13.1 Introduction
Iron-deficiency anemia (IDA) is the most common nutritional disorder in the world, constituting a public health condition of epidemic proportions. It particularly affects women in the reproductive age group and young children in tropical and subtropical regions. The World Bank estimates that the direct contribution of IDA to global burden of disease is 14 disability-adjusted life-years per 1000 population. It has the greatest overall effect in terms of ill-health, premature death and lost earnings.
Prevalence
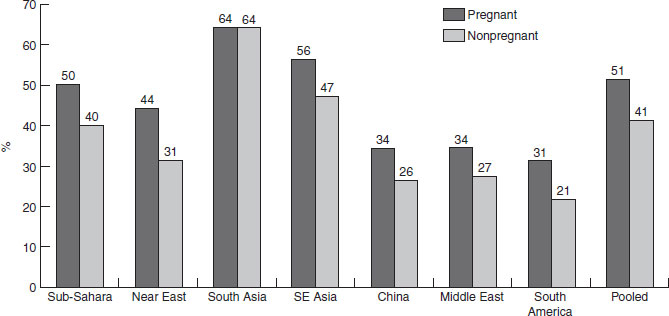
IDA affects over 2 billion people in the world. In the developing countries alone, 370 million women suffer from IDA. The average prevalence is higher in pregnant women (51%) than in the nonpregnant women (41%). The prevalence among pregnant women varies from 31% in South America to 64% in south Asia (Figure 13.1). South and south-east Asia combined contribute to 58% of the total anemic people in the developing world. In North America, Europe and Australia IDA during pregnancy is less common. Even in the USA, about 5% of young children and 5–10% of women of reproductive age suffer from IDA. About 20–30% of pregnant women in low socioeconomic strata in the USA exhibit iron deficiency during the third trimester of pregnancy. Surveys from North America and Europe indicate that, among pregnant women, the prevalence of anemia ranges between 10 and 30%. In the developing countries, the problem of iron deficiency is high. In India about 88% of pregnant women are anemic and in other parts of Asia almost 60% of women are anemic; in China, however, the prevalence does not exceed 40%.
Figure 13.1 Prevalence of anemia in women in different regions.
13.2 Definition and clinical features of iron-deficiency anemia
Anemia occurs when hemoglobin production is considerably reduced, leading to a fall in its levels in the blood. The World Health Organization (WHO) recommends cut-off values to determine IDA in different age, gender and physiological groups (Table 13.1).
Although most anemia is due to iron deficiency, the role of other causes (such as folate and vitamin B12 deficiency or anemia of chronic disease) should be distinguished.
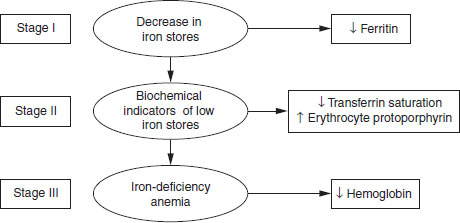
Iron depletion can be categorized into three stages of varying degrees of severity ranging from mild to severe (Figure 13.2).
- The first stage involves a decrease in iron stores, which is diagnosed by a decreased serum ferritin level. Although not associated with adverse physiological consequences, this represents increased vulnerability from long-term marginal iron balance that could lead to severe iron deficiency.
- The second stage is characterized by biochemical changes reflecting a lack of iron for normal hemoglobin production. There is decreased tranferrin saturation or increased erythrocyte protoporphyrin, and increased serum transferrin receptor levels.
- The third stage of iron deficiency is anemia. In severe IDA the levels of hemoglobin are less than 7 g/dl.
Table 13.1 Cut-off points for hemoglobin values for the diagnosis of anemia
Source: WHO (1972). Reproduced with permission from the WHO.
| Population group | Hemoglobin (g/l) |
| Adult men | <120 |
| Nonpregnant and nonlactating | |
| adult women | <120 |
| Pregnant women | <110 |
| Lactating women | <120 |
| Children <6 years | <110 |
| Children <6 years | <120 |
Iron nutritional status can be assessed by the following biochemical and hematological tests.
Serum iron concentration
In IDA, serum iron may either be low or even normal. It is regulated by reticuloendothelial release. The normal values vary between 50 and 175 μg/dl. There is a considerable diurnal variation; the levels are highest in the morning and lowest during the night. It is reduced in inflammation and malignancy and during menstruation. Therefore, this test cannot be really considered as of real diagnostic value.
Total iron binding capacity
Total iron binding capacity (TIBC) and transferrin saturation indicate iron supply to tissues. The normal value is about 300 μg/dl. TIBC is lowered in chronic disease and raised in iron deficiency.
Figure 13.2 Stages of iron depletion.
Transferrin saturation
This is a ratio (expressed as a percentage) of serum iron and TIBC. The normal value is 33%. In iron deficiency, there is a decreased saturation, while in chronic diseases the saturation is normal.
Protoporphyrin
Protoporphyrin is the precursor of heme. Free red blood cell (RBC) protoporphyrin is raised when there is an insufficient supply of iron for heme synthesis. It is also high in IDA, caused by lead toxicity and other sideroblastic anemias.
Serum ferritin
Serum ferritin reflects the status of total body iron stores. It is generally considered as the test of choice for estimating iron stores. A value below about 10 ng/ml is considered as diagnostic of iron deficiency. However, its levels are raised in inflammation, infections and liver disease. The effect of infections on serum ferritin often limits the usefulness of serum ferritin as a sensitive indicator of iron stores, particularly in areas where the incidence of infections is very high, as in the developing countries of south-east Asia. The criteria generally used to diagnose iron deficiency are listed in Table 13.2.
Transferrin receptors
Transferrin receptors become elevated on cell surfaces and in plasma whenever there is insufficient iron supply to cells or iron depletion. The ratio of transferrin to ferritin may be a good discriminator between iron deficiency and anemia of chronic inflammation.
Table 13.2 Diagnostic criteria for iron-deficiency anemia
| Indicator | Cut-off guideline |
| Serum iron (μg/dl) | <60 |
| Total iron binding capacity (μg/dl) | >300 |
| Transferrin saturation (%) | <15 |
| Erythrocyte protoporphyrin (μg/dl) | >100 |
| Serum ferritin (μg/l) | <12 |
Because of the cost implications for multiple biochemical tests, the parameter used to indicate iron status in population studies of IDA is measured by hemoglobin.
Clinical features of iron-deficiency anemia
The symptoms of IDA depend on the rate at which anemia develops in an individual. Symptoms may relate to rate of fall in hemoglobin. Since lowering of hemoglobin affects oxygen carrying capacity, in IDA, any physical exertion leads to shortness of breath. Initially, most patients complain of increasing lethargy and fatigue. More unusual symptoms are headache, tinnitus and disturbance in taste. There is often a poor correlation between hemoglobin level and symptoms. As the severity of deficiency increases, the patients develop pallor of the conjunctiva, tongue, nailbeds and soft palate. In IDA of longer duration, there may be papillary atrophy of the tongue and, the nails may become spoon shaped (koilnychia). There may also be enlargement of the spleen (splenomegaly). In children, chronic IDA may lead to behavioral changes; they may have impairment of cognitive function and short attention spans and appear withdrawn.
13.3 Iron metabolism
The human body requires iron for the synthesis of the oxygen transport proteins, hemoglobin and myoglobin in the body, and other iron-containing enzymes that participate in electron transfer and oxidation–reduction reactions. An active process in the duodenum absorbs iron. The iron thus absorbed is mobilized across the mucosal and serosal membranes into the blood where the plasma transport protein (transferrin) transports it to the cells or the bone marrow for erythropoiesis. Transferrin delivers iron to the tissues by transferrin-specific cell membrane receptors. The cell receptors bind the transferrin–iron complex at the cell surface and carry it into the cell to release iron.
In the human body, iron is distributed in six compartments (Box 13.1). Total body iron in men is about 3.8 g, while in women it is 2.3 g. In men, about one-third of the total body iron is storage iron, whereas in women it forms only about one-eighth. Approximately two-thirds of the total iron is functional, serving either a metabolic or an enzymic function. Almost all of this is in the form of hemoglobin, circulating within the RBC. Myoglobin and other iron-containing enzymes constitute about 15% of functional iron.
The factors influencing iron balance are intake of iron, iron stores and iron loss. Adult males require about 1 mg of absorbed iron daily to replace the losses in gut secretions, epithelial cells, urine and skin. In menstruating females this can increase to 1.4 mg.
Iron homeostasis, as with the most of the other metals, is maintained by controlling absorption, which increases during deficiency and decreases when erythropoisis is depressed. The body can excrete iron in a limited capacity and the excess is stored either as ferritin or as hemosiderin in the liver, spleen and bone marrow.
Inadequate iron intake will:
- enhance absorption of dietary iron
- mobilize the body’s iron stores
- reduce the transport of iron to the bone marrow
- lower the hemoglobin levels, leading finally to IDA.
- Hemoglobin (2–2.5 g of iron)
- Storage iron as ferritin and hemosiderin (1 g in men; 600 mg in women)
- Myoglobin in skeletal and cardiac muscle (130 mg of iron)
- Labile iron pool (80–90 mg iron)
- Tissue iron consisting of heme and flavoproteins (6–8 mg of iron)
- Transport iron forming (3 mg of iron)
Hemoglobin plays a crucial role in the transport of oxygen. With moderate IDA, there is a compensatory mechanism by biochemical changes to compensate for the reduced oxygen carrying capacity of blood. In contrast, in severe IDA, the markedly reduced hemoglobin content decreases the oxygen carrying capacity, leading to chronic tissue hypoxia.
Iron absorption
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree