Chapter Outline
HYPERLEUKOCYTOSIS AND LEUKOSTASIS
Expert and timely management of emergent situations is crucial in the practice of pediatric oncology. Although many emergencies are common across the spectrum of pediatric patients, such as seizure, respiratory arrest, and electrolyte abnormalities, others stem from the underlying cancer or cancer-directed therapy and are unique to this population of patients. These types of emergencies require special consideration and specific management to minimize adverse outcomes. Emergencies related to space-occupying lesions, metabolic derangements caused by breakdown of tumor cells, and disruption of the hematopoietic system by invasion of cancer cells all in some way relate to disruption of the normal function of a cell or organ as a result of the abnormal presence of cancer cells. This disruption can be rapidly progressive and life-threatening, as when patients with T-cell lymphoblastic leukemia present with a mediastinal mass and respiratory compromise. The treatments directed toward cancer also carry significant toxicities and can result in uniquely emergent complications. Many chemotherapy agents result in a state of significant immune compromise and put patients at risk for unusual or opportunistic infections, as well as fulminant presentations of infection with more common organisms. All chemotherapy drugs carry extensive profiles of possible adverse effects, and errors of administration or dosage can magnify toxicity; any available therapeutic interventions must be administered immediately. Appropriate management of an emergent pediatric oncologic situation requires a clear understanding of the specific underlying disease process and of the effects and consequences of cancer-directed therapies.
Symptoms in children who present with new diagnoses of cancer or are receiving cancer treatment are myriad. Therefore recognition of an emergency is not always straightforward, particularly in the young pediatric population, for whom obtaining an accurate history can be challenging. It is therefore imperative that the clinician maintain a high level of vigilance for the presentation of potentially life-threatening situations. A child with cough and mild respiratory distress is most likely to have a viral infection, but these symptoms also might be consistent with a child presenting with a marked mediastinal mass, developing leukostasis syndrome, or the onset of Pneumocystis pneumonia. A complaint of lower back pain in a child may relate to new physical activity or could represent developing spinal cord compression from a tumor. Pediatric oncologists need to be familiar with all pediatric oncologic emergent situations to evaluate and manage these high-risk patients appropriately. Rapid and appropriate triage of patient complaints is critical, because overlooked diagnoses, as well as inaction or inappropriate action, can have severe consequences.
Grouping of some emergent pediatric oncologic conditions into disease- and treatment-related events is presented in Box 68-1 . Clear overlap exists among the conditions listed; for example, tumor lysis syndrome (TLS) can occur on presentation with a new diagnosis of leukemia or high-grade lymphoma, but it can be markedly worsened by initiation of chemotherapy. Similarly, superior vena cava syndrome (SVCS) can result from a new or recurrent presence of a mass lesion or as a complication of an indwelling catheter placed to deliver antineoplastic therapy. In this chapter our discussion will focus on several emergent situations unique to pediatric oncology patients, with a review of available data and overviews of current management strategies. The topics addressed in the chapter are anterior mediastinal mass, increased intracranial pressure, spinal cord compression, hyperleukocytosis and leukostasis, TLS, and selected chemotherapy toxicities. Several topics unique to the pediatric oncology patient are covered elsewhere in this text and will not be readdressed in this chapter (e.g., management of fever and neutropenia, typhlitis, veno-occlusive disease, all- trans -retinoic acid syndrome, transfusion medicine, and pain management). Situations that can occur in the pediatric oncology patient but that are not unique to this population, such as seizure, cerebral vascular injury, disseminated intravascular coagulation, and pancreatitis, will not be discussed here.
Disease-Related
Anterior mediastinal mass
Increased intracranial pressure
Spinal cord compression
Leukocytosis and leukostasis
Tumor lysis syndrome
Disseminated intravascular coagulation
Treatment-Related
Fever and neutropenia, sepsis
Typhlitis
Pancreatitis
Vesicant infiltration
Methotrexate toxicity
All- trans -retinoic acid syndrome
Veno-occlusive disease
Intrathecal chemotherapy overdose
Central nervous system events such as seizure or CVA
Severe allergic reactions to chemotherapy agents or blood products
Although optimal management of many of these emergent situations has been difficult to study, given the relatively small number of patients presenting to each institution, a literature is evolving for many of these topics. Many institutions have established clinical practice guidelines to help manage the most common pediatric oncologic emergencies. Clinical practice guidelines provide uniform standards for management but must allow for adaptations for each specific clinical scenario. Uniform use of clinical guidelines with collection and analysis of outcomes has the potential to guide improvements in clinical practice.
Anterior Mediastinal Masses
Definition
A commonly encountered oncologic emergency is the entity of anterior mediastinal mass. A clinician should always consider this entity in the differential diagnosis of a pediatric patient with new respiratory distress and must consider evaluation, even in asymptomatic patients presenting with any of the variety of cancers known to be associated with the occurrence of anterior mediastinal masses.
Space-occupying lesions occurring in the anterior mediastinum can impinge on vital structures, causing significant cardiac or airway compromise. The anterior mediastinum is defined as the anatomic compartment bounded by the sternum, thoracic inlet, and anterior border of the heart. The presence of a mass in this region can cause SVCS, tracheobronchial compression, or compression of the heart or pulmonary vessels. The term superior mediastinal syndrome (SMS) has been used to denote the combination of great vessel and tracheobronchial tree compression. However, because these phenomena may be overlapping, particularly in the pediatric population, the terms SVCS and SMS are often used interchangeably.
Neoplasms that cause anterior mediastinal masses in the pediatric population and are associated with SVCS and/or tracheal compression include non-Hodgkin lymphoma (NHL), acute lymphoblastic leukemia (ALL), and Hodgkin lymphoma (HL), as well as neuroblastoma, paraganglioma, germ cell tumors, sarcoma (including myeloid sarcoma), thymoma, desmoplastic round cell tumor, lymphangioma, inflammatory myofibroblastic tumor, and thyroid and parathyroid carcinaomas. In a retrospective review of children with mediastinal masses treated at the Mayo Clinic, NHL and HL represented the majority of malignant causes of anterior mediastinal masses, although this review did not include patients with leukemia. In a report on 3721 children with cancer treated at St. Jude Children’s Research Hospital over a 15-year period, 24 children had superior vena cava (SVC) obstruction at initial presentation or with disease recurrence. Patients presenting with SVC obstruction at initial presentation included eight children with NHL, four with ALL, two with HL, one with neuroblastoma, and one with yolk sac tumor. In contrast, in the adult population, small cell bronchogenic carcinoma is the most common underlying diagnosis associated with SVCS.
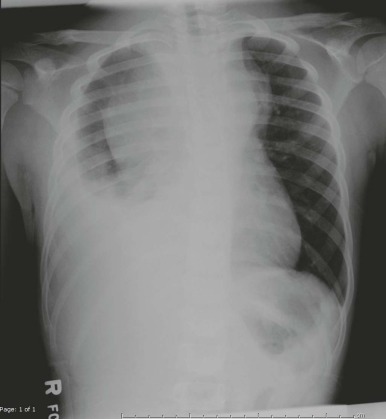
SVCS was first described in a patient with a syphilitic aneurysm of the aorta in 1757. The SVC is a thin-walled vessel with blood flowing under low pressure and is easily compressible by the enlargement of surrounding structures in the thorax. In persons with SVCS, compression and obstruction of the SVC cause restriction of blood return to the right atrium and impaired venous drainage from the upper extremities, head, and neck. Occlusion of the SVC by thrombus—for example, as a complication of a central venous line—can also result in SVCS. Just as the SVC can be compressed by an anterior mediastinal mass, the trachea and main stem bronchi in children are relatively compressible and have smaller intraluminal diameters compared with these structures in adults, making the pediatric patient with an enlarging mass susceptible to respiratory compromise ( Fig. 68-1 ).
Clinical Presentation
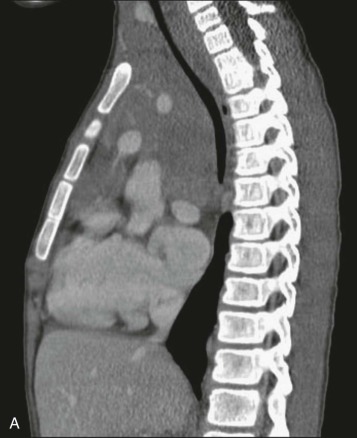
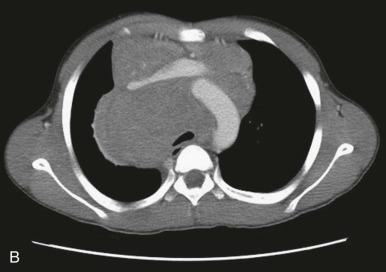
Children with SMS can present with a constellation of signs and symptoms related to SVC and airway obstruction, including swelling, plethora, venous engorgement of the face, neck, and upper extremities, cough, hoarseness, orthopnea, cyanosis, anxiety, syncope, and altered mental status. The child can present in extremis, with shock caused by cardiac compromise, or the child can present with respiratory failure, which more commonly occurs in malignancies with a higher growth fraction such as B- or T-lymphoblastic or mature B-cell lymphomas. Conversely, patients with more indolent tumors, such as HL, can present with impressively large masses and surprisingly little symptomatology ( Fig. 68-2 ). Symptoms are often more profound with the patient in the supine position and may improve in the upright or prone position. In the supine position, thoracic volume is decreased because of decreased rib cage dimension and diaphragm position. In addition, blood flow to the mass itself can increase, causing further expansion of the mass. Therefore pursuing testing such as computed tomography (CT) scanning in the supine position should be carefully considered, and patient tolerance of supine positioning should be evaluated before initiating cross-sectional imaging. Patients who are unable to tolerate supine positioning may require decubitus or prone positioning if CT evaluation is necessary during the initial workup, and some patients with extreme compression may not be able to tolerate any horizontal positioning prior to emergent cytoreductive therapy.

An anterior mediastinal mass may be suspected on the basis of medical history and physical examination and can be identified on a chest radiograph. Identifying patients at greatest risk for anesthetic complications is important in planning the diagnostic evaluation. Procedural sedation and anesthesia pose particular risks for patients with an anterior mediastinal mass, with the potential for cardiopulmonary collapse and death. A patient whose respiratory status is stable while awake, with spontaneous respirations, may experience significant obstruction when anesthesia is induced. General anesthesia alters ventilatory mechanics, decreasing inspiratory muscle tone and functional residual capacity of the lung. Chest wall tone is decreased by neuromuscular blockade. Airways are more compressible because of bronchial smooth muscle relaxation. Positive pressure ventilation may further restrict an already narrowed airway. In addition, general anesthetic agents can have negative inotropic effects and may contribute to decreasing cardiac output, precipitating hypoxemia, hypotension, or even cardiac arrest in patients with compression of the great vessels.
Respiratory symptoms occur in 40% to 60% of children with anterior mediastinal masses, but correlation of symptoms with anesthetic risk can be challenging. Orthopnea has been associated with increased anesthetic risk. Posterior anterior and lateral chest radiographs can demonstrate tracheal compression but may not detect all cases. CT can provide additional anatomic information, and patients with less than 50% of expected tracheal cross-sectional area on CT have been shown to be a high-risk group. However, obstruction distal to the trachea may be more difficult to appreciate. Authors of a prospective study assessing tracheal area and pulmonary function testing have found that inducement of general anesthesia can be used safely in children who meet the minimum criteria of a tracheal area larger than 50% and predicted peak expiratory flow rate (PEFR) greater than 50%. Thirty-one children with mediastinal masses underwent pulmonary function testing in the sitting and supine positions along with CT prior to undergoing 34 surgical procedures. For children with less than 50% of predicted cross-sectional tracheal area (or with less than 50% of PEFR), only local rather than general anesthetic agents were used. Five patients had predicted tracheal areas larger than 50% but had a low PEFR and thus only local anesthetic agents were used. No intraoperative complications occurred in patients receiving anesthetic drugs when these criteria were used.
Evaluation and Imaging
A definitive diagnosis is needed to initiate tumor-specific therapy, and the least invasive diagnostic methods possible should be pursued. If the peripheral blood smear is abnormal, a bone marrow aspiration or biopsy with use of a local anesthetic may prove sufficiently diagnostic. A biopsy of a peripheral lymph node, if present, may prove diagnostic. Serum α-fetoprotein or β-human chorionic gonadotropin may be positive in patients with mediastinal germ cell tumors, and these tests can be performed rapidly in many institutions. If a pleural effusion is present, pleural fluid aspiration may be possible with use of local anesthesia and may provide fluid for pathologic review and immunophenotypic and cytogenetic evaluation ( Fig. 68-3 ). Some patients whose only source of potential diagnostic tissue is the mediastinal mass will be able to tolerate percutaneous needle biopsy with use of a local anesthetic and anxiolytic-only sedation.
An accurate tissue diagnosis allows for initiation of the optimal specific treatment regimen. For many patients with anterior mediastinal masses, a general state of anesthesia will be able to be induced safely. However, the cases of patients in extremis or those with rapidly evolving respiratory compromise who require sedation or inducement of general anesthesia are particularly challenging to manage. When proceeding with general anesthesia, some persons have advocated the preservation of spontaneous respiration when at all possible, avoidance of the use of muscle relaxants, being prepared to change the patient’s position, and having an expert bronchoscopist and emergency cardiopulmonary bypass available. Use of bilevel positive airway pressure for safe anesthesia in these settings has been reported recently. Successful suprasedative dosing of dexmedetomidine has also been reported. Use of extracorporeal membrane oxygenation in persons with anterior mediastinal masses who are experiencing cardiopulmonary failure has been reported. Use of inhaled heliox, a helium-oxygen mixture, has been advocated in case reports to decrease resistance to airflow in obstructed airways, although its use in the setting of anterior mediastinal masses has not been studied.
In recent years the surgical literature has included an increased focus on novel operative techniques to allow safe biopsy and/or resection in patients with large anterior mediastinal masses. Percutaneous biopsy has long been a standard approach in patients too unstable for inducement of general anesthesia, but advances in imaging technique allow for additional applications of this methodology, such as real-time ultrasound fusion with CT for biopsy guidance. Additional modalities recently described as successful in a majority of patients include awake video-assisted thoracoscopic biopsy, the hemiclamshell approach, and the Chamberlain procedure (also known as anterior mediastinotomy).
Treatment
In some clinical situations the risk of proceeding with diagnostic procedures is considered too great, and empiric therapy aimed at reducing the mass must be initiated. Emergent treatment modalities include radiation, intravenous steroids, and combination chemotherapy. Radiation has traditionally been used in emergency management of anterior mediastinal masses, because most lymphomas are radiosensitive. Daily radiation doses are based on those used for the presumed diagnosis that best fits the clinical data. Depending on the radioresponsiveness of the mass, prebiopsy radiation treatment will frequently obscure the tissue diagnosis, even with low-dose radiation therapy. Loeffler and colleagues reported that in 8 of 10 patients younger than 30 years who underwent emergent prebiopsy radiation for a mediastinal mass, a tissue diagnosis could not be established because of uninterpretable pathologic specimens. Also of concern in the pediatric population is a risk of postradiation respiratory deterioration caused by swelling of the trachea after radiation. Concurrent administration of a steroid (e.g., dexamethasone at a dosage for airway edema, 0.5 to 2 mg/kg/day in divided doses every 6 hours) can be considered with the goal of preventing this complication but may also contribute to obscurity of the diagnosis because most untreated lymphomas are sensitive to steroids.
The emergent use of empiric chemotherapy includes the use of intravenous (IV) steroids or combination chemotherapy. Steroid therapy should be considered in the emergent management of proven or presumed ALL or lymphoblastic lymphoma. Careful attention must be directed to fluid management given concerns for TLS in the patient with the potential for cardiovascular compromise. Consideration of the use of rasburicase may be warranted. Steroid dosing based on ALL treatment protocols can be used (e.g., methylprednisolone, 32 to 48 mg/m 2 /day IV, divided every 8 to 12 hours). Steroid therapy may also be effective in the setting of other malignancies because of its potent antiinflammatory effect. Other chemotherapeutic agents for consideration in the emergent management of anterior mediastinal mass include cyclophosphamide, vincristine, and anthracyclines. Just as radiation may eliminate the ability to establish a definitive diagnosis, steroid therapy has also been reported to inhibit accurate diagnosis. Without a definitive pathologic diagnosis, continued treatment of the patient for the presumed diagnosis that best fits the clinical data has been advocated. Masses that are not responsive to the previously described emergent therapies may require surgical intervention.
Conclusions
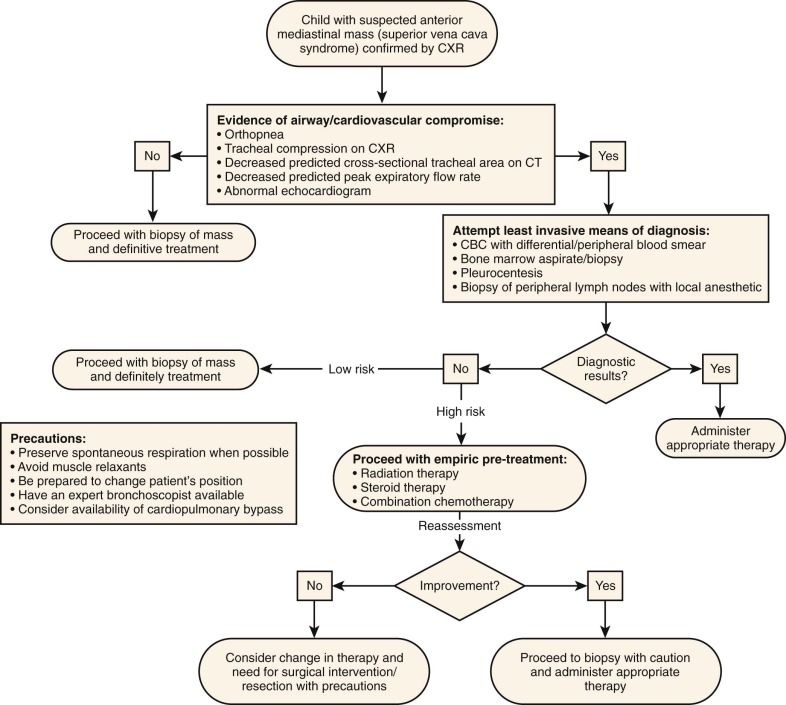
The evaluation of a child with a suspected anterior mediastinal mass or SVCS should be initiated rapidly once the diagnosis is considered to be a possibility. Great variability in the course of evaluation and treatment may be encountered, depending on the specifics of each clinical situation, including the clinical stability of the patient and the underlying diagnosis. A representative evaluation and treatment algorithm published in 2008 is outlined in Figure 68-4 . The clinician must adapt his or her care to the particular circumstances of the individual patient.
Spinal Cord Compression
Spinal cord compression is overall a rare event in children, but several studies have shown an incidence of up to 5% in children with cancer. The most frequent occurrence of spinal cord compression in children with cancer is during the initial presentation of the malignant process (67% of the time). Although such a presentation often occurs with widely disseminated disease, spinal cord compression can also occur with a singular lesion as the initial presentation of a new diagnosis of cancer. In this scenario, along with preservation or restoration of lost neurologic functioning, proper diagnosis of the cancer through adequate tissue sampling is a management priority. Procurement of sufficient tissue for specialized testing and possible study enrollment is a lesser priority but still should be thoughtfully considered in the overall management plan. Spinal cord compression is also encountered in patients with relapsed and refractory disease, again as a single metastatic lesion or as widespread metastatic disease. In such situations, management strategies must take into account the likelihood of cure, the effect of interventions on the patient’s quality of life, and the role of palliation.
The majority (71%) of tumors that cause spinal cord compression are extradural. More than 50% of tumors causing spinal cord compression are found to be neuroblastoma, soft tissue sarcomas, Ewing sarcoma, or rhabdomyosarcoma. A wide variety of other tumors have been reported as causes of spinal cord compression and must be considered in the differential diagnosis. Tumors reported to date include osteosarcoma, Wilms tumor, metastatic central nervous system tumors, meningioma, pleuropulmonary blastoma, juvenile xanthogranuloma, adrenocortical carcinoma, acute myelogenous leukemia (AML), and lymphoma.
Clinical Presentation
Back pain is the most common presenting sign of spinal cord compression and was present in 94% of 70 pediatric patients presenting with solid tumors and spinal cord compression. Because recurrent back pain without known injury is a fairly uncommon complaint in children, spinal lesions and spinal cord compression should always be considered in the differential diagnosis of this complaint, even in the absence of neurologic findings. In pediatric patients known to have cancer, the index of suspicion must be particularly high. Any delay in diagnosis of spinal cord compression may contribute significantly to morbidity. A thorough physical examination should accompany a complete history. The history should include characterization of pain, including onset and duration. Neurologic findings, if present, will vary, depending on the spinal level of the lesion and the degree of compression. In their series of children with systemic cancer and spinal cord disease, Lewis and coworkers noted that localized spine tenderness is the most reliable clinical finding. A spectrum of ataxia, gait disturbance, and paraplegia can be observed. Sphincter dysfunction is most commonly seen as urinary retention or constipation. Localization of the level of epidural cord compression along the spine is suggested by the specific effects on strength, tendon reflexes, sensory level, Babinski reflex, sphincter abnormalities, and rate of progression ( Table 68-1 ).
| Sign | LOCATION | ||
|---|---|---|---|
| Spinal Cord | Conus Medullaris | Cauda Equina | |
| Weakness | Symmetrical; profound | Symmetrical; variable | Asymmetrical; may be mild |
| Tendon reflexes | Increased or absent | Increased knee; decreased ankle | Decreased; asymmetrical |
| Babinski | Extensor | Extensor | Plantar |
| Sensory | Symmetrical; sensory level | Symmetrical; saddle | Asymmetrical; radicular |
| Sphincter abnormality | Spared until late | Early involvement | May be spared |
| Progression | Rapid | Variable; may be rapid | Variable; may be slow |
Evaluation and Imaging
When the possibility of spinal cord compression is considered, immediate evaluation should be undertaken. Spinal radiography is often the first-choice modality because it is quickly and easily undertaken. Plain films may be able to provide valuable clinical information rapidly, but one must be aware that they are positive in only approximately 30% of patients with spinal cord compression. The presence of a compressive spinal lesion may be seen on a radiograph as a paraspinal soft tissue mass. In cases of neuroblastoma, calcifications are often present within the paraspinal mass. Classically, neuroblastoma invading the spinal canal and intervertebral foramina may be visualized as a dumbbell-shaped lesion surrounding the spine. Other tumors can also have this appearance, emphasizing the importance of obtaining adequate tissue for diagnosis ( Fig. 68-5 ). In many spinal lesions, lytic or sclerotic changes in adjacent bone are often observed on a radiograph. Diagnostic widening of interpeduncular distances and enlargement of the neural foramina can sometimes also be observed on radiographs.
Emergent magnetic resonance imaging (MRI) is essential when plain radiographs are negative and clinical suspicion still exists. MRI is also necessary for further delineation of lesions observed on plain radiographs. MRI has become widely accessible and thus it has replaced the previously used techniques of radionucleotide bone scanning, spinal CT with intrathecal contrast, and lumbar myelography. MRI offers the advantage of increased accuracy and a less invasive nature than previously used modalities. Careful comparison of axial, coronal, and sagittal images can help categorize spinal tumors as extradural, intradural-extramedullary, or intramedullary, thereby aiding in the differential diagnosis. MRI sequences with and without use of gadolinium should be obtained for patients being evaluated for spinal cord compression. Contrast enhancement is less helpful for extradural tumors than for intradural-extramedullary or intramedullary lesions. Fat suppression techniques are most helpful for evaluating extradural lesions. It should also be remembered that it is important to image the entire spine to identify the possibility of involvement at several levels, even if the physical examination has localized the most likely area of compression.
MRI can also accurately detect compression and dislocation of the spinal cord. Coronal images can elucidate the degree of cranial and caudal extension on the intraspinal component of a mass lesion. Because of increased cellularity and high nuclear-to-cytoplasmic ratio, lesions are usually isodense to hypodense on T2-weighted images. Cord edema can be seen as a hyperintense signal in T2-weighted images. Contrast enhancement is generally marked in the presence of intraspinal lesions. Diffusion tensor imaging has been recently described as a potentially beneficial additive sequence.
If the clinical situation allows time for further radiologic evaluation, CT with bone windows can provide complementary information to MRI findings. Detailed information on bone architecture and cortical appearance is better obtained via CT.
Treatment
Treatment options for a patient presenting with spinal cord compression include surgery (laminectomy or laminotomy), chemotherapy, steroid therapy, and radiation therapy. The best choice of initial therapy will vary depending on the clinical situation. In many clinical situations use of more than one modality may be appropriate, and in some situations it is appropriate for more than one modality to be used simultaneously. The complexity of the choice of the best initial therapy stems largely from multiple competing therapeutic goals—the desire to act quickly to prevent further loss of neurologic function, the need to make an accurate diagnosis with sufficient biologic information for assignment of appropriate treatment, the hope of regaining any lost function existing at presentation, and the concern regarding the late effects of the available emergent treatment modalities. After providing a brief overview of the basic treatment options, we will present the conclusions of several clinical studies.
Surgery
Laminectomy is the most common surgical approach to relieve spinal cord compression caused by a solid tumor. Laminectomy involves removal of the bony arch, or lamina, of a vertebra. This procedure is often combined with biopsy and/or debulking of the tumor. The advantage of a laminectomy is the rapid decompression of the spinal cord. Laminectomy is generally accepted as the most likely modality to stabilize or improve lost neurologic function. Another significant advantage of laminectomy is the procurement of tumor tissue for proper histologic diagnosis and the option of obtaining tissue for relevant biology studies or possible protocol enrollment. The disadvantage of laminectomy is the high risk for late effects, with a significant percentage of patients experiencing kyphosis or scoliosis. Depending on the degree of spinal cord injury and how long symptoms have been present, laminectomy may not provide an advantage compared with treatment with chemotherapy only and may expose the child to risks of the surgical procedure and greater risks of long-term orthopedic sequelae.
The technique of osteoplastic laminotomy involves removal of part of the lamina above and below an affected nerve, with subsequent bracing for a period of 6 to 8 weeks. Although historically this technique is not commonly used, it may offer a decreased incidence of long-term sequelae and may be considered by the surgeon. Minimally invasive thoracoscopic approaches for decompression have been described. Additionally, percutaneous approaches for decompression are increasing reported.
Chemotherapy
Chemotherapy can be used successfully to reduce some neoplastic masses that cause spinal cord compression. Use of chemotherapy has been most studied in cases of neuroblastoma or Ewing sarcoma that present with spinal cord compression, and chemotherapy has been used successfully to treat germ cell tumors as well. In the rare cases of leukemic or lymphomatous infiltrates, chemotherapy is the clear standard initial therapy once the diagnosis has been established.
Advantages of upfront chemotherapy for lesions causing spinal cord compression are that chemotherapy is anatomically noninvasive and long-term surgical consequences may be avoided. In addition, patients may proceed with tumor-specific therapy without the need for delay from postoperative healing. However, the use of chemotherapy depends on first establishing an accurate pathologic diagnosis. In cases of metastatic tumor, it may be possible to make a diagnosis by biopsy of other primary or metastatic lesions. Identifying isolated paraspinal masses can be more diagnostically difficult. If surgery is avoided completely and chemotherapy is started presumptively, the possibility of an incorrect diagnosis will exist. Also, if no tissue is obtained, even though an isolated paraspinal mass may eventually be diagnosed as neuroblastoma on the basis of elevated urine catecholamine levels or positive metaiodobenzylguanidine activity, the opportunity to obtain valuable biologic information, such as histology, v-myc avian myelocytomatosis viral oncogene neuroblastoma derived homolog (MYCN) status, and other cytogenetic and molecular markers used to direct therapy could be lost.
In patients who experience spinal cord compression in the setting of relapsed disease, the choice of using chemotherapy rather than surgery or radiation therapy can become more complex, because the rate and overall likelihood of response can be substantially decreased in previously treated patients. Radiation therapy or surgeries may have been used previously and may limit these options in a relapse setting. Such clinical situations must be carefully weighed individually. The decision to initiate chemotherapy rather than to use surgery, radiation therapy, or comfort palliation must include consideration of the patient’s overall chance of long-term cure, the history of treatment and response, and most importantly, the goals of treatment of the patient and family and the desired level of intensity of therapy.
In the patient with a new presentation of cancer, the choice of a chemotherapy regimen must be based on a clear identification of the malignant process. Once a diagnosis is known and a decision has been made to avoid surgery or radiation and commence with chemotherapy, standard frontline therapy for the malignancy that has been identified is accepted as the best initial chemotherapy in patients with spinal cord compression.
Dexamethasone
Dexamethasone is routinely recommended throughout the literature for the treatment of pediatric spinal cord compression. It is theorized that decreased swelling of the cord will help minimize neurologic damage through minimizing the effect of vasogenic edema and venous congestion. In a review of the adult literature compiled to create an evidence-based guideline for the emergency treatment of extradural spinal cord compression in adult patients, Loblaw and Laperriere reviewed the available studies on the use of dexamethasone. They found strong evidence to support the use of high-dose dexamethasone with radiation therapy with improved outcome but significant associated toxicity. They also reported evidence judged to be fair that dexamethasone does not need to be given to asymptomatic ambulatory patients with spinal cord compression receiving x-ray therapy. Although no corresponding evidence is available to support the role of dexamethasone in improving outcome in spinal cord compression caused by solid tumors in pediatric patients, little substantial detrimental effect has been observed with the routine use of dexamethasone in the setting of acute spinal cord compression. Increased rates of infection and delayed wound healing are possible concerns. The use of gastric protectants is advised when administering dexamethasone in this setting and should include H2 blockers and/or proton pump inhibitors. Blood pressure and glucose levels should be monitored, and any increase above age norms should be treated appropriately.
In the case of a child presenting with a high index of suspicion of spinal cord compression caused by tumor and any neurologic compromise, rapid administration of dexamethasone at 1 to 2 mg/kg IV to be administered over 30 minutes is suggested. This dose should be administered prior to imaging. If no neurologic symptoms are present, lower oral dosing, 0.25 to 0.5 mg/kg every 6 hours, may be initiated while imaging is arranged. If the child is found to have a spinal lesion and radiation therapy is chosen as the modality for treatment of cord compression, use of moderate-dose dexamethasone is sometimes continued to decrease the effects of radiation-induced swelling and vasogenic edema.
Radiotherapy
Most tumors causing spinal cord compression in the pediatric population will respond to radiotherapy, and thus this modality can often be used emergently to relieve symptoms of spinal cord compression effectively. The benefits of its use include possible rapid response, reasonable tolerance, and avoidance of more invasive procedures. Potential drawbacks include interference with establishing a correct pathologic diagnosis, long-term effects on the spinal column (including stunted growth, cord damage, and scoliosis), thyroid dysfunction, and the risk of secondary malignancies. Additionally, young pediatric patients may require anesthesia for planning and delivery of radiotherapy fractions. For safety and accuracy, adequate imaging, usually through MRI, is required to define the length, depth, and width of the planned treatment field.
Several approaches can be used to deliver a therapeutic treatment dose. Midline lesions, as well as lesions in the lumbar spine, may be treated through a parallel opposed anteroposterior-posteroanterior beam arrangement. A posterior field alone can be used for lesions in the thoracic spine. The cervical spine may be treated with opposed lateral fields in an effort to avoid the oral cavity. Conformal techniques can allow for more precise localization of the radiation therapy to the tumor volume. However, these methods require more elaborate planning and may delay the initiation of therapy. The potential benefit of a more conformal field must be weighed against the additional time and planning required.
Radiotherapy dose and schedule are determined by the pathologic diagnosis, extent of spinal involvement, possible coadministration of chemotherapy, and the patient’s clinical situation (e.g., previously ambulatory or nonambulatory, overall life expectancy, likelihood of response to systemic therapy, and previous history of radiation therapy). A dosing range of 18 to 40 Gy can be used. Commonly used schedules include 30 Gy delivered in 10 fractions, 20 Gy given in 5 fractions, or up to 35 to 40 Gy spread over a more protracted course of 3 to 4 weeks. However, 8-Gy single fraction radiotherapy has been successfully used for palliation in patients with a grim prognosis and spinal cord compression, and a recent systematic review suggested that 30 Gy over 10 fractions was a preferred regimen for patients with a good prognosis.
The concomitant use of dexamethasone with radiation therapy is often recommended. Dexamethasone is usually begun at a high dose prior to starting radiotherapy, continued at a lesser dose during the procedure, and tapered off after completion. The use of dexamethasone with a taper is thought to decrease some of the edema that can occur as a result of the radiotherapy. Although newer radiation modalities such as Gamma Knife radiosurgery and CyberKnife radiosurgery have been used in adult patients with spinal cord compression, pediatric use of these technologies for spinal cord compression has not been reported to date.
Clinical Studies
Although much laudable investigation has been undertaken to address the best choice of initial therapy for children with spinal cord compression, no consensus has been reached to date. The available data are limited by relatively small sample sizes because of the overall rarity of spinal cord compression. Larger series tend to group multiple histologic diagnoses, which may respond differently to the interventions of chemotherapy, radiation, and surgery. Many of the studies also span long intervals and cannot necessarily take into account modernization of practice, improved imaging modalities, and improved therapeutic options.
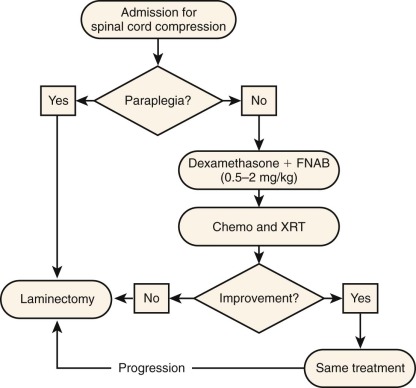
Pollono and colleagues have reported on a review of 70 pediatric patients with spinal cord compression treated at a single institution from 1984 to 2001; 71% were extradural tumors and 54% were soft tissue sarcomas or neuroblastoma. Figure 68-6 presents the algorithm by which these patients were diagnosed and treated. Patients with paraplegia of less than 96 hours’ duration underwent laminectomy, as did patients with primary spinal cord tumor or isolated recurrence of main tumor, progression of symptoms after radiation or chemotherapy, or, if known, a radiation- and chemotherapy-resistant tumor. Surgery was avoided when the prognosis of the primary tumor was poor or risks were thought to outweigh benefits. Of patients with paraplegia, 57% became ambulatory after undergoing a laminectomy, whereas only 14% improved after use of chemotherapy and radiation. In patients who presented without paraplegia and received medical treatment without surgery, 87% remained ambulatory. Overall survival was related to the original malignancy. These data suggest that patients with chemotherapy- and radiotherapy-sensitive tumors without evidence of progressive neurologic damage might be treated with chemotherapy and/or radiation therapy but that any indication of neurologic progression is a strong indication for laminectomy without delay.
The question of upfront chemotherapy versus surgery or radiation therapy for spinal cord compression has been most extensively studied in pediatric patients with neuroblastoma, because rapid improvement with initial chemotherapy has been demonstrated. The first reports on the use of chemotherapy as frontline therapy in children presenting with dumbbell neuroblastoma was published in 1984 by Hayes et al. Eleven children with presenting neurologic symptoms were treated; 9 had complete recovery and 2 demonstrated sustained paraplegia. In 1989, Sanderson and coworkers also reported encouraging results of two children with paraplegia and two with paraparesis who both demonstrated full neurologic recovery after treatment with vincristine, cisplatin, etoposide, and cyclophosphamide therapy. Since then, more investigations have been conducted on the use of frontline chemotherapy compared with neurosurgical intervention. Both prospective and retrospective studies have attempted to elucidate whether chemotherapy or surgery is the best initial choice of therapy, but the results yielded have conflicting conclusions. The variability in outcome likely reflects the difficulty of appropriately grouping patients based on initial presenting signs and extent of disease, as well as institutional practice variations and availability of resources.
A series of these studies has been well summarized by De Bernardi and colleagues. Studies from several major international cooperative groups (Italy, Germany, Poland, United Kingdom, and United States) were reviewed. The Italian Cooperative Group for Neuroblastoma has reported a prospective study in which 26 patients with neuroblastoma and spinal cord compression were preferentially given chemotherapy rather than surgery or radiation therapy. Prior to this study, patients followed up between 1979 and 1998 by this group had been analyzed. Retrospective analysis of the earlier patient group showed that radiotherapy, laminectomy, and chemotherapy have comparable ability to improve spinal cord compression. The prospective study also concluded that chemotherapy adequately relieves neurologic symptoms in most patients. Neurologic and orthopedic sequelae were seen in a large number of patients, including those who received chemotherapy only.
In another prospective study carried out by the French Society of Paediatric Oncology, 78 patients with intraspinal extension of neuroblastoma were treated, beginning in 1990. Of these patients, 86% were initially treated with chemotherapy; 63% had a complete neurologic recovery and 21% had a partial neurologic recovery. Also, 13% failed to improve with chemotherapy, the disease of 1 patient progressed, 14% underwent primary surgery, 23% of initially symptomatic patients (10 of 43) had severe neurologic sequelae, 16% had mild sequelae, and 23% were reported to have severe orthopedic sequelae.
One of the largest series contained 83 patients registered for the Pediatric Oncology Group Neuroblastoma Biology Protocol 9047, in which 66 patients received upfront chemotherapy, 23 underwent primary laminectomy, 8 received initial radiotherapy, and 31 underwent initial surgical resection without laminectomy. From this series of retrospectively collected data, it was concluded that patients with neurologic symptoms can be managed without laminectomy and that laminectomy should be reserved for patients who demonstrate progressive neurologic deterioration after initial chemotherapy. The likelihood of neurologic sequelae was found to be inversely related to the length of symptoms prior to initiation of treatment, and more severe sequelae were seen in patients presenting with more severe symptoms. Twenty-nine percent of patients had orthopedic sequelae, and the incidence was significantly lower in patients managed with chemotherapy compared with those who underwent laminectomy.
After a review of multiple studies, the primary conclusion of the neuroblastoma workshop was that a need exists for the creation of an international spinal cord compression registry. The workshop group specified the need to standardize language and criteria to group patients with comparable degrees of neurologic compromise at presentation accurately and to characterize the late effects uniformly. A functional grading system proposed by Gilbert and colleagues was used in some studies to aid in outcome analysis, but it has not been universally adopted ( Table 68-2 ). This grading system notably does not take into account sphincter dysfunction, an important neurologic finding.
| Grade | Patient Function |
|---|---|
| 1 | Ambulatory, with or without weakness of lower extremities or ataxia |
| 2 | Not ambulatory, but able to lift legs against gravity when supine |
| 3 | Paraplegic and unable to move legs against gravity |
It was also concluded that chemotherapy is a reasonable initial approach, but laminectomy should be used for patients who demonstrate progressive neurologic symptoms after presentation or no restoration in the short term. Patients with symptoms occurring within 96 hours of presentation may also be considered candidates for surgical intervention. It was also proposed that radiotherapy may be underused in spinal cord compression caused by neuroblastoma.
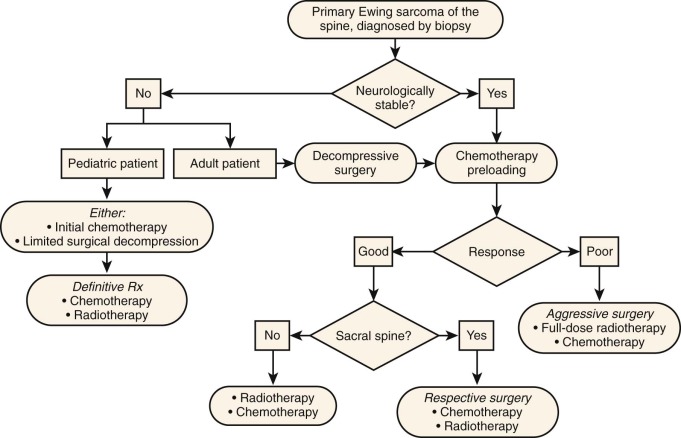
Because of the heterogeneity of pediatric malignancies, the findings of the neuroblastoma group cannot automatically be generalized for other types of cancer. Smaller series of outcomes of patients with Ewing sarcoma and intraspinal lesions have also described mixed use of upfront therapies, with variable outcomes. In a report of a series of seven patients with primary Ewing sarcoma of the spine, as well as a review of the literature, Sharafuddin and colleagues noted the importance of the location (sacral or nonsacral) of the presenting lesion in the response to treatment. Differences in response in adult and pediatric patients were also noted. Figure 68-7 shows the algorithm that they proposed for the management of primary Ewing sarcoma of the spine. Initial chemotherapy or limited surgical decompression is recommended for all pediatric patients; surgical intervention is clearly indicated for patients who demonstrate progressive findings with medical treatment.
The rare cases of leukemia and lymphoma that present with spinal cord compression should be treated with initial chemotherapy. Radiotherapy can be considered for some special cases.
Late Effects
The long-term consequences of spinal cord compression and its management include neurologic compromise and orthopedic effects, and the choice of initial management heavily takes into account an attempt to minimize those late effects. A number of reports have supported the observation that neurologic sequelae are most related to the degree of presenting neurologic compromise; the more severe the neurologic compromise at presentation, the more likely long-term sequelae will be seen. Other studies have also shown that the duration of time between the development of neurologic symptoms and the initiation of therapy is inversely correlated with the degree of neurologic recovery. Although laminectomy may be the most likely method to correct or prevent neurologic sequelae, it is clearly not necessary in all cases of spinal cord compression and carries the greatest risk of orthopedic sequelae. For patients with neuroblastoma in particular, the initial treatment for spinal cord compression did not demonstrate a clear impact on late effect frequency at a median follow-up of 8 years. Orthopedic sequelae include scoliosis, growth impairment, and spinal instability. Orthopedic sequelae are related to the spinal level of the lesion and the age of the patient at treatment. The most severe sequelae are seen in patients with thoracic versus lumbar lesions and age younger than 12 months. Although a wide range of rates of sequelae has been reported, almost all studies have supported the observation of significantly greater orthopedic sequelae in patients who undergo surgery. In a study by Hoover and coworkers, a scoliosis incidence of 67% was found in patients who underwent laminectomy compared with an incidence of 36% in patients who did not undergo laminectomy. Correlation between late effects, including differences in health-related quality of life and initial treatment, remain at the center of investigations into the optimal choice of initial therapy.
Hyperleukocytosis and Leukostasis
Hyperleukocytosis is a clear, life-threatening pediatric oncologic emergency that requires immediate initiation of appropriate therapy. Marked metabolic abnormalities and mild to severe pulmonary and neurologic symptoms can accompany the initial finding of hyperleukocytosis or can develop shortly after presentation. Stasis of leukemic cells within blood vessels and migration of blast cells into tissues can lead to the clinical entity of leukostasis, with mortality rates reported as high as 20% to 40%. Despite the relative frequency of this true oncologic emergency and several varied therapeutic options, no data-driven management guidelines are available because of overall small numbers of patients, variability of clinical presentation, molecular differences in the underlying disease, and the difficulty of conducting randomized clinical trials with these constraints.
Definition
Hyperleukocytosis is generally accepted as a white blood cell (WBC) count higher than 100 × 10 9 /L, although some studies have included patients with a WBC count higher than 50 × 10 9 /L. It is estimated to occur in 5% to 20% of childhood leukemias and is seen most frequently in infant ALL and T-cell ALL. Hyperleukocytosis is more associated with certain subtypes of ALL and AML and is seen in almost all cases of childhood chronic myelogenous leukemia ( Table 28-2 ). It has long been observed that the occurrence of leukostasis does not correlate directly with degree of hyperleukocytosis. Although hyperleukocytosis is seen more frequently in children with ALL, clinical leukostasis is seen more frequently in children with AML, particularly in M4 and M5 French-American-British subtypes. Clinical symptoms from hyperleukocytosis generally occur at WBC counts higher than 200 to 300 × 10 9 /L in patients with AML and higher than 400 to 500 × 10 9 /L in patients with ALL. However, symptoms of leukostasis have also been observed in patients with WBC counts lower than 100 × 10 9 /L.
Clinical Presentation
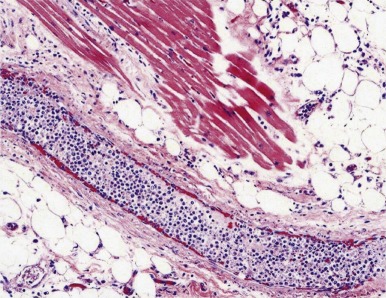
Clinical symptoms of leukostasis are dependent on the system affected. Any small vessels, and therefore any organs, can be at risk. Although pulmonary and neurologic symptoms are most often observed, involvement of many other systems has also been reported. Hyperleukocytosis has been reported to result in leukostasis, causing renal failure, papilledema, dactylitis, priapism or clitorism, acute myocardial infarction, and cardiac failure. Histopathologic studies have demonstrated aggregates of blast cells and thrombi leading to occlusion of small vessels in multiple organs of patients with hyperleukocytosis. Corresponding to observed clinical symptoms, postmortem examinations have shown profound infiltration of multiple organs, including brain (including retinal vessels), liver, spleen, adrenals, and pulmonary vasculature, and in the alveoli and connective tissue of the lungs. A striking image of leukemic infiltration of a coronary artery was obtained by Thornton and Levis ( Fig. 68-8 , Table 68-3 ).