Fig. 3.1
Structural layers of the esophagus. (a) H&E stain. The innermost layer (or tunica) is the mucosa. The mucosa is composed of an epithelial lining (squamous epithelium), the underlying lamina propria (loose connective tissue that supports the epithelium), and the muscularis mucosae (a thin layer of smooth muscle). Deep to the mucosa is the submucosa, which contains more cellular connective tissue, prominent blood vessels (with muscularized arteries), nerve fibers (Meissner plexus), and submucosal mucus (exocrine) glands. A submucosal gland is present in this figure. The thick outer muscular layer is called the muscularis mucosa (or muscularis externa) and in the distal esophagus (as shown in this figure) is composed of an inner circular layer and an outer longitudinal layer of smooth muscle; the layers are separated by the myenteric plexus. Skeletal muscle is admixed with smooth muscle of the muscularis propria in the mid esophagus, and in the proximal esophagus, the outer muscular layers are primarily composed of skeletal muscle. The outermost layer of the esophagus is the adventitia, which is composed of loose connective tissue and helps to anchor the esophagus in place (H&E stain, 20×). (b) Schematic diagram illustrating the structural layers of the esophagus
Neoplastic transformation can involve any of the cell types found in the esophagus. However, the vast majority of malignant tumors that arise from the esophagus are epithelial in origin. This review will focus on the malignant epithelial lesions of the esophagus, namely, adenocarcinoma and squamous cell carcinoma, and their precursor lesions.
Esophageal cancer affects more than 450,000 people worldwide, and squamous cell carcinoma is the predominant histologic type [1]. In the United States, Australia, the United Kingdom, and some Western European countries, the incidence of adenocarcinoma is increasing rapidly and now exceeds that of squamous cell carcinoma [1]. The estimated number of new cases of esophageal cancer in the United States in 2014 is 18,170, and the vast majority (14,660) will affect men [2]. Esophageal cancer is estimated to be the 7th leading cause of cancer deaths in men in 2014 in the United States, accounting for 12,450 or 4 % of all cancer deaths (following lung, prostate, colorectal, pancreatic, liver cancer, and leukemia) [2].
Pathology of Adenocarcinoma and Its Precursor Lesions
Adenocarcinoma typically arises in the distal third of the esophagus and is associated clinically with dysphagia and weight loss. Several risk factors have been described for esophageal adenocarcinoma, including increased age, male gender, white ethnicity, high body mass index, low fruit and vegetable intake, absence of Helicobacter pylori infection, presence of a hiatal hernia, and history of reflux disease [3–10]. Clinical observations and studies in animal models suggest a linear sequence in the development of esophageal adenocarcinoma. Initially, the replacement of the normal squamous epithelium by glandular mucosa (gastric type or intestinal type) occurs secondary to repeated injury from bile and acid reflux. This is followed by dysplastic change within this metaplastic tissue, leading to the development of invasive carcinoma after molecular evolution of the dysplastic epithelium.
Precursor Lesions of Esophageal Adenocarcinoma
In a medical position statement on the management of Barrett’s esophagus by the American Gastroenterological Association, Barrett’s esophagus is defined as the condition in which any extent of metaplastic columnar epithelium that predisposes to cancer development replaces the stratified squamous epithelium that normally lines the distal esophagus; presently, intestinal metaplasia is required for the diagnosis of Barrett’s esophagus as it is the only type of esophageal columnar epithelium that predisposes to malignancy [11]. However, not all professional organizations (including the British Society of Gastroenterology) require the histologic documentation of intestinal metaplasia [12].
Macroscopically, Barrett’s esophagus appears as a well-demarcated area of erythematous or “velvety” mucosa within the squamous-lined tubular esophagus. Histologically, three different types of columnar or glandular metaplasia can be seen on biopsies from patients with “Barrett’s” esophagus: (1) gastric cardia-type mucosa (composed of mucin-secreting glands); (2) gastric oxyntic (or fundus)-type mucosa composed of parietal cells, chief cells, and mucus-secreting cells; and (3) specialized columnar epithelium or intestinal metaplasia containing round, bluish, barrel-shaped cells called goblet cells, often admixed with gastric cardia-type glands (Fig. 3.2). From the pathologist’s perspective, the gastric-type mucosa ± goblet cells seen in the tubular esophagus is indistinguishable from the true gastric mucosa seen in the cardia. Hence, if the pathologist does not see submucosal glands on a biopsy and does not know what the endoscopist observed at the time of biopsy, a diagnosis of “gastric-type mucosa with intestinal metaplasia” may be rendered instead of a diagnosis of “Barrett’s esophagus.” Goblet cells produce acid-rich mucins and thus stain intensely blue with an Alcian blue stain at a pH of 2.5. In addition, the intestinal mucosa stains positive for immunohistochemical stains such as CDX2, villin, and MUC-2 (markers of intestinal differentiation) [13–16]. Although ancillary stains can detect goblet cells and intestinalized epithelium, these stains are not required for a diagnosis of intestinal metaplasia, as the goblet cells can often be readily identified on the routine hematoxylin and eosin (H&E) stain (Fig. 3.2).
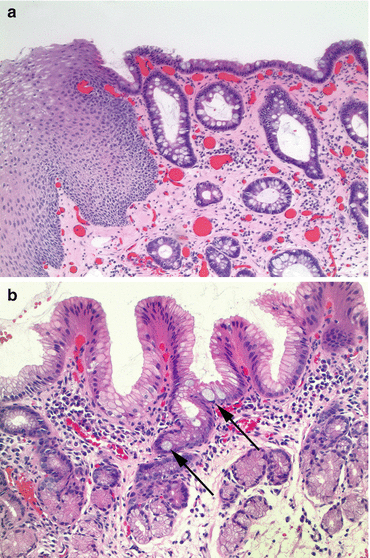

Fig. 3.2
Intestinal metaplasia. (a) In this example, the squamous mucosa has been replaced by glandular mucosa of intestinal type. Some intact squamous epithelium is present on the left. All of the glands present on the right contain goblet cells (specialized columnar epithelium or intestinal metaplasia). If this biopsy was obtained from the tubular esophagus, these findings are consistent with Barrett’s esophagus (H&E stain, 100×). (b) Gastric-type mucosa with rare goblet cells (focal intestinal metaplasia). Many biopsies from islands of velvety mucosa within the tubular esophagus contain gastric cardiac-type glands. If even one goblet cell is identified, a diagnosis of (focal) intestinal metaplasia can be rendered. As shown in this example, there are about five goblet cells (two are highlighted by arrows) present within the gastric-type mucosa. If this biopsy was obtained from the tubular esophagus, these findings are consistent with Barrett’s esophagus (H&E stain, 100×)
It is worth noting that in addition to designating the presence and/or absence of intestinal metaplasia, pathologic reports may detail several other histologic mimics of metaplasia. “Pseudogoblet cells” refer to barrel-shaped gastric foveolar cells that may look like goblet cells on low magnification, but have an eosinophilic tinge and do not stain for Alcian blue due to a production of neutral mucins. The term “columnar blues” refers to the identification of mucus cells that contain bluish mucin on the H&E stain and stain positive for Alcian blue but lack goblet cell morphology. The term “multilayered epithelium” may be noted in reports. This finding refers to the identification of an epithelium that contains flattened squamous-appearing cells in the basal layers with an overlying columnar mucus cell layer. Studies have shown that this epithelium can show immunohistochemical features similar to intestinal-type epithelium, indicating that it may represent an early or intermediate phase in the development of intestinal metaplasia and Barrett’s esophagus [17]. Finally, pathologists may report the presence of “subsquamous intestinal metaplasia” (also called “buried Barrett’s” or “squamous overgrowth”), particularly in endoscopic mucosal resection specimens. Although subsquamous intestinal metaplasia is present in the majority of endoscopic mucosal resection specimens and is no longer felt to be a post-ablative phenomenon, the long-term clinical significance is uncertain, and histologic evaluation of squamous-lined mucosa adjacent to areas of BE may be indicated [18, 19].
Once a diagnosis of Barrett’s esophagus has been rendered, increased surveillance is indicated in order to monitor for the presence of dysplasia and/or adenocarcinoma. Studies have shown that 0.12–0.38 % of patients with nondysplastic Barrett’s esophagus will develop adenocarcinoma each year [20]; thus, screening efforts have focused largely on identifying metaplasia in at-risk individuals. Definitive dysplastic change falls into one of two general categories: low-grade or high-grade dysplasia. Features of low-grade dysplasia (Fig. 3.3a) include increased epithelial proliferation characterized by nuclear crowding and minimal distortion of glandular architecture. Mild cytologic atypia including nuclear enlargement, hyperchromasia, stratification, and irregular nuclear contours is also observed. Nuclear-to-cytoplasmic ratios and mitotic counts remain low, basal nuclear polarity is preserved, and in general, the glandular architecture is retained, though there may be increased glandular crowding. Low-grade dysplasia in Barrett’s esophagus often resembles the epithelium of a colonic-type adenoma. For a diagnosis of low-grade dysplasia, the cytologic atypia should extend to the surface. When surface involvement is not present or cannot be evaluated (e.g., due to squamous overgrowth), the term “indefinite for dysplasia” may be used.
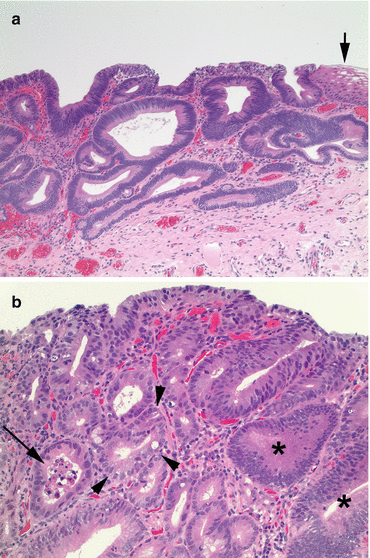

Fig. 3.3
Dysplasia in Barrett’s esophagus. (a) Low-grade dysplasia. All of the glandular epithelium in this example is involved by low-grade dysplasia; there is a small portion of intact squamous mucosa present (arrow). When dysplasia develops in Barrett’s esophagus, the normal goblet cells are often lost because they are replaced by neoplastic epithelial cells that are replicating without normal inhibition and maturation. Characteristic features of low-grade dysplasia include elongation and crowding of the cell nuclei (penicillate nuclei) and increased nuclear-cytoplasmic ratios. These features are typically present from the deep glands all the way to the luminal surface, indicating the lack of maturation. As opposed to high-grade dysplasia, the cell nuclei are still basally oriented (nuclear polarity is maintained), the nuclei are not round, and nucleoli are inconspicuous (H&E stain, 100×). (b) High-grade dysplasia. All of the glands in this example show evidence of dysplasia; two glands (*) show features of low-grade dysplasia, but most of the remaining glands, especially towards the left and towards the luminal surface, show features of high-grade dysplasia. In high-grade dysplasia, there are both cytologic changes and architectural changes. Cytologically, the nuclear-cytoplasmic ratio is increased, and the nuclei “round up,” often contain prominent nucleoli, and lose polarity (several nuclei appear detached from the basal aspect of the cells and are present towards the luminal surface of the glands). Architecturally, the cells proliferate within the lumens of the dysplastic glands, creating cribriform architecture or “gland-within-gland” morphology (arrowheads). Necrotic and apoptotic cells are also often present within glandular lumens (arrow) (H&E stain, 100×)
In contrast to low-grade dysplasia, high-grade dysplasia (Fig. 3.3b) is characterized by increased cellular atypia and complex architecture. Cells have high nuclear-to-cytoplasmic ratios, and there is a loss of basal nuclear polarity with the large often rounded nuclei containing prominent nucleoli. There is also distortion of the normal glandular architecture, often with intraglandular bridging or cribriforming. Cellular or apoptotic debris may be found within glandular lumens. Mitotic activity is evident and often extends beyond the deep proliferative zone and may reach the luminal surface. Deep glands with dilated lumens lined by attenuated epithelium may also indicate the presence of high-grade dysplasia or a lesion of greater clinical significance (Fig. 3.4). In the 7th ed of the AJCC staging manual, the term “Tis” (or “carcinoma in situ”) was removed from all epithelial neoplasia of the gastrointestinal tract and was replaced by “high-grade dysplasia” [21]. Since there are some cases of intraepithelial neoplasia that are more atypical than expected for high-grade dysplasia, or cases where definitive invasion cannot be documented, some pathologists may use the term “carcinoma in situ” or “at least high-grade dysplasia.” If these terms are noted in the pathology report without an explanatory comment, a discussion with the pathologist may help clarify its meaning in individual cases.
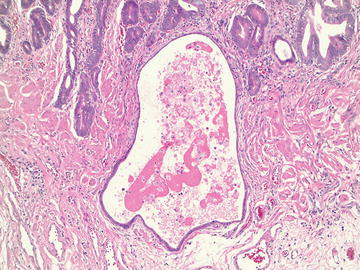

Fig. 3.4
Attenuated gland in the background of high-grade dysplasia. Even though the epithelial lining of the dilated gland is flat and appears bland, the presence of luminal necrotic debris and its presence within muscle bundles make this gland suspicious for attenuated high-grade dysplasia, and it may actually represent early intramucosal adenocarcinoma. If present in the submucosa or within the muscularis propria, attenuated glands with these same histologic features would be diagnostic of invasive adenocarcinoma (H&E stain, 100×)
Esophageal Adenocarcinoma
Once adenocarcinoma develops, it can be managed by different modalities. Small early lesions may be amenable to endoscopic mucosal resection. Surgery ± neoadjuvant therapy is the treatment of choice for more deeply invasive or more advanced lesions. Grossly, esophageal adenocarcinoma often appears as an infiltrative mass in the distal third of the esophagus, although fungating, polypoid, and flat growths can be seen (Fig. 3.5a). Histologically, adenocarcinoma is characterized by invasion beyond the basement membrane. Invasion beyond the basement membrane into the lamina propria of the esophagus is a significant development, as it allows access to lymphatic channels that are not found in other areas of the alimentary track, such as the colon. Hence, as per the AJCC staging recommendations, early esophageal adenocarcinoma is staged as either T1a (intramucosal invasion) or T1b (submucosal invasion) [21].
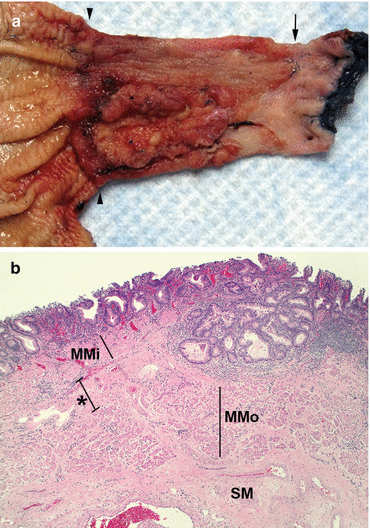
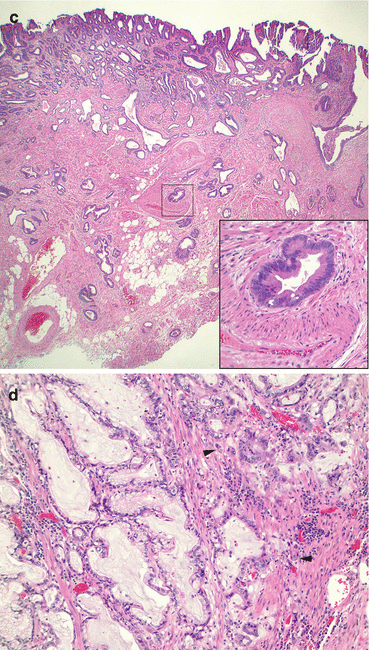


Fig. 3.5
Adenocarcinoma. (a) Esophagogastrectomy specimen with adenocarcinoma. The stomach, with normal rugal folds, is towards the left, while the inked squamous-lined esophageal margin is towards the right. The arrowheads highlight the esophagogastric junction. The majority of the esophagus is lined by velvety-appearing mucosa, which represents Barrett’s esophagus. The arrow indicates the squamocolumnar junction, which is several centimeters from the esophagogastric junction. Within the distal esophagus and within the Barrett’s segment, there is an exophytic mass that is an adenocarcinoma. This patient did not receive neoadjuvant therapy prior to the resection. (b) Intramucosal adenocarcinoma. A small focus of adenocarcinoma is invading with a pushing boarder into the space (*) between the two layers of the duplicated muscularis mucosae, making this a pT1a adenocarcinoma. Even though this tumor has invaded through the internal layer of the muscularis mucosae (MMi), it has not invaded through the outer (duplicated) layer (MMo), so this is not invasive into the submucosa (SM) (H&E stain, 40×). (c) Well-differentiated adenocarcinoma. This endoscopic mucosal resection specimen shows dysplasia towards the luminal surface and glandular structures “dripping” through the muscularis mucosae into the submucosa. On low magnification (H&E stain, 20×), there is no obvious desmoplastic response. On high magnification (inset, H&E stain, 200×), if taken out of context, the malignant gland could represent a dysplastic gland, but its presence in the submucosa (as noted by the large muscular artery adjacent to the gland) indicated that this is an invasive adenocarcinoma present in the submucosa. (d) Invasive adenocarcinoma. This example is primarily well differentiated as most of the malignant cells are forming glands. There is not much desmoplasia in this example, but the malignant glands are present within the muscularis propria, and the smooth muscle fibers can be seen in the background stroma. Compared to high-grade dysplasia, there is a bit more cytologic and nuclear atypia in this example of adenocarcinoma, including more nuclear pleomorphism, as well as irregularly shaped glands (upper right corner) and some small nests and single cell infiltration (arrowheads) (H&E stain, 100×)
In pathology, most early adenocarcinomas are evaluated on either small biopsies or endoscopic mucosal resection (EMR) specimens. Determining the depth of invasion can be challenging on such specimens because of a phenomenon known as duplication of the muscularis mucosae. For unknown mechanistic reasons, in areas of intestinal metaplasia (Barrett’s esophagus), a new internal layer of the muscularis mucosa is created [22, 23] (Fig. 3.5b). In order to become a T1b lesion, an adenocarcinoma arising in these areas needs to invade through three layers: the “new” superficial muscularis mucosae, the loose connective tissue between the two layers, and then the deep or true muscularis mucosae. Hence, true submucosal invasion is difficult to determine on small biopsies or superficial EMR specimens.
Since EMR specimens are small but contain pertinent information, recommendations for the handling of such specimens have been reported [24]. Ideally, fresh specimens should be pinned to cork or foam prior to formalin fixation. Photographic documentation is recommended. Then the entire specimen should be submitted for histologic evaluation. In addition to reporting the main pathology (dysplasia or intramucosal carcinoma), pathologists must comment on the status of the lateral (mucosal) and deep margins. If an adenocarcinoma is present, the depth of invasion must be assessed. As noted above, the pitfall of overcalling a T1a lesion as a T1b tumor due to duplicated muscularis mucosae must be avoided. The AJCC further subdivides both intramucosal (m1, m2, and m3) and submucosal (sm1, sm2, sm3) invasion [25]. Although the AJCC does not take into account the different layers of the duplicated MM, other studies have [26]. Hence, pathologists should attempt to describe the specific depth of invasion, for example, “intramucosal adenocarcinoma, invasive into the superficial muscularis mucosal layer.” However, only the depth of mucosal invasion may be discernable on biopsies or small EMR specimens. Determining the different levels of submucosal invasion (i.e., sm1, sm2, sm3) is not practical in biopsies or small EMR specimens where the outer limit of the measurement (border of the muscularis propria) is absent. However, given the high risk of nodal involvement with submucosal invasion, the precise depth of invasion on these types of specimens should not alter patient management.
Grading of adenocarcinoma has prognostic significance and falls into one of three categories: well (G1), moderately (G2), or poorly (G3) differentiated; undifferentiated tumors are uncommon, often cannot be subtyped as squamous or glandular, and are considered to be grade 4 (G4) tumors. The importance of accurate tumor grading is critical for clinical management, as AJCC guidelines for clinical staging of adenocarcinoma integrate tumor grade into the algorithm for determining clinical stage [21]. For example, a well- or moderately differentiated T2 N0 M0 tumor is stage IB, while a poorly differentiated T2 N0 M0 tumor is stage IIA. For adenocarcinoma, grading involves determining the percent of tumor that is composed of glands: well-differentiated tumors contain >95 % glands, moderately differentiated tumors contain 50–95 % glands, and poorly differentiated tumors display <50 % glandular architecture [21]. When tumors contain areas of multiple grades, the highest grade is documented. Histologically, well-differentiated tumors are composed of glands with irregular shapes or profiles (often with focal cribriform formation) lined by cuboidal to columnar cells with mild to moderate atypia. Not all adenocarcinomas illicit a desmoplastic response. In the absence of desmoplasia, a well-differentiated adenocarcinoma could be easily misdiagnosed as dysplasia if assessed out of context. Hence, even the most bland-appearing glands are adenocarcinoma if they are present in the submucosa or muscularis propria (Fig. 3.5c). Esophageal adenocarcinomas are often well to moderately differentiated (Fig. 3.5d). As tumors become more poorly differentiated, a more sheetlike appearance is identified. Signet ring cells, single infiltrating cells, and/or wildly atypical tumor cells may be present. Often, poorly differentiated tumors elicit a strong desmoplastic response from neighboring stromal cells. When dealing with a poorly differentiated adenocarcinoma on biopsy specimens, metastatic disease should be considered and excluded, either clinically or immunohistochemically (e.g., immunostains for breast markers could help diagnose metastatic breast carcinoma). Unfortunately, there are no immunomarkers that are diagnostic of esophageal adenocarcinoma, although most tumors express CK7 and may express CDX2 and CK20.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree