18.1 Introduction
The fetal origins hypothesis proposes that alterations in fetal nutrition and endocrine status result in developmental adaptations that permanently change structure, physiology and metabolism, thereby predisposing to cardiovascular, metabolic and endocrine disease in adult life. Coronary heart disease (CHD) is now thought to be a consequence of fetal adaptations to undernutrition that are beneficial for short-term survival, even though they are detrimental to health in postreproductive life.
In fetal life the tissues and organs of the body go through critical periods of development. These may coincide with periods of rapid cell division. In common with other living creatures, humans are plastic in their early life, and are moulded by the environment. Although the growth of a fetus is influenced by its genes, studies in humans and animals suggest that it is usually limited by the environment, in particular the nutrients and oxygen it receives from the mother. There are many possible evolutionary advantages in the body remaining plastic during development, rather than having its development driven only by genetic instructions acquired at conception.
Programming describes the process whereby a stimulus or insult at a critical period of development has lasting or lifelong effects. Experimental studies in animals have documented many examples of fetal programming, with recent studies showing that alterations in maternal nutrition can have long-term effects on the offspring that are of relevance to human cardiovascular disease. These long-term effects of alterations in maternal nutrition include changes in vascular structure and function, insulin secretion, renal development, and glucose and cholesterol metabolism. For example, feeding pregnant rats a low-protein diet results in lifelong elevation of blood pressure in the offspring; rats whose mothers had been fed a diet with a low ratio of protein to energy during pregnancy also exhibited a permanently altered balance between hepatic glucose production and utilization.
Although some effects of maternal nutrition may be direct consequences of alterations in substrate availability, others are thought to be mediated by hormonal effects. Experiments in animals have shown that alterations in maternal and fetal hormonal status can have important long-term consequences. Substrate availability and hormonal milieu may alter the development of specific fetal tissues during critical periods of development, or may lead to long-lasting changes in hormone secretion or tissue hormone sensitivity. The fetal hypothalamus is implicated as a key site that can be programmed by transient changes in prenatal endocrine status.
18.2 Observations establishing the link between size at birth and later cardiovascular disease
Ecological observations
At the start of the twentieth century the incidence of CHD rose steeply in Western countries to become the most common cause of death. In many of these countries the steep rise has been followed by a fall over recent decades that cannot be accounted for by changes in adult lifestyle. The incidence of CHD is now rising in other parts of the world to which Western influences are extending, including China, India and eastern Europe.
An important clue suggesting that CHD might originate during fetal development came from studies of death rates among newborn babies in Britain during the early 1900s. The usual certified cause of death in newborn babies at that time was low birth weight (LBW). Death rates in the newborn period differed considerably between one part of the country and another, being highest in some of the northern industrial towns and the poorer rural areas in the north and west. This geographical pattern in death rates closely resembled today’s large variations in death rates from CHD, variations that form one aspect of the continuing north–south divide in health in Britain. One possible conclusion suggested by this observation was that low rates of growth before birth are in some way linked to the development of CHD in adult life. The suggestion that events in childhood influence the pathogenesis of CHD was not new. A focus on intrauterine life, however, offered a novel point of departure for research.
Cohort studies of birth weight and coronary heart disease
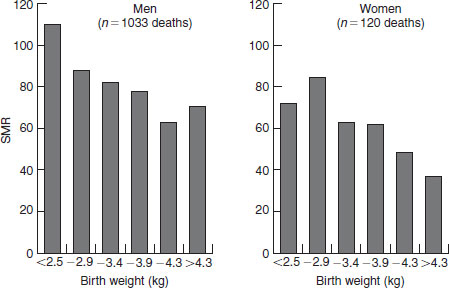
More direct evidence that an adverse intrauterine environment might have long-term consequences came from follow-up studies of men and women in middle and late life whose body measurements at birth had been recorded. A study of men and women born in Hertfordshire, UK, showed for the first time that those who had had low birth weights had increased death rates from CHD in adult life. Thus, among 15 726 men and women born during 1911–1930, death rates from CHD fell progressively with increasing birth weight in both men and women (Figure 18.1). A small rise in the highest birth weights in men could relate to the macrosomic infants of women with gestational diabetes. Another study, of 1586 men born in Sheffield, UK, during 1907–1925, showed that it was particularly people who were small at birth as a result of growth retardation, rather than those born prematurely, who were at increased risk of the disease.
Replication of the UK findings has led to wide acceptance that low rates of fetal growth are associated with CHD in later life. For example, confirmation of a link between LBW and adult CHD has come from studies of 1200 men in Caerphilly, South Wales, and of 70 297 nurses in the USA. The latter study found a two-fold fall in the relative risk of nonfatal CHD across the range of birth weight. Similarly, among 517 men and women in Mysore, south India, the prevalence of CHD in men and women aged 45 years or older fell from 15% in those who weighed 2.5 kg or less at birth to 4% in those who weighed 3.2 kg or more.
Body proportions at birth and cardiovascular disease
The Hertfordshire records and the Nurses and Caerphilly studies did not include measurements of body size at birth other than weight. The weight of a newborn baby without a measure of its length is as crude a summary of its physique as is the weight of a child or an adult without a measure of height. The addition of birth length allows the thin baby to be distinguished from the short, fat baby. With the addition of head circumference the baby whose body and trunk are small in relation to its head, as a result of brain-sparing, can also be distinguished. Thinness, shortness and a small trunk are thought to reflect differing fetal adaptations to undernutrition, hypoxia and other influences, and they have different long-term consequences.
Figure 18.1 Death rates from coronary heart disease, expressed as standardized mortality ratios (SMR), in 10 141 men and 5585 women born in Hertfordshire, UK, according to birth weight. Derived from Osmond et al. (1993), with permission from the BMJ Publishing Group.
In Sheffield, death rates for CHD were higher in men who had had a short crown–heel length at birth. The mortality ratio for CHD in men who were 47 cm or less in length was 138 compared with 98 in the remainder. Thinness at birth, as measured by a low ponderal index (birth weight/length3), was also associated with CHD. Table 18.1 shows data for a group of men born in Helsinki, Finland. While LBW was associated with raised death rates for CHD, there was a stronger association with thinness at birth, especially in men born at term. Men who had a low ponderal index had death rates that were twice those of men who had a high ponderal index.
In Finland, raised death rates from CHD were associated with low placental weight. In Sheffield, however, CHD did not vary with placental weight, but showed a U-shaped relation with the ratio of placental weight to birth weight, the highest mortality ratios being at either end of the distribution. The pattern of body proportions at birth that predicts death from CHD may be therefore summarized as a small head circumference, shortness or thinness, which reflect retarded fetal growth, and either low placental weight or an altered ratio of placental weight to birth weight. The pattern for stroke, which has been reported in Sheffield and Helsinki, is different. Whereas stroke was similarly associated with LBW it was not associated with thinness or shortness. Instead, there were increased rates among men who had a low ratio of birth weight to head circumference. One interpretation of these associations is that normal head growth has been sustained at the cost of interrupted growth of the body in late gestation.
Table 18.1 Standardized mortality ratios (SMR) for coronary heart disease in 3302 Finnish men during 1924–1933
| Birth weight (kg) | SMR (no. of deaths) |
| ≤2.5 | 84 (11) |
| 2.6–3.0 | 83 (44) |
| 3.1–3.5 | 99 (124) |
| 3.6–4.0 | 76 (80) |
| >4.0 | 66 (27) |
| All | 85 (286) |
| p-Value for trend | 0.09 |
| Term babies only Ponderal index at birth (kg/m3) | SMR (no. of deaths) |
| ≤25.0 | 116 (59) |
| 25.1–27.0 | 105 (88) |
| 27.1–29.0 | 72 (64) |
| >29.0 | 56 (33) |
| All | 86 (244) |
| p-Value for trend | <0.0001 |
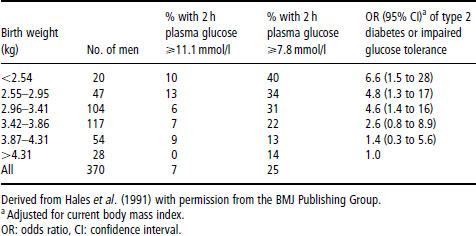
Table 18.2 Prevalence of type 2 diabetes (2 h glucose ≥ 11.1 mmol/l) and impaired glucose tolerance (2 h glucose 7.8–11.0 mmol/l) in 370 men aged 59–70 years
18.3 Potential confounding influences
The results of research discussed so far in this chapter suggest that influences linked to fetal and placental growth have an important effect on the risk of CHD and stroke. It has been argued, however, that people whose growth was impaired in utero may continue to be exposed to an adverse environment in childhood and adult life, and it is this later environment that produces the effects attributed to programming. There is strong evidence that this argument cannot be sustained. In three of the studies that have replicated the association between birth weight and CHD data on lifestyle factors, including smoking, employment, alcohol consumption and exercise, were collected. Allowance for them had little effect on the association between birth weight and CHD.
In studies exploring the mechanisms underlying these associations, the trends in CHD with birth weight were found to be paralleled by similar trends in two of its major risk factors: hypertension and type 2 (noninsulin-dependent) diabetes mellitus. Table 18.2 illustrates the size of these trends, with the prevalence of type 2 diabetes and impaired glucose tolerance falling three-fold between men who weighed 2.5 kg (5.5 lb) at birth and those who weighed 4.3 kg (9.5 lb). These associations with small size at birth were again independent of social class, cigarette smoking and alcohol consumption. Influences in adult life, however, add to the effects of the intrauterine environment. For example, the prevalence of impaired glucose tolerance is highest in people who had low birth weight, but became obese as adults.
18.4 Findings for particular cardiovascular and metabolic disorders
Hypertension
Associations between LBW and raised blood pressure in childhood and adult life have been extensively demonstrated around the world. Figure 18.2 shows the results of a systematic review of published papers describing the association between birth weight and blood pressure; this review was based on 34 studies of more than 66 000 people of all ages in many countries. Each point on the figure with its confidence interval represents a study population and the populations are ordered by their ages. The horizontal position of each population describes the change in blood pressure that was associated with a 1kg (2.2 lb) increase in birth weight. In almost all the studies an increase in birth weight was associated with a fall in blood pressure, and there was no exception to this in the studies of adults, which now total nearly 8000 men and women. The associations are less consistent in adolescence, presumably because the tracking of blood pressure from childhood through adult life is perturbed by the adolescent growth spurt. These associations were not confounded by socioeconomic conditions at the time of birth or in adult life. The difference in systolic pressure associated with a 1 kg difference in birth weight was around 3.5 mmHg. In clinical practice this would be a small difference, but these are large differences between the mean values of populations. Available data suggest that lowering the mean systolic pressure in a population by 10 mmHg would correspond to a 30% reduction in total attributable mortality.
Figure 18.2 Difference in systolic pressure (mmHg), with confidence intervals, per 1 kg increase in birth weight (adjusted for weight in children and body mass index in adults) in published studies of people of different ages.
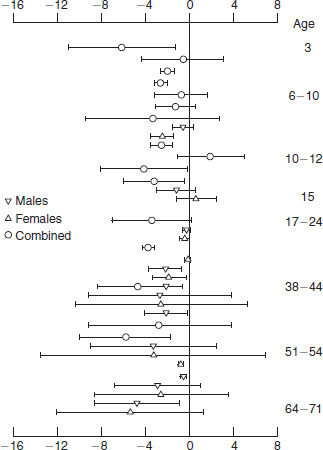
The association between LBW and raised blood pressure depends on babies who were small for dates, after reduced fetal growth, rather than on babies who were born preterm. Although in these studies alcohol consumption and higher body mass were also associated with raised blood pressure, the associations between birth weight and blood pressure were independent of them. Nevertheless, body mass remains an important influence on blood pressure and, in humans and animals, the highest pressures are found in those who were small at birth, but become overweight as adults.
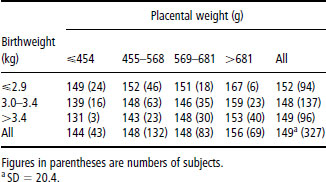
Table 18.3 Mean systolic blood pressure (mmHg) of men and women aged 50 years, born after 38 completed weeks of gestation, according to placental weight and birth weight
As has already been discussed, birth weight is a crude measure of fetal growth that does not distinguish thinness or short length, differences in head size, or variations in the balance of fetal and placental size. Analyses in Preston, UK, defined two groups of babies who developed raised blood pressures. The first group had below-average placental weight and were thin with a low ponderal index and a below-average head circumference. The second had above-average placental weight and a short crown–heel length in relation to their head circumference; such short babies tend to be fat and may have above-average birth weight. In contrast to the associations between birth size and CHD, those between birth weight and blood pressure are generally as strong as those between thinness, shortness and blood pressure. Associations between blood pressure, thinness and shortness have been found in some studies, but not in others. In a longitudinal study of young people in Adelaide, Australia, associations between blood pressure, thinness and shortness were not apparent at the age of 8 years, but emerged at the age of 20.
Placental size and blood pressure
Table 18.3 shows the systolic pressure of a group of men and women who were born, at term, in Preston, UK, in the 1950s. The subjects are grouped according to their birth weights and placental weights. Consistent with findings in other studies, systolic pressure falls between subjects with low and high birth weight. In addition, however, there is an increase in blood pressure with increasing placental weight. Subjects with a mean systolic pressure of 150 mmHg or more, a level sometimes used to define hypertension in clinical practice, comprise a group who as babies were relatively small in relation to the size of their placentas. There are similar trends with diastolic pressure. A rise in blood pressure with increasing placental weight was also found in 4-year-old children in Salisbury, UK, and among 8-year-old children in Adelaide. In studies of children and adults the association between placental enlargement and raised blood pressure has, however, been inconsistent. For example, among men and women born in Aberdeen, Scotland, after World War II, at a time when food was still rationed, raised blood pressure was associated with small placental size. As referred to later, animal studies offer a possible explanation of this inconsistency. In sheep the placenta enlarges in response to moderate undernutrition in midpregnancy. This effect, thought to be an adaptive response to extract more nutrients from the mother, is only seen in ewes that were well nourished before pregnancy.
Mother’s blood pressure
In some studies the blood pressures of the mother during and after pregnancy have been recorded. They correlate with the offspring’s blood pressure. However, the associations between body size and proportions at birth and later blood pressure are independent of the mother’s blood pressure. Recent observations show that if the mother’s blood pressure is measured throughout a 24 h period, rather than by isolated readings at antenatal clinics, there is a continuous inverse association between a woman’s blood pressure and her infant’s weight at birth. It could be argued, therefore, that the association between LBW and raised adult blood pressure reflects an association, possibly genetic, between a mother’s ambulatory blood pressure and the blood pressure of her offspring. The demonstration that experimental undernutrition during gestation programs blood pressure in animals argues against this interpretation. One possibility is that raised blood pressure during pregnancy reflects failure of maternal cardiovascular adaptations to pregnancy; this may reduce uteroplacental blood flow and fetal nutrient supply, leading to LBW and raised blood pressure in the offspring.
Mechanisms that may underlie the programming of hypertension
There are several possible mechanisms by which restricted intrauterine growth could either initiate raised blood pressure or lead to accentuated amplification of blood pressure in later life. Studies in the USA, the UK and the Netherlands have shown that blood pressure in childhood predicts the likelihood of developing hypertension in adult life. These predictions are strongest after adolescence. In children the rise in blood pressure with age is closely related to growth and is accelerated by the adolescent growth spurt. These observations led Lever and Harrap (1992) to propose that essential hypertension is a disorder of growth. The hypothesis that hypertension is a disorder of accelerated childhood growth can be reconciled with the association with LBW by postulating that postnatal catch-up growth plays an important role in amplifying changes established in utero.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree