| Factors | Percentage |
|---|---|
| Infectious etiology | 68.8 |
| Endocarditis | 15.3 |
| Septic syndrome | 11.9 |
| Miscellaneous | 11.9 |
| Urinary infection | 7.1 |
| Otitis | 3.3 |
| Appendicitis | 2.8 |
| Pneumonia | 2.8 |
| Brucellosis | 2.3 |
| Lung abscess | 2.3 |
| Malaria | 1.9 |
| Diverticulitis | 1.9 |
| Amebiasis | 0.95 |
| Noninfectious etiology | 31.2 |
| Contiguous diseases | 23.0 |
| Trauma | 16.7 |
| Hemoglobinopathies | 11.9 |
Metastatic hematogenous infections
Infective endocarditis is the most common condition predisposing a patient to splenic abscess (Table 56.1, Figure 56.1). Although the exact incidence is difficult to determine, several studies demonstrated the occurrence of splenic embolization in 31% to 44% of the patients with endocarditis. Histologic examination disclosed splenitis in at least 20% of patients. Splenic infarction occurred in 30% to 67% of patients with endocarditis during the pre-antibiotic era, and in 33% to 44% of these patients during the antibiotic era. In 1977, Pelletier and Petersdorf reported the incidence of splenic abscess in patients with subacute bacterial endocarditis to be approximately 2.4%. Mycotic aneurysms are seen angiographically within abscesses, but whether these predispose a patient to, or result from, splenic abscess remains uncertain.
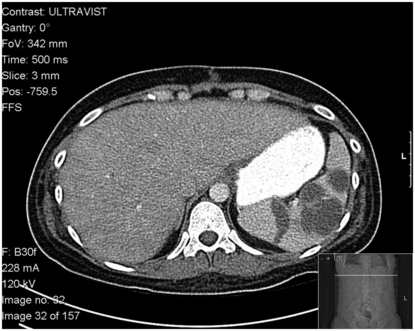
Figure 56.1 Multilocular splenic abscess in an intravenous drug abuse patient with bacterial endocarditis.
In addition to endocarditis, a multitude of other infections have been reported as primary causes of splenic abscess (see Table 56.1). Miscellaneous infections include dental abscess, bacteremia after dental extraction, tonsillectomy, peritonsillar abscess, acute parotitis, bronchiectasis, perinephric abscess, decubitus ulcer, complicated infectious mononucleosis, tuberculosis, yellow fever, typhoid fever, diphtheria, cat scratch disease, and anthrax.
Conditions resulting in splenic ischemia and infarct have been implicated in the development of splenic abscess, probably from hematogenous spread of pathogens. Splenic abscess has been reported after splenic artery ligation in the course of liver transplantation surgery, division of the short gastric vessels during laparoscopic Nissen fundoplication, and accidental injection of the splenic artery during endoscopic procedures for gastric bleeding.
Contiguous infection
On occasion, splenic abscess can result from the direct extension of disease having its primary focus in adjacent organs. Contiguous extension from diverticulitis, pancreatic pseudocyst or carcinoma, gastric ulcer, carcinoma of the stomach, perihepatic abscess, perinephric and subphrenic abscess, and carcinoma of the descending colon have been reported. Splenic abscess has rarely been reported as an extraintestinal manifestation of inflammatory bowel disease.
Traumatic abscess
Traumatic abscess results from secondary infection and suppuration of contused parenchyma or of a hematoma arising from injury to splenic tissue. In a report by Phillips, the initial traumatic injury was not easily recognized or reported, and most patients developed signs and symptoms of splenic infection after a latent period of 2 weeks to 4 months after sustaining injuries to the left upper quadrant. Splenic abscess has been reported after operative repair of splenic injury (splenorrhaphy), and nonoperative management of blunt splenic injuries diagnosed by computed tomography (CT) scan (Figure 56.2). On occasion, radiologic procedures such as splenic artery embolization for hemorrhage control following traumatic injury, splenoportography for portocaval shunt evaluation, and percutaneous transluminal coronary angioplasty have been implicated as causes of splenic abscess, sometimes up to 4 months afterward.
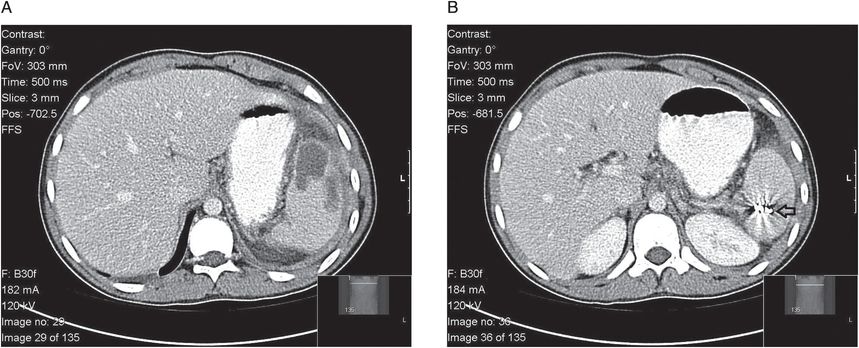
Figure 56.2 (A, B) Splenic abscess following nonoperative management of blunt splenic injury including splenic artery embolization. Note embolization coil (B, see arrow).
Hematologic disorders
Hemoglobinopathies accounted for approximately 12% of splenic abscesses reported by Alsono-Cohen. Patients with sickle cell disease have an increased risk of acquiring invasive bacterial infections as a result of hyposplenism, including functional defects in opsonization, phagocytic function, and cell-mediated immunity. If a patient with sickle cell disease and prior splenic infarcts develops a transient bacteremia from a central line infection or cholecystitis, bacteria may seed the infarcted regions with resultant abscess formation.
The spleen may also be a site of infection in patients with collagen vascular diseases. Splenic abscesses have been reported in patients with rheumatoid arthritis, systemic lupus erythematosus, myelodysplastic syndrome, and polyarteritis nodosa. Pathologic features of the spleen in these illnesses include capsulitis and small infarcts.
Immunodeficiency states
Splenic abscess has been reported complicating acquired immunodeficiency syndrome (AIDS), chemotherapy, cancer (leukemia, lymphoma), bone marrow and solid organ transplantation, long-term steroid use, monoclonal antibody immunosuppressive medications, and conditions such as diabetes mellitus and alcoholism.
Diagnosis
History and physical examination
The signs and symptoms of a splenic abscess are often insidious, nonspecific, and related to the underlying disease. Table 56.2 characterizes the clinical findings in 227 patients. Fever is the most common symptom along with pain in the left hypochondrium or vague abdominal pain. Pain is probably caused by splenitis with capsular involvement. Abscesses located in the upper pole of the spleen tend to irritate the diaphragm, causing radiation of pain toward the left shoulder (Kehr’s sign) and an elevated, immobile left hemidiaphragm. Splenic rupture also commonly manifests as left shoulder pain. An abscess located in the lower pole of the spleen more often irritates the peritoneal surface, resulting in peritonitis. A deep-seated abscess that does not involve the splenic capsule may be accompanied only by nonspecific symptoms of infection without pain or other localizing signs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree