SELECTIVE ESTROGEN RECEPTOR MODULATORS
Tamoxifen
Tamoxifen continues to be an important hormonal therapy for the prevention and treatment of breast cancer worldwide. The continued importance of tamoxifen is reflected in the fact that it is the only hormonal agent approved by the U.S. Food and Drug Administration (FDA) for the prevention of premenopausal breast cancer,1 the treatment of ductal carcinoma in situ (DCIS),2 and the treatment of surgically resected premenopausal estrogen receptor (ER)–positive breast cancer.3
The standard daily dose of tamoxifen is 20 mg, and the optimal duration depends on the underlying clinical setting. Although the recommended duration in the prevention and DCIS settings is 5 years, recently published prospective studies have demonstrated that for the adjuvant treatment of invasive breast cancer, a duration of 10 years (compared to 5 years) further reduced the risk of breast cancer mortality and improved overall survival.4
The most common toxicity from tamoxifen is hot flashes, affecting approximately 50% of treated women. These hot flashes are of varying intensity and duration. Tamoxifen-induced hot flashes appear to increase over the first 3 months of therapy and then plateau. They appear to be more prominent in women with a history of hot flashes or estrogen replacement use. Tamoxifen-induced hot flashes can be ameliorated by a number of different pharmacotherapies, including low doses of megestrol5; antidepressants such as venlafaxine,6 desvenlafaxine,7 citalopram,8 escitalopram,9 and paroxetine10; and the anticonvulsant drugs gabapentin11 and pregabalin.12 There is evidence that drugs that inhibit CYP2D6 (e.g., paroxetine) alter the metabolic activation of tamoxifen to endoxifen, a critical metabolite associated with in vivo tamoxifen efficacy.13
The estrogenic properties of tamoxifen are responsible for both beneficial and deleterious side effects. Tamoxifen increases the incidence of endometrial cancer in postmenopausal (but not premenopausal) women, with the increase in the annual incidence of endometrial cancer being approximately 2.58 (ratio of incidence rates).14 The absolute risk depends on the duration of tamoxifen administration. For women who receive 10 years of adjuvant tamoxifen, the cumulative risk is 3.1% (mortality, 0.4%) versus 1.6% (mortality, 0.2%) for 5 years of tamoxifen.4 The incidence of a rarer form of uterine cancer, uterine sarcoma, is also increased after tamoxifen use.15 This form of endometrial cancer comprises approximately 15% of all uterine malignancies that develop after tamoxifen use.15 Beneficial estrogenic effects from tamoxifen include a decrease in total cholesterol16 and the preservation of bone density in postmenopausal women.17 In premenopausal women, however, tamoxifen has a negative effect on bone density.18 Although most patients do not complain of vaginal symptoms, a few complain of vaginal dryness, whereas others have increased vaginal secretions and discharge, the latter of which is an indication of the estrogenic activity of tamoxifen on the vagina. In the Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial, a commonly observed tamoxifen side effect was vaginal bleeding, leading to a higher hysterectomy rate for patients randomized to tamoxifen (5%) compared to anastrozole (1%).19 An uncommon effect from tamoxifen is retinal toxicity. This drug can also increase the risk of cataracts. However, no difference in the rate of vision-threatening ocular toxicity has been seen among prospectively treated tamoxifen patients.20 Tamoxifen predisposes patients to thromboembolic phenomena, especially if used with concomitant chemotherapy. Depression has also been described, but the association with tamoxifen is not clear. Although liver cancers have been noted in laboratory animals, there is no established association between tamoxifen and liver cancers in humans.
Pharmacology
Tamoxifen acts by blocking estrogen stimulation of breast cancer cells, inhibiting both translocation and nuclear binding of the ER. This alters transcriptional and posttranscriptional events mediated by this receptor.21 Tamoxifen has agonistic, partial agonistic, or antagonistic effects depending on the species, tissue, or endpoints that have been assessed. Additionally, there are marked differences between the antiproliferative properties of tamoxifen and its metabolites.22
Resistance to tamoxifen can be intrinsic or acquired, and the potential mechanisms for this resistance are reviewed in the following paragraphs. At each step of the signal transduction pathway with which tamoxifen or its metabolites interferes, there is the potential for an alteration in response. The most important factor appears to be the level of ER, which is highly predictive for a response to tamoxifen. Tamoxifen is ineffective in ER-negative breast cancer. Although decreased or absent expression of the progesterone receptor (PR) is associated with a worse prognosis, the relative risk reduction in tamoxifen-treated patients is the same regardless of the presence or absence of the PR.
Following binding to the ER, subsequent translocation of the tamoxifen/ER complex to the nucleus and binding to an estrogen-response element may occur. This binding prevents transcriptional activation of estrogen-responsive genes. Laboratory and clinical data have demonstrated that ER-positive breast cancers that overexpress HER2 may be less responsive to tamoxifen and to hormonal therapy in general.23–26 In these tumors, ligand-independent activation of the ER by mitogen-activated protein kinase (MAPK) pathways may contribute to resistance.27–29 In addition, the expression of AIB1, an estrogen-receptor coactivator, has been associated with tamoxifen resistance in patients whose breast cancers overexpress HER2.30 In some cases, resistance may result from a decrease or loss of ER expression.31,32 Although mutations in the ER ligand binding domain (LBD) are rare in newly diagnosed breast cancer, ER mutations are present in up to 20% of recurrent breast cancers.33–36 These mutations lead to a conformational change in the LBD, which mimics the conformation of activated ligand-bound receptor and constitutive, ligand-independent transcriptional activity, resulting in resistance to hormonal therapy. Preclinical studies suggest that some of these mutations, although insensitive to aromatase inhibitors, retain sensitivity to higher dose selective estrogen-receptor modulators (SERM), such as endoxifen, as well as fulvestrant.35
The carcinogenic potential of tamoxifen has been recognized in rat studies37–39 and in humans (endometrial cancer).40 It has been proposed that the generation of reactive intermediates that bind covalently to macromolecules underlies the process. Such reactive intermediates have been demonstrated in vitro.40–43 In addition, the induction of covalent DNA adducts in rat livers treated with tamoxifen has been reported.44 Both constitutive and inducible cytochrome P-450 (CYP) enzymes have been implicated in the formation of metabolites with tamoxifen,45,46 and the flavone-containing monooxygenase has been implicated in the formation of the N-oxide of tamoxifen. Reactive intermediates from such metabolic steps are being evaluated for their carcinogenic potential in vitro and in vivo.
Multiple studies to evaluate tumor gene expression profiling have identified gene expression patterns or specific genes associated with resistance to tamoxifen therapy. A commonly utilized gene expression assay, Oncotype DX 21 gene assay (Genomic Health, Redwood City, California), measures the expression of genes known to be involved in estrogen signaling (e.g., ER, PR), HER2, proliferation (e.g., Ki-67), and others. In multiple different data sets, the recurrence score has been associated with a higher risk of breast cancer recurrence in patients treated with hormonal therapy (e.g., tamoxifen or aromatase inhibitors) without concomitant chemotherapy.47–49
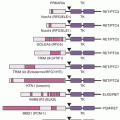
The pharmacokinetics of tamoxifen is complex. The chemical structure and metabolic pathway of tamoxifen are shown in Figure 27.1. Metabolic activation of tamoxifen is associated with greater pharmacologic activity. The two most active tamoxifen metabolites are 4-hydroxytamoxifen (4-OH tamoxifen) and 4-OH-N-desmethyltamoxifen (endoxifen). A series of studies carried out to characterize endoxifen pharmacology have demonstrated that it has equivalent potency in vitro to 4-hydroxytamoxifen in ER-α and -beta (ER-β) binding,50 for the suppression of ER-dependent human breast cancer cell line proliferation,22,50 and in global ER-responsive gene expression.51 A recent study suggests that endoxifen’s effect on the ER may differ from 4-hydroxytamoxifen based on the observation of ER-α degradation.52
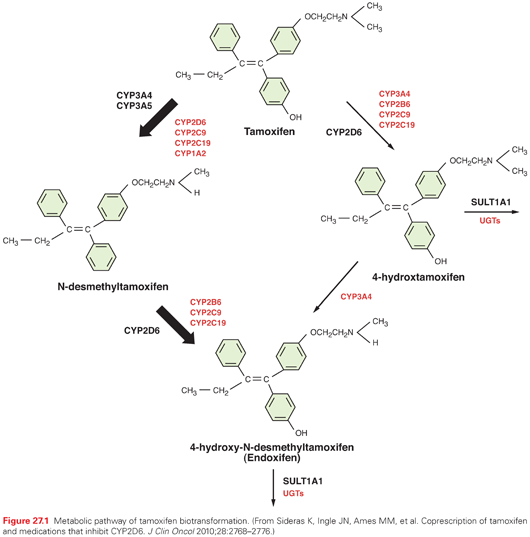
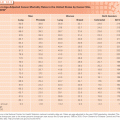
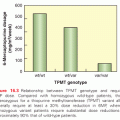
In women who receive tamoxifen at a dose of 20 mg per day, plasma endoxifen steady-state concentrations are generally 6 to 10 times higher than 4-hydroxytamoxifen.53 Although the metabolism of tamoxifen to 4-OH-tamoxifen is catalyzed by multiple enzymes, endoxifen is formed predominantly by the CYP2D6-mediated oxidation of N-desmethyltamoxifen, the most abundant tamoxifen metabolite (see Fig. 27.1).54 Multiple clinical studies have demonstrated that common CYP2D6 genetic variation (leading to low or absent CYP2D6 activity) or the drug-induced inhibition of CYP2D6 significantly lowers endoxifen concentrations.53,55 The CYP2D6 gene is highly polymorphic, with more than 70 major alleles with four well-defined phenotypes: poor metabolizers (PM), intermediate metabolizers (IM), extensive metabolizers (EM), and ultrarapid metabolizers (UM).
The clinical studies to evaluate the association between CYP2D6 polymorphisms and tamoxifen outcomes have yielded conflicting results. Initial56 and follow-up data57,58 demonstrated that CYP2D6 PM had an approximately two- to threefold higher risk of breast cancer recurrence (compared to CYP2D6 EM) and these data led an FDA special emphasis panel to recommend a tamoxifen label change to incorporate data that the CYP2D6 genotype was an important biomarker associated with tamoxifen efficacy.5 However, this label change has been delayed, in part because of conflicting data from secondary analyses of 5-year tamoxifen prospective trials (ATAC,59 BIG 1-98,60 and ABCSG861) as well as meta-analyses,62 which demonstrate that the CYP2D6 genotype is associated with tamoxifen efficacy when tamoxifen is administered as monotherapy for the adjuvant treatment of postmenopausal, ER-positive breast cancer. Additional support for the importance of endoxifen concentrations came from a secondary analysis of a prospective study, which demonstrated a higher risk of recurrence for women with low endoxifen concentrations.13
Many drugs are known to inhibit CYP2D6 activity. In tamoxifen-treated women, the coadministration of potent CYP2D6 inhibitors, such as paroxetine, converts a patient with normal CYP2D6 metabolism to a phenotypic PM.63 Many other clinically important drugs have been reported to inhibit the CYP2D6 enzyme system, but their effects on tamoxifen metabolism have not been prospectively studied. As with the data regarding CYP2D6 genotype, the data regarding CYP2D6 inhibitors has additionally been controversial, including two studies that reported opposite findings with regard to CYP2D6 inhibitor use and breast cancer recurrence or death.64,65 Although the CYP2D6 data remain controversial, we conclude that until results from prospective adjuvant studies are available, women should be counseled regarding the potential impact of the CYP2D6 genotype on the effectiveness of adjuvant tamoxifen, and potent CYP2D6 inhibitors should be avoided. Additional caution should be used with drugs that induce CYP3A, such as rifampicin, as a these drugs have been demonstrated to substantially reduce (up to 86%) the concentrations of tamoxifen and its metabolites.66
Strategies to overcome low endoxifen concentrations include dose escalation of tamoxifen to 40 mg per day, which has been demonstrated to significantly increase endoxifen concentrations,67,68 as well as the direct administration of endoxifen itself. The latter strategy is ongoing in multiple different clinical trials, and early reports suggest clinical activity in aromatase inhibitors (AI)-resistant breast cancer.69
Following the metabolic activation of tamoxifen, the hydroxylated metabolites undergo both glucuronidation and sulfation. Peak plasma levels of tamoxifen (maximum concentration [Cmax]) are seen 3 to 7 hours after oral administration. Assuming an oral bioavailability of 30%, the volume of distribution has been calculated to be 20 L/kg, and plasma clearance ranges from 1.2 to 5.1 L per hour.70 The terminal half-life of tamoxifen has been reported to range between 4 and 11 days.71,72 The elimination half-life of tamoxifen increases with successive doses, which is consistent with saturable kinetics.71,73 The drug’s distribution in tissues is extensive. Levels of the parent drug and metabolites have been reported to be higher in tissue than in plasma in animal studies.74,75 Reports of tamoxifen concentrations 10- to 60-fold higher than plasma concentrations in the liver, lungs, brain, pancreas, skin, and bones are reported.76,77 Elevated levels of tamoxifen with biliary obstruction have been reported.78
Tamoxifen has been reported to interact with warfarin,73,79–81 digitoxin, phenytoin,82 and medroxyprogesterone.73 Tamoxifen-induced activation of human transcription factor pregnane X receptor (hPXR), resulting in the induction of CYP3A4, may increase the elimination of concomitantly administered CYP3A substrates,83 such as anastrozole.84
Toremifene
Toremifene is an agent similar to tamoxifen. It is available in the United States for the treatment of patients with metastatic breast cancer, and is approved in other countries for the adjuvant treatment of ER-positive breast cancer. Clinical trials have demonstrated no difference in either disease-free or overall survival when toremifene was compared with tamoxifen for the treatment of ER-positive breast cancer,85,86and evidence exists for major cross-resistance between tamoxifen and toremifene.87,88
Pharmacology
Toremifene is an antiestrogen with a chemical structure that differs from that of tamoxifen by the substitution of a chlorine for a hydrogen atom that is retained when toremifene undergoes metabolism.89 Like tamoxifen, toremifene is metabolized by CYP3A,90 with a secondary metabolism to form hydroxylated metabolites that appear to have similar binding affinities to 4-OH tamoxifen.89,91 The importance of these metabolites or the role of metabolism to the hydroxylated metabolites is unknown, but may play a role given the structural similarity of toremifene to tamoxifen. Although the oral bioavailability has not been defined, toremifene’s oral absorption appears to be good. The time to peak plasma concentrations after oral administration ranges from 1.5 to 6.0 hours,92 with the terminal half-lives for toremifene and one metabolite, 4-hydroxytoremifene, being 5 to 6 days.93,94 The apparent clearance is 5.1 L per hour. The terminal half-life for the major metabolite, N-desmethyltoremifene, is 21 days.95 The time to reach plasma steady-state concentrations is 1 to 5 weeks. Plasma protein binding is more than 99%. As with tamoxifen, toremifene is present at higher concentrations in tissues compared to plasma with a high apparent volume of distribution (958 L). Seventy percent of the drug is excreted in feces as metabolites. Studies in patients with impaired liver function or those on anticonvulsants known to induce CYP3A have demonstrated that hepatic dysfunction decreases the clearance of toremifene and N-desmethyltoremifene,95 whereas those patients on anticonvulsants had an increased clearance. Although toremifene appeared to be less carcinogenic than tamoxifen in preclinical models,43,96,97 of the rates of endometrial cancer in the adjuvant studies have been similar to tamoxifen.85
Raloxifene
Raloxifene is an estrogen agonist and antagonist originally developed to treat osteoporosis. Large placebo-controlled randomized trials demonstrated reduced rates of osteoporosis and a reduction in new breast cancers in treated women, leading to the development of a second-generation breast cancer chemoprevention trial (National Surgical Adjuvant Breast and Bowel Project, NSAPB P2) in which raloxifene was compared with tamoxifen in high-risk postmenopausal women. In this study, tamoxifen was superior to raloxifene in terms of both invasive and noninvasive cancer events, but was associated with a higher risk of thromboembolic events and endometrial cancer.98
Pharmacology
Raloxifene is partially estrogenic in bone99 and lowers cholesterol.100 It is antiestrogenic in mammary tissue101,102 and uterine tissue.103
The pharmacokinetics of raloxifene have been studied principally in postmenopausal women.104–106 Pharmacokinetic parameters of raloxifene show considerable interindividual variation. Limited information is available on the pharmacokinetics of raloxifene in individuals with hepatic impairment, renal impairment, or both.
Raloxifene is rapidly absorbed from the gastrointestinal tract. Because raloxifene undergoes extensive first-pass glucuronidation, oral bioavailability of unchanged drug is low. Although approximately 60% of an oral dose is absorbed, the absolute bioavailability as unchanged raloxifene is only 2%. However, systemic availability of raloxifene may be greater than that indicated in bioavailability studies, because circulating glucuronide conjugates are converted back to the parent drug in various tissues.
After the oral administration of a single 120- or 150-mg dose of raloxifene hydrochloride, peak plasma concentrations of raloxifene and its glucuronide conjugates are achieved at 6 hours and 1 hour, respectively. After the oral administration of radiolabeled raloxifene, less than 1% of total circulating radiolabeled material in plasma represents the parent drug.
Results of a single-dose study in patients with liver dysfunction indicate that plasma raloxifene concentrations correlate with serum bilirubin concentrations and are 2.5 times higher than individuals with normal hepatic function. In postmenopausal women who received raloxifene in clinical trials, plasma concentrations of raloxifene and the glucuronide conjugates in those with renal impairment (i.e., estimated creatinine clearance values as low as 23 mL per minute) were similar to values in women with normal renal function.
Raloxifene and its monoglucuronide conjugates are more than 95% bound to plasma proteins. Raloxifene binds to albumin and α1-acid glycoprotein. Raloxifene undergoes extensive first-pass metabolism to the glucuronide conjugates raloxifene 4′-glucuronide, 6-glucuronide, and 6,4′-diglucuronide. UGT1A1 and -1A8 have been found to catalyze the formation of both the 6-β-and 4′-β-glucuronides, whereas UGT1A10 formed only the 4′-β-glucuronide.107 The metabolism of raloxifene does not appear to be mediated by CYP enzymes (such as CYP2D6), because metabolites other than glucuronide conjugates have not been identified.
The plasma elimination half-life of raloxifene at steady state averages 32.5 hours (range, 15.8 to 86.6 hours). Raloxifene is excreted principally in feces as an unabsorbed drug and via biliary elimination as glucuronide conjugates, which, subsequently, are metabolized by bacteria in the gastrointestinal tract to the parent drug. After oral administration, less than 0.2% of a raloxifene dose is excreted as the parent compound and less than 6% as glucuronide conjugates in urine.
Fulvestrant
Fulvestrant is an ER antagonist that has no known agonist activity and results in ER downregulation.108–111 Like tamoxifen, fulvestrant competitively binds to the ER but with a higher affinity—approximately 100 times greater than that of tamoxifen,108–114—thus preventing endogenous estrogen from exerting its effect in target cells.
Results from two phase III clinical trials using the 250 mg per month dose demonstrated fulvestrant to be as effective as anastrozole in the treatment of postmenopausal women with advanced hormone receptor–positive breast cancer previously treated with antiestrogen therapy (mainly tamoxifen).112–116 In the setting of first-line hormone-responsive metastatic breast cancer, a randomized phase III clinical trial to compare tamoxifen to fulvestrant (250 mg per month) demonstrated no differences in response or time to progression.117 Because of pharmacology data (discussed in the following paragraphs), the 500 mg per day dose was developed. A randomized trial comparing the 250 mg per month with 500 mg per month dose demonstrated a 4-month improvement in median overall survival advantage for the higher dose.118 For this reason, the higher dose is now the standard recommended dose.
Fulvestrant is well tolerated. The most common drug-related events (greater than 10% incidence) from the randomized phase III studies were injection-site reactions and hot flashes. Common events (1% to 10% incidence) included asthenia, headache, and gastrointestinal disturbances such as nausea, vomiting, and diarrhea, with minor gastrointestinal disturbances being the most commonly described adverse event.
Pharmacology
Fulvestrant is a steroidal molecule derived from E2 with an alkylsulphonyl side chain in the 7-α position (Fig. 27.2). Because fulvestrant is poorly soluble and has low and unpredictable oral bioavailability, a parenteral formulation of fulvestrant was developed in an attempt to maximize delivery of the drug.111 The intramuscular formulation provides prolonged release of the drug over several weeks. The pharmacokinetics of three different single doses of fulvestrant (50, 125, and 250 mg) have been published.111 In this phase I/II multicenter study, postmenopausal women with primary breast cancer who were awaiting curative surgery received either fulvestrant, tamoxifen, or placebo. After single intramuscular injections of fulvestrant, the time of maximal concentration (tmax) ranged from 2 to 19 days, with the median being 7 days for each dose group. At the interval of 28 days, Cmin values were two- to fivefold lower than the Cmax values. For most patients in the 125- and 250-mg dose groups, significant levels of fulvestrant were still measurable 84 days after administration. Pharmacokinetic modeling of the pooled data from the 250-mg cohort was best described by a two-compartment model in which a longer terminal phase began approximately 3 weeks after administration. Because of the long time needed to reach a steady state, the 500-mg loading dose regimen was prospectively studied and determined to be superior to the 250 mg per month dose, both in terms of steady state concentrations achieved within 1 month119as well as progression-free and overall survival.118
At menopause, the synthesis of ovarian hormones ceases. However, estrogen continues to be converted from androgens (produced by the adrenal glands) by aromatase, an enzyme of the CYP superfamily. Aromatase is the enzyme complex responsible for the final step in estrogen synthesis via the conversion of androgens, androstenedione and testosterone, to estrogens, estrone (E1) and E2. This biologic pathway served as the basis for the development of the antiaromatase class of compounds. Alterations in aromatase expression have been implicated in the pathogenesis of estrogen-dependent disease, including breast cancer, endometrial cancer, and endometriosis. The importance of this enzyme is also highlighted by the fact that selective aromatase inhibitors are commonly used as first-line therapy for the treatment of postmenopausal women with estrogen-responsive breast cancer. Aminoglutethimide was the first clinically used aromatase inhibitor. When it became available, it was used to cause a medical adrenalectomy. Because of the lack of selectivity for aromatase and the resultant suppression of aldosterone and cortisol, aminoglutethimide is no longer recommended for treating metastatic breast cancer. Aminoglutethimide is also occasionally used to try to reverse excess hormone production by adrenocortical cancers.120
Aromatase (cytochrome P-450 19 [CYP19]) is encoded by the CYP19 gene, which is highly polymorphic. Some of these variants are functionally important121 and may have clinical significance.122,123
Aromatase inhibitors have been classified in a number of different ways, including first, second, and third generation; steroidal and nonsteroidal; and reversible (ionic binding) and irreversible (suicide inhibitor, covalent binding).124 The nonsteroidal aromatase inhibitors include aminoglutethimide (first generation), rogletimide and fadrozole (second generation), and anastrozole, letrozole, and vorozole (third generation). The steroidal aromatase inhibitors include formestane (second generation) and exemestane (third generation).
Steroidal and nonsteroidal aromatase inhibitors differ in their modes of interaction with, and their inactivation of, the aromatase enzyme. Steroidal inhibitors compete with the endogenous substrates, androstenedione and testosterone, for the active site of the enzyme and are processed into intermediates that bind irreversibly to the active site, causing irreversible enzyme inhibition.19 Nonsteroidal inhibitors also compete with the endogenous substrates for access to the active site, where they then form a reversible bond to the heme iron atom so that enzyme activity can recover if the inhibitor is removed; however, inhibition is sustained whenever the inhibitor is present.19
Letrozole and Anastrozole
Both letrozole and anastrozole have been extensively studied in the metastatic and adjuvant settings. When compared to tamoxifen, both letrozole and anastrozole have demonstrated superior response rates and progression-free survival in the metastatic setting.124,125 In the adjuvant setting, two trials have been performed and demonstrated superiority in terms of relapse-free survivals of both anastrozole (ATAC)126 and letrozole (BIG 1-98).127 Additionally, anastrozole has been studied in a sequential approach, and the sequence of tamoxifen followed by anastrozole is superior to 5 years of tamoxifen alone.128 Anastrozole has recently been compared to placebo in women at an increased risk of developing breast cancer and was demonstrated to significantly reduce the incidence of invasive breast cancer.129
The side effects of both anastrozole and letrozole are similar and include arthralgias and myalgias in up to 50% of patients. Both letrozole and anastrozole are associated with a higher rate of bone fracture, compared with the tamoxifen.130 At the present time, minimal long-term (longer than 5 years) clinical data regarding the effect of aromatase inhibitors on bones are available. When offering anastrozole for extended periods of time to patients with early breast cancer, attention to bone health is paramount, and bone density should be monitored in all patients. Prospective studies have demonstrated that bisphosphonates prevent aromatase-inhibitor–induced bone loss and a meta-analysis presented at the 2013 San Antonio Breast Cancer Symposium demonstrated that bisphosphonates reduce bone recurrences and prolong overall survival. Therefore, bisphosphonates should be considered in AI-treated patients, both in those with and without an increased risk of bone fractures.
A meta-analysis of toxicities comparing aromatase inhibitors with tamoxifen has demonstrated a 30% increase in grade 3 and 4 cardiac events with aromatase inhibitors.131 However, prospective data demonstrate no differences in myocardial events comparing anastrozole with placebo, although an increase in hypertension was observed.129
No impact has been seen with anastrozole on adrenal steroidogenesis at up to 10 times the clinically recommended dose.132 Although letrozole may decrease basal and adrenocorticotropic hormone–stimulated cortisol synthesis,133,134 the clinical effect appears to be minimal. Aromatase inhibitors appear to have differential effects on lipids. In a study of over 900 patients with metastatic disease, anastrozole showed no marked effect on lipid profiles compared with baseline.135 Conversely, the administration of letrozole in women with advanced breast cancer resulted in significant increases in total cholesterol and low-density lipoprotein, from baseline, after 8 and 16 weeks of therapy.136 In the Breast International Group 1-98 trial, more women who received letrozole experienced grade 1 hypercholesterolemia compared to women who received tamoxifen.127
Letrozole is a nonsteroidal aromatase inhibitor with a high specificity for the inhibition of estrogen production (Fig. 27.3). Letrozole is 180 times more potent than aminoglutethimide as an inhibitor of aromatase in vitro. Aldosterone production in vitro is inhibited by concentrations 10,000 times higher than those required for inhibition of estrogen synthesis.137,138 In a normal male volunteer study, letrozole was shown to decrease E2 and serum E1 levels to 10% of baseline with a single 3-mg dose. In phase I studies, letrozole caused a significant decline in plasma E1 and E2 within 24 hours of a single oral dose of 0.1 mg.139,140 After 2 weeks of treatment, the blood levels of E2, E1, and estrone sulfate were suppressed 95% or more from baseline. This continued over the 12 weeks of therapy. There was no apparent alteration in plasma levels of cortisol and aldosterone with letrozole or after corticotropin stimulation.139 In postmenopausal women with advanced breast cancer, the drug did not have any effect on follicle-stimulating hormone (FSH), luteinizing hormone (LH), thyrotropin (previously thyroid-stimulating hormone), cortisol, 17-α-hydroxyprogesterone, androstenedione, or aldosterone blood concentrations.141,142
Anastrozole is a nonsteroidal aromatase inhibitor that is 200-fold more potent than aminoglutethimide.143 No effect on the adrenal glands has been detected. In human studies, the tmax is 2 to 3 hours after oral ingestion.144 Elimination is primarily via hepatic metabolism, with 85% excreted by that route and only 10% excreted unchanged in urine. The main circulating metabolite is triazole after cleavage of the two rings in anastrozole by N-dealkylation. Linear pharmacokinetics have been observed in the dose range of 1 to 20 mg and do not change with repeat dosing. The terminal half-life is approximately 50 hours, and steady-state concentrations are achieved in approximately 10 days with once-a-day dosing and are three to four times higher than peak concentrations after a single dose. Plasma protein binding is approximately 40%.145 In one study, anastrozole 1 mg and 10 mg daily, inhibited in vivo aromatization by 96.7% and 98.1%, respectively, and plasma E1 and E2 levels were suppressed 86.5% and 83.5%, respectively, regardless of dose.146 Thus, 1 mg of anastrozole achieves near maximal aromatase inhibition and plasma estrogen suppression in breast cancer patients.
A recent prospective study to evaluate the pharmacokinetics of anastrozole (1 mg per day) demonstrated large interindividual variations in plasma anastrozole and anastrozole metabolite concentrations, as well as pretreatment and postdrug plasma E1, E2, and E1 conjugate and estrogen precursor (androstenedione and testosterone) concentrations.147 Further research is needed to determine the basis for the wide variability in the pharmacokinetics of anastrozole and whether these findings are clinically relevant.
Exemestane
Exemestane has a steroidal structure and is classified as a type 1 aromatase inhibitor, also known as an aromatase inactivator, because it irreversibly binds with and permanently inactivates the enzyme.134 Exemestane has been compared to tamoxifen in both the metastatic and adjuvant settings. In the setting of tamoxifen-refractory metastatic breast cancer, exemestane is superior to megestrol acetate, as demonstrated in a phase III trial in which improvements in both median time to tumor progression and median survival were observed.148 In the adjuvant setting, the international exemestane study compared 2 to 3 years of tamoxifen with 2 to 3 years of exemestane in women who had previously competed 2 to 3 years of adjuvant tamoxifen. In this trial, a switch to exemestane resulted in superior disease-free and overall survival in the hormone receptor–positive subtype. Furthermore, exemestane has been compared with the nonsteroidal agent anastrozole in the adjuvant treatment of ER-positive breast cancer, and there were no differences in disease-free or overall survival.149 Finally, exemestane has been compared to placebo in patients at increased risk of breast cancer, and a significant reduction in the risk of developing invasive breast cancer was observed.150
Side Effects of Exemestane
Although preclinical studies have suggested that exemestane prevented bone loss in ovariectomized rats,151 the Intergroup Exemestane adjuvant trial still demonstrated a higher rate of bone fracture for patients randomized to the exemestane arm and there were no differences in fracture rates comparing anastrozole with exemestane.149 Side effects, including arthralgias and myalgias, appear to be similar to the other AIs. With regard to steroidogenesis, no impact on either cortisol or aldosterone levels was seen in a small study after the administration of exemestane for 7 days.152 Finally, exemestane has weak androgenic properties, and its use at higher doses has been associated with steroidal-like side effects, such as weight gain and acne.153,154 However, these side effects have not been observed with the FDA-approved dose (25 mg per day).155
Pharmacology
Exemestane is administered once daily by mouth, with the recommended daily dose being 25 mg. The time needed to reach maximal E2 suppression is 7 days,156 and its half-life is 27 hours.157 At daily doses of 10 to 25 mg, exemestane suppresses estrogen concentrations to 6% to 15% of pretreatment levels. This activity is more pronounced than that produced by formestane and comparable to that produced by the nonsteroidal AIs, anastrozole and letrozole.158–160 Exemestane does not appear to affect cortisol or aldosterone levels when evaluated after 7 days of treatment based on dose-ranging studies, including doses from 0.5 to 800 mg.152 Exemestane is metabolized by CYP3A4.134 Although drug–drug interactions have not been formally reported for exemestane, there is the potential for interactions with drugs that affect CYP3A4.134
GONADOTROPIN-RELEASING HORMONE ANALOGS
Gonadotropin-releasing hormone (GnRH) analogs result in a medical orchiectomy in men and are used as a means of providing androgen ablation for hormone-sensitive and castration refractory metastatic prostate cancer.161 Because the initial agonist activity of GnRH analogs can cause a tumor flare from temporarily increased androgen levels, concomitant use of the antiandrogen flutamide or bicalutamide has been used to prevent this effect. GnRH analogs can also cause tumor regressions in hormonally responsive breast cancers162 and have received FDA approval for the treatment of metastatic breast cancer in premenopausal women. Data suggest that these drugs may be useful as adjuvant therapy of premenopausal women with resected breast cancer.163 The use of these drugs in combination with tamoxifen or exemestane in premenopausal women with primary breast cancer is the subject of large, ongoing, international clinical trials. The primary toxicities of GnRH analogs are secondary to the ablation of sex steroid concentrations and include hot flashes, sweating, and nausea.164 These symptoms can be reversed with low doses of progesterone analogs.5 In males treated with GnRH analogs for prostate cancer, an alternate strategy of intermittent schedule of GnRH administration may result in improved tolerability and quality of life, with comparable efficacy compared with continuous GnRH analog administration in well-selected advanced prostate cancer patient cohorts.165 However, in a recent trial comparing intermittent with continuous androgen ablation in newly diagnosed metastatic hormone sensitive prostate cancer patients, a greater risk for death from an intermittent strategy could not be conclusively ruled out although intermittent therapy resulted in small improvements in quality of life.166
GnRH analogs available for clinical use include goserelin167,168 and leuprolide.169 Both are available in depot intramuscular preparations to be given at monthly intervals. The recommended monthly dose of leuprolide is 7.5 mg and of goserelin is 3.6 mg. There are also longer acting depot preparations to be administered every 3, 4, 6, and 12 months.
Pharmacology
Analogs of the decapeptide GnRH167,169,170
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree