Abstract
This chapter describes the causes, clinical manifestations, diagnosis, and treatment of various types of histiocytosis disorders.
Keywords
Histiocytosis, Langerhans cell histiocytosis, Rosai–Dorfman disease, emophagocytic lymphohistiocytosis, Erdheim–Chester disease, Hand–Schüller–Christian disease, eosinophilic granuloma, Abt–Letterer–Siwe disease, Langerhans cell, dendritic cell, inus histiocytosis with massive lymphadenopathy, dermal dendrocytes, macrophages, erythrophagocytosis, infection-associated hemophagocytic syndrome, malignancy-associated hemophagocytic syndrome, macrophage activation syndrome, perforin, syntaxin, Munc genes, cytolytic granules
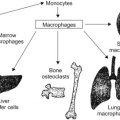
The term histiocytosis syndrome identifies a group of disorders that have in common the proliferation of cells of the mononuclear phagocyte system and/or the dendritic cell system.
Macrophages function predominantly as antigen-processing cells and dendritic cells function as accessory cells or antigen-presenting cells. Although the principal pathological cells of these syndromes are histiocytes, the term histiocytosis syndrome is a very selective term in the sense that it does not include storage diseases, hyperlipidemic xanthomatosis, or granulomatous reaction in chronic infections such as tuberculosis or foreign body granuloma.
The histiocytoses thus represent a diverse group of diseases characterized by the excess proliferation and accumulation of dendritic cells, monocytes, and macrophages in addition to other immune effector cells such as eosinophils and lymphocytes depending upon the specific type of disorder ( Table 20.1 ).
|
|
|
Langerhans Cell Histiocytosis
Incidence
The prevalence has been estimated to be between 2 and 10 cases per million children under age 15 years annually. The male to female ratio is between 1.3 and 1.9:1. The peak incidence is between the ages of 1 and 4 years. Isolated pulmonary Langerhans cell histiocytosis (LCH) occurs primarily in adults who smoke cigarettes (>90% of cases). LCH has also been shown to be commonly concordant in identical twins and less frequently in nonidentical twin siblings and relatives.
Pathology
The characteristic histopathology required for a “presumptive diagnosis” usually shows a granulomatous-like lesion with immature dendritic-appearing cells that have characteristic bean-shaped, folded nuclei and pale cytoplasm. Often, multinucleated giant cells are present. A “definitive diagnosis” of LCH requires the immunohistochemical identification of the presence of Langerhans cell antigen expression of cell surface CD1a, CD207 (langerin) or by the presence of cells with Birbeck granules by electron microscopy ( Table 20.2 ). Early in the course of the disease the lesions are usually proliferative and locally destructive. In later or healing stages, they can become more fibrotic.
|
Pathogenesis
LCH can most accurately be considered a clonal neoplasm that arises from an immature dendritic cell or in some cases an earlier hematopoietic precursor cell. Multiple reports have furthermore now documented the presence of BRAF V600E mutations in 50–60% of cases. This mutation is nearly always monoallelic and thus appears to act like a dominant driving oncogene. There also appears to be no predilection for this BRAF V600E mutation in terms of the extent of organ involvement or age ( Badalian-Very et al., 2013 ). Of particular interest, however, is that the cases which appear not to have BRAF V600E mutations do have evidence for activation of the RAS-RAF-MEK-ERK signaling pathway. This suggested that alternative activating mutations were present, which has been confirmed with the subsequent identification of mutations involving genes encoding CSF-1 receptor, RAS, and MAP2K1. Evidence for circulating CD34-positive hematopoietic progenitors harboring V600E mutations, usually in the more severe forms of LCH, along with RNA expression patterns placing the pathologic LCH LC in a category of an activated myeloid dendritic cell rather than a epidermal Langerhans cell, have contributed to LCH now being considered an oncogene-driven neoplasm of the myeloid lineage.
Clinical Features
Clinical manifestations depend on the site of lesions, number of involved areas, and extent to which the function of key involved organs is compromised. Although replaced by more prognostic categories, the classic designations, eosinophilic granuloma, Hand–Schüller–Christian disease, and Abt–Letterer–Siwe disease, are still present in the literature and are useful descriptions of the various clinical manifestations of LCH.
Eosinophilic granuloma , solitary (SEG) or multifocal (MEG), are found predominantly in older children, as well as in young adults, usually within the first three decades of life with the incidence peaking between 5 and 10 years of age. SEG and MEG represent approximately 60–80% of all instances of LCH. Patients with systemic involvement frequently have similar bone lesions in addition to other manifestations of disease.
Hand–Schüller–Christian disease (multisystem disease) was historically described as the clinical triad of lytic lesions of bone, exophthalmos, and diabetes insipidus (DI), although this form of LCH is now usually considered to be more typical multisystem LCH without key organ dysfunction. It most commonly occurs in younger children, 2–5 years of age, and represents 15–40% of such patients; this type of involvement can be observed, however, in all ages. Signs and symptoms include bone lesions with exophthalmos due to tumor mass in the orbital cavity. This usually occurs from involvement of the roof and lateral wall of the orbital bones. Orbital involvement may sometimes result in vision loss or strabismus due to optic nerve or orbital muscle involvement, respectively. The most frequent sites of skeletal involvement include the flat bones of the skull, ribs, pelvis, and scapulae. There may be extensive involvement of the skull, with irregularly shaped, lytic lesions. Less frequently, long bones and lumbosacral vertebrae, usually the anterior portion of the vertebral body, are involved. Oral involvement commonly affects the gums and/or palate, resulting in the characteristic floating tooth seen on dental radiographs. Large sections of the mandible may be involved, with loss of bone leading to diminished height of the mandibular rami. Chronic otitis media, due to involvement of the mastoid and petrous portion of the temporal bone, and otitis externa are common.
Abt–Letterer–Siwe disease occurs in about 10% of cases and represents the most severe manifestation of LCH. Typically, patients are less than 2 years of age and present with a scaly seborrheic, eczematoid, sometimes purpuric rash that involves the scalp, ear canals, abdomen, and intertriginous areas of the neck and face. The rash may be maculopapular or nodulopapular. Ulceration may result, especially in intertriginous areas. Draining ears, lymphadenopathy, hepatosplenomegaly, and, in severe cases, hepatic dysfunction with hypoproteinemia and diminished synthesis of clotting factors can occur. Gastrointestinal involvement may give rise to diarrhea, malabsorption, and bleeding. Anorexia, irritability, failure to thrive, and significant pulmonary symptoms such as cough, tachypnea, and pneumothorax may occur as well. Involvement of the hematopoietic system, with bone marrow infiltration, can result in pancytopenia.
Other presentations of LCH are commonly seen. LCH can have a strictly nodal presentation , not to be confused with sinus histiocytosis with massive lymphadenopathy (SHML) (Rosai–Dorfman disease). This presentation is characterized by significant enlargement of multiple lymph node groups, with little or no other signs of disease. Pulmonary disease, usually seen in young adults in their third or fourth decade (occasionally in adolescents) and associated with cigarette smoking, may follow a severe and often chronic, debilitating course; patients may present with pneuomthorax. Pulmonary disease may also occur as part of multisystem disease. Cutaneous disease with no evidence of dissemination has been described in infants, children, and adults.
Involvement by Site of Disease
Skeleton
Painful bone lesions affecting hematopoietically active bones are common. Radiographically, the lesions are lytic and sometimes may have sclerotic edges or bone islands within the lytic area. They occur commonly in the skull as punched-out lytic lesions. Bone involvement of the mandible and maxilla and soft-tissue involvement of the gingivae may result in loss of teeth. Involvement of vertebrae can result in vertebral collapse (vertebra plana) and lesions of long bones can result in fractures. There is often an inability to bear weight and tender, sometimes warm, swelling due to soft-tissue infiltration overlying the bone lesions occur. Radionuclide bone scan ( 99m Tc-polyphosphate) or PET/CT may show localized increased uptake at the site of involvement. The differential diagnoses include osteomyelitis, malignant bone tumors, and bony cysts. Only rarely are bones involving the wrists, hands, and feet observed. Table 20.3 lists the distribution of the sites of bone lesions.
| Site | Incidence (%) |
|---|---|
| Skull | 49 |
| Innominate bone | 23 |
| Femur | 17 |
| Orbit | 11 |
| Ribs | 8 |
| Humerus | 7 |
| Mandible | 7 |
| Tibia | 7 |
| Vertebra | 7 |
| Clavicle | 5 |
| Scapula | 3 |
| Fibula | 2 |
| Sternum | 1 |
| Radius | 1 |
| Metacarpal | 1 |
Skin
Cutaneous eruptions consist of:
- 1.
Diffuse papular scaling lesions, resembling seborrheic eczema (most common).
- 2.
Petechiae and purpura.
- 3.
Granulomatous ulcerative lesions.
- 4.
Xanthomatous lesions.
Lungs
Lung involvement may result in pulmonary dysfunction with tachypnea and/or dyspnea, cyanosis, cough, pneumothorax, or pleural effusion. Radiographic infiltrates consisting of diffuse cystic changes, nodular infiltrations, or extensive fibrosis can occur. The radiographic appearance may resemble miliary tuberculosis.
Liver
The liver may be enlarged with dysfunction that results in hypoproteinemia (total protein less than 5.5 g/dl and/or albumin less than 2.5 g/dl), edema, ascites, and/or hyperbilirubinemia (bilirubin level greater than 1.5 mg/dl, not attributable to hemolysis). Pretreatment liver biopsy more often reveals portal triditis and less often fibrohistiocytic infiltrates or bile duct proliferation. Sclerosing cholangitis, fibrosis, and liver failure may lead to the need for liver transplantation.
Hematopoietic System
The pathophysiology of hematopoietic dysfunction can be due to hypersplenism as well as direct involvement of the bone marrow by Langerhans cells and/or reactive macrophages.
Hematopoietic system dysfunction may consist of the following:
Anemia (hemoglobin level less than 10 g/dl, not due to iron deficiency or superimposed infection), leukopenia (neutrophils less than 1500/mm 3 ), or thrombocytopenia (less than 100,000/mm 3 ). Excessive number of histiocytes in the marrow aspirate is not considered evidence of dysfunction.
Lymph Nodes
Occasionally, there is massive lymph node enlargement of cervical nodes occurs without other evidence of histiocytosis.
Endocrine System
Short stature has been found in up to 40% of children with systemic LCH. Chronic illness and steroid therapy play an important role in its causation. However, short stature may also be a consequence of anterior pituitary involvement and growth hormone deficiency, which may occur in up to about half of the patients with initial anterior pituitary dysfunction. Posterior pituitary involvement with DI is characteristic of systemic LCH. Other endocrine manifestations include hyperprolactinemia and hypogonadism due to hypothalamic infiltration. Pancreatic and thyroid involvement have also been reported.
Gastrointestinal System
Gastrointestinal tract disease can occur and 2–13% of patients have biopsy-proven gastrointestinal involvement and/or digestive tract symptoms, although this may be an underestimate. Diarrhea, malabsorption, and hematochezia are common manifestations.
Central Nervous System
Four groups of patients can be clinically distinguished:
- 1.
Patients who present with hypothalamic pituitary system involvement.
- 2.
Patients who present with site-dependent symptoms of space-occupying lesions leading to headache and seizures.
- 3.
Patients who exhibit neurologic dysfunction characterized by a neurodegenerative picture of reflex abnormalities, ataxia, intellectual impairment, sometimes hydrocephalus, tremor, and dysarthria with variable progression to severe CNS deterioration.
- 4.
Patients who present with an overlap of the aforementioned symptoms.
Patients who develop CNS disease are more likely to have multisystem disease and skull lesions. Table 20.4 shows the clinical characteristics of patients with LCH who developed CNS disease compared to those who did not develop CNS disease. It reveals that patients who developed CNS disease are more likely to have multisystem disease with skull and temporal bone lesions, orbital involvement, DI, and endocrinopathies. Table 20.5 shows the classification of CNS lesions according to MRI morphology.
| Percentage in 38 CNS patients | Percentage in 275 LCH patients | |
|---|---|---|
| Multisystem disease | 72 | 40 |
| Single-system bone disease | 18 | 53 |
| Single-system skin, lymph node | 0 | 7 |
| Primary CNS disease | 10 | 0 |
| Bone | 84 | 79 |
| Skull | 74 | 40 |
| Temporal bone | 34 | 8 |
| Skin | 58 | 25 |
| Diabetes insipidus | 31 | 6 |
| Orbits | 24 | 2 |
| Endocrinopathies | 18 | 3 |
| Lungs | 16 | 6 |
| Gastrointestinal tract | 10 | 5 |
| Liver | 10 | 11 |
| Spleen | 10 | 9 |
| Type | Number | Percentage | |
|---|---|---|---|
| Ia | White-matter lesions without enhancement | 21 | 55 |
| Ib | White-matter lesions with enhancement | 9 | 24 |
| IIa | Gray-matter lesions without enhancement | 19 | 50 |
| IIb | Gray-matter lesions with enhancement | 3 | 8 |
| IIIa | Extraparenchymal dural based | 12 | 32 |
| IIIb | Extraparenchymal arachnoidal based | 2 | 5 |
| IIIc | Extraparenchymal choroid plexus based | 3 | 8 |
| IVa | Infundibular thickening | 8 | 21 |
| IVb | (Partial) empty sella | 14 | 37 |
| IVc | Hypothalamic mass lesions | 4 | 10 |
| Va | Atrophy: diffuse | 10 | 26 |
| Vb | Atrophy: localized | 6 | 16 |
| VI | Therapy-related with enhancement | 6 | 15 |
Histopathology of CNS Lesions
There are likely four stages of LCH involvement of the CNS as follows:
- 1.
Hyperplastic proliferative.
- 2.
Granulomatous.
- 3.
Xanthomatous.
- 4.
Fibrosis.
Lesions in the hyperplastic proliferative stage are most likely to contain the diagnostic LCH cells. In the cerebellum and cerebrum, white matter may show demyelination and there may also be destruction of Purkinje cells in the absence of histiocytes. Gliosis with plasma cell infiltrates may also be found.
Hypothalamic Pituitary Involvement
Signs and symptoms: Disturbances in social behavior, appetite, temperature regulation, and sleep patterns are common.
Posterior pituitary involvement: DI, polyuria, polydipsia.
Anterior pituitary involvement: Growth failure, precocious or delayed puberty, amenorrhea, hypothyroidism.
Of these, DI is the most common manifestation. The incidence of this complication ranges from 5% to 35% depending upon the extent and location of disease. Most present within 4 years of diagnosis. DI is due to infiltration by the disease into the hypothalamus with or without involvement of the posterior pituitary gland. Local tissue damage may be a consequence of IL-1 and prostaglandin E2 production. Polydipsia and polyuria may develop at presentation, during active disease (even when there is improvement in other areas), or after therapy is discontinued and there is no other apparent active disease.
Laboratory Studies for the Diagnosis of DI
- 1.
Water deprivation test with at least 3 h of no intake and with serum and urine electrolytes and osmolalities before and after deprivation plus arginine vasopressin levels is a definitive approach to diagnosis. Correction with a dose of desmopressin (DDAVP) further confirms the diagnosis. An early morning fasting urinalysis for specific gravity is also helpful as a screen.
- 2.
Gadolinium-enhanced MRI studies show thickening of the hypothalamic pituitary stalk (>2.5 mm) and absence of a posterior pituitary bright signal in T1 weighted images. These lesions are caused by pathologic infiltrates. There is no convincing evidence that established DI can be reversed by any treatment modality, but new-onset DI is considered to represent active disease and rapid initiation of treatment, usually LCH-directed chemotherapy, is recommended, although there are few data about the true response rate.
Replacement therapy with DDAVP is recommended for patients with DI. The rapid institution of effective systemic chemotherapy for disseminated disease may prevent the occurrence of DI and might be responsible for the low frequency of DI, although this has not been definitively proven. A small pituitary or empty sella indicates combined anterior and posterior pituitary insufficiency. This may be a result of disease or may be observed following cranial radiotherapy for DI.
Patients presenting with isolated idiopathic DI and morphologic changes in the suprasellar area should be closely observed. Stereotactic biopsy performed because of an enlarged pituitary stalk can distinguish a variety of conditions such as sarcoidosis, granulomatosis, tuberculosis, nonspecific lymphocytic hypophysitis, and LCH. A biopsy is not always possible. Cerebrospinal fluid (CSF) analysis for markers of germ cell tumors can be an important differentiator. Patients without a definitive diagnosis and with DI are often followed with serial contrast MRIs. If the CNS lesion enlarges and no definitive diagnosis has been made, then a biopsy may be indicated.
Space-Occupying Central Nervous System Lesions
These lesions most often arise from adjacent bone lesions, brain meninges, or choroid plexus. They usually give rise to signs and symptoms of increased intracranial pressure. They are also site-specific and size-dependent. Symptoms include headaches, vomiting, papilledema, optic atrophy, seizures, and other focal symptoms. Even diffuse meningitis-like manifestations can occur. These lesions may occur without any other evidence of LCH. Mass lesions respond well to treatment, leaving minimal or no residual defects.
Neurodegenerative Disease
The cerebellum is the second most common site of LCH CNS involvement, the first being the hypothalamic–hypophyseal axis. Neurologic symptoms occasionally predate the diagnosis of LCH. Symptoms mainly follow the pontine–cerebellar pattern, beginning as a discrete reflex abnormality or gait disturbance, and/or nystagmus. Sometimes patients may also present with hydrocephalus. They can progress to disabling ataxia. Pontine symptoms include dysarthria, dysphagia, and other cranial nerve deficits, ultimately leading to fatal neurodegeneration. On MRI, enhancing lesions involving the pons, basal ganglion, and cerebellar peduncles are observed. MRI of the cerebral hemispheres may show white-matter lesions in the periventricular area.
Biopsy shows a primarily inflammatory, lymphocyte response associated with gliosis, demyelination, and neuronal cell death. The etiology of this neurodegenerative process is unknown, but is believed to be an immune-mediated paraneoplastic response. In some cases, the onset of CNS symptoms occurs many years after the initial diagnosis of LCH.
Clinical and Laboratory Evaluation of LCH
Diagnostic Evaluation
Physical Findings: Patients should have a thorough physical examination including temperature, height, weight and head circumference, pubertal status, skin and scalp for rash, pallor or jaundice, external and middle ear, face and orbits, oropharynx, dentition, chest and lungs, abdomen for organomegaly, extremity and spine. There are few evidence-based guidelines, but the ones listed represent general best practice recommendations ( Tables 20.6 and 20.7 ).
| Follow-up test interval when organ system is: | |||
|---|---|---|---|
| Test | Involved | Not involved | Single-bone lesion |
| Hemoglobin and/or hematocrit | Monthly | 6 months | None |
| White blood cell count and differential count | Monthly | 6 months | None |
| Platelet count | Monthly | 6 months | None |
| Ferritin, iron, transferrin ESR | Monthly | 6 months | None |
| Liver function tests (SGOT, SGPT, alkaline phosphatase, bilirubin, total proteins, albumin) | Monthly | 6 months | None |
| Coagulation studies (PT, PTT, fibrinogen) | Monthly | 6 months | None |
| Chest radiograph (PA and lateral) | Monthly | 6 months | None |
| Skeletal radiograph survey a | 6 months | None | Once, at 6 months |
| Urine osmolality measurement after overnight water deprivation | 6 months | 6 months | None |
| Bone marrow aspirate and biopsy b | |||
| HLA-typing c | |||
a Radionuclide bone scan is not as sensitive as the skeletal radiograph survey in most patients. It may be performed optionally but should not replace the skeletal survey. If suspicion of lesion exists (e.g., pain) and both radiographic and radionuclide tests are negative, an MRI should be performed.
b From patients with multisystem disease.
c From patients with high-risk disease (multisystem with major organ dysfunction).
| Clinical scenario and recommended additional testing |
|---|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree