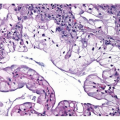
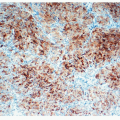
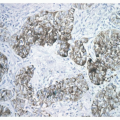
carcinomas (BAC).11,12,13 and 14 However, studies that applied strict 2004 WHO definition of BAC failed to demonstrate this association. Subsequently, several reports showed that mixed subtype of invasive adenocarcinoma with a BAC component were commonly associated with EGFR mutations, although other studies did not confirm this correlation. More recent studies found a link between papillary differentiation and EGFR mutations.15,16 In our experience, there is a large overlap in histologic growth patterns between different adenocarcinomas with a different mutational profile. EGFR mutations are extremely rare in large cell carcinomas, small cell carcinomas, and large cell neuroendocrine carcinomas.
Table 22-1 Investigational agents in non-small cell lung cancer | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
trials demonstrated that tumors harboring BRAF mutation (V600E) showed regression induced by a specific MEK inhibitor, CI-104045.24,25
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree