiratory tract epithelium and superinfection with SAg-producing S. aureus (Figures 18.2 and 18.3). This illness is highly fatal in children. RED syndrome is a recalcitrant erythematous desquamating disorder in patients with acquired immunodeficiency syndrome (AIDS) that may last 70 or more days until the patient succumbs. Recently, staphylococcal TSS has been associated with enterocolitis. Until enterocolitis became highly associated with Clostridium difficile infection, staphylococcal enterotoxins were associated with the illness. Although sometimes forgotten, staphylococcal SAgs should be considered as causes of enterocolitis with TSS symptoms when C. difficile cannot be isolated from patients. Finally, nonmenstrual TSS associated with use of diaphragms may be similar to menstrual TSS.
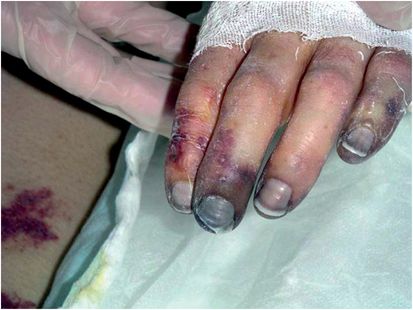
Figure 18.2 Gangrenous fingers and skin peeling with nonmenstrual staphylococcal toxic shock syndrome (pulmonary infection). (Courtesy of Gary R. Kravitz, MD, St. Paul Infectious Disease Associates, St. Paul, MN.)
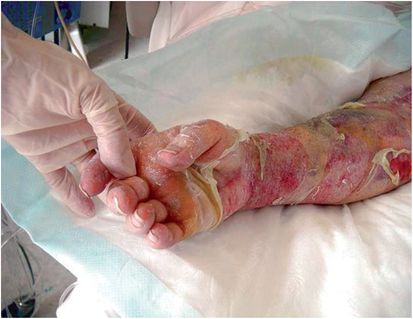
Figure 18.3 Same patient as in Figure 18.2, demonstrating extensive peeling and tissue damage. (Courtesy of Gary R. Kravitz, MD, St. Paul Infectious Disease Associates, St. Paul, MN.)
Streptococcal TSS is primarily associated with group A streptococcal infections, particularly M serotypes 1, 3, and 18. The illness may or may not be associated with necrotizing fasciitis or myositis (Figure 18.4). The case:fatality rate is higher in cases with necrotizing myositis than when this condition is absent. Occasionally, streptococcal TSS is caused by other groups of streptococci, primarily groups B, C, and G.
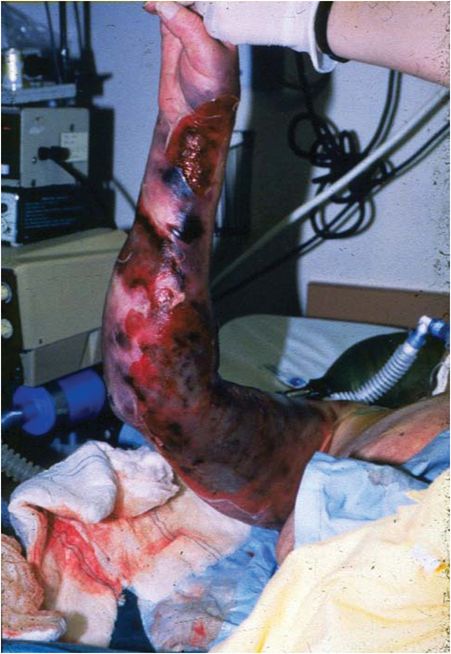
Figure 18.4 Necrotizing fasciitis: note the extensive necrosis in this patient’s arm. At the time, this patient was hypotensive with multiple organ failure and severe pain at the site of infection.
Group A streptococcal strains that cause TSS produce streptococcal pyrogenic exotoxin SAgs. The major associations have been with serotypes A and C, but other family members contribute significantly. Nongroup A streptococcal strains associated with TSS also produce streptococcal pyrogenic exotoxin SAgs that are related or identical to the group A streptococcal SAgs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree