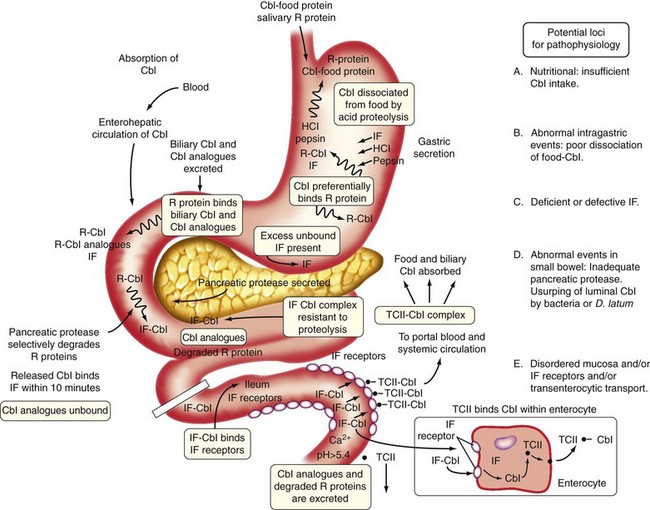
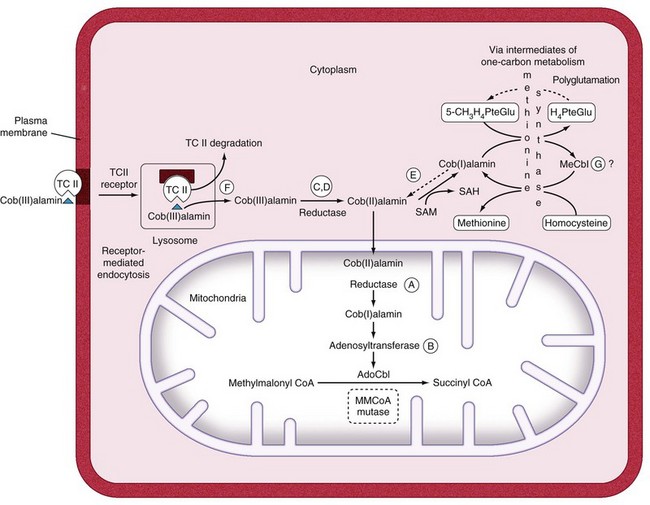
Chapter 12 Megaloblastic Anemias
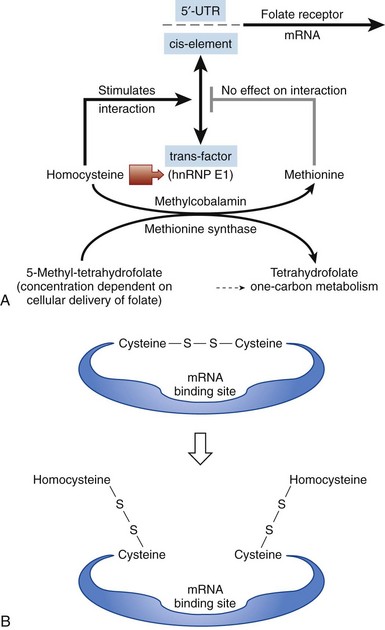
Figure 12-3 MODEL FOR HOW THE CELL SENSES FOLATE DEFICIENCY AND RESPONDS BY UPREGULATING FOLATE RECEPTORS.
(From Tang YS, Khan RA, Zhang Y, et al: Incrimination of heterogeneous nuclear ribonucleoprotein E1 (hnRNP-E1) as a candidate sensor of physiological folate deficiency. J Biol Chem 286:39100, 2011.)
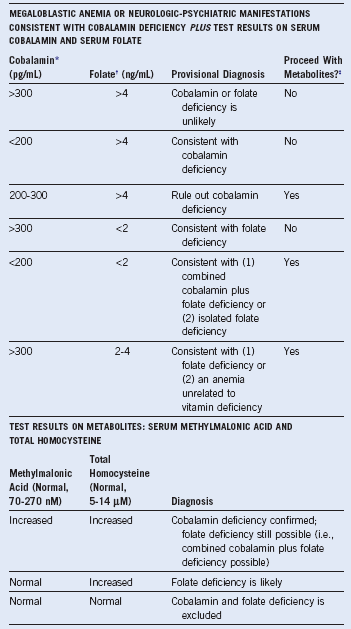
Serum Homocysteine and Methylmalonic Acid Levels in Cobalamin and Folate Deficiencies
The combined use of homocysteine and methylmalonic acid (MMA) levels can differentiate cobalamin from folate deficiency, because most patients with folate deficiency have normal MMA levels, and the remainder have only mild elevations.2 These two tests are useful diagnostically. The abnormally high levels of metabolites return to normal only when the patient receives replacement with the appropriate (deficient) vitamin. A positive response to cobalamin, documented by falling levels of homocysteine and MMA, is evidence of cobalamin deficiency. Conversely, therapy with folate results in a decrease in the isolated homocysteine level if folate deficiency is present.2 Indeed, because several variables that are not related to vitamin deficiency (such as age, mild renal dysfunction) can falsely elevate serum homocysteine and MMA levels, if there is ambiguity, proof of vitamin deficiency would require clear-cut demonstration of a reduction in metabolite levels after specific vitamin supplementation.2,3
Modified Therapeutic Trials
The traditional therapeutic trial using physiologic doses of vitamins (100 mcg of folate or 1 mcg of cobalamin given daily while monitoring the reticulocyte response)1 has given way to a modified therapeutic trial. Rather than making the diagnosis of a deficiency, the intention is often to confirm the clinical suspicion that the patient does not have deficiency. This can be demonstrated by lack of response to full replacement doses of both vitamins (1 mg of folic acid orally for 10 days and 1 mg of cobalamin intramuscularly or subcutaneously daily for 10 days). Clinical scenarios in which such trials may be applicable (after drawing blood for serum cobalamin and folate levels) are as follows:
1. There is a clinical suspicion that the underlying disease is not caused by a vitamin deficiency, but this idea is not supported by results of clinical, morphologic, and biochemical evaluations. Such conditions include anemia with a megaloblastic bone marrow that may be secondary to chemotherapy, myelodysplastic syndromes, or acute leukemia; when time is of the essence in making the diagnosis; when the levels of cobalamin are likely to be falsely abnormal because of these diseases; or when there is underlying dehydration or renal dysfunction that predictably gives falsely high levels of metabolites.
2. In other situations (i.e., pregnancy, acquired immunodeficiency syndrome [AIDS], or alcoholism) with a multifactorial basis for anemia, the response or lack thereof to full replacement doses can eliminate cobalamin or folate deficiency and thereby narrow the (often extensive) differential diagnosis.
3. In instances when severe anemia with megaloblastosis is clinically obvious and so serious that the physician cannot wait for the results of specific tests for deficiency. Full doses of both vitamins are administered, and if there is a response manifested by brisk reticulocytosis by days 5 to 7, retrospective assignment of the deficiency is based on the results of blood samples drawn before beginning the trial.
Table 12-2 Serum Cobalamin: False-Positive and False-Negative Test Results
| FALSELY LOW SERUM COBALAMIN IN THE ABSENCE OF TRUE COBALAMIN DEFICIENCY |
| FALSELY RAISED COBALAMIN LEVELS IN THE PRESENCE OF A TRUE DEFICIENCY* |
IF, Intrinsic factor; TC, transcobalamin.
*Although a low serum cobalamin level is not synonymous with cobalamin deficiency, 5% of patients with true cobalamin deficiency have low-normal cobalamin levels, a potentially serious problem because the patient’s underlying cobalamin deficiency will progress if uncorrected.
Diagnosing Folate Deficiency
When combined with a clinical picture of megaloblastic anemia and additional results of cobalamin levels, the serum folate concentration is the cheapest and most useful initial biochemical test to diagnose folate deficiency2 (see Table 12-1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree