Age
Adrenals
Ovaries
Others
Prenatal/neonatal
46,XX CCAH:
Maternal androgenizing tumors
Maternal drugs
Prepubertal
46,XX NCCAH
Adrenal adenomas
Adrenal carcinomas
Adrenal hyperplasia
11β-HSD1 deficiency
Androgen producing tumors (sex cord stromal; gonadoblastoma)
Pubertal/adolescence
Adrenal adenomas
Adrenal carcinomas
Adrenal hyperplasia
Cushing’s syndrome
Androgen producing tumors (sex cord stromal; gonadoblastoma)
Hyperprolactinemia
Adult
Adrenal adenomas
Adrenal carcinomas
Adrenal hyperplasia
Cushing’s syndrome
Androgen producing tumors (sex cord stromal)
Hyperthechosis
Hyperprolactinemia
Exogenous DHEA intake
Pregnancy
Maternal adenomas/carcinomas
Lutheoma
Placental aromatase deficiency
Fetal PORD
Post-menopausal
Adrenal adenomas
Adrenal carcinomas
Androgen producing tumors (sex cord stromal)
Hyperthechosis
Pathophysiology of Hirsutism and Virilization
There are three structural types of hair on the human body: lanugo is a soft hair that covers the skin of the fetus, but disappears soon after the birth; vellus is a soft hair, usually non-pigmented and with a diameter less than 0.03 mm covering much of the body in men and women; terminal hair is longer, pigmented and coarser in texture and with different extents of expression in men and women. In particular, females have terminal hairs only in the eyebrows, eyelashes, scalp, pubis, and axillae [20]. Hair arises from a complex and highly dynamic structure—the hair follicle, which consists of several components and has a rhythmic growth cycle [21]. The hair follicle growth cycle is made up of three major phases: anagen (a stage of rapid growth), telogen (a stage of relative quiescence), and catagen (apoptosis-mediated regression) [22]. Hirsutism follows an alteration of the hair follicle cycle, in particular a prolongation of the anagen phase with a consequent transformation of vellus into terminal hairs. This alteration appears under the effect of androgens that are triggered and involved in the regulation of growth of sexual hair. Androgens active on hair follicle are testosterone (T) and dihydrotestosterone (DHT), which may be generated via a de novo synthetic pathway from cholesterol, and/or via a shortcut pathway from circulating dehydroepiandrosterone-sulfate (DHEA-S) [23]. Cutaneous testosterone may arrive from the circulation, principally synthesized in the adrenals and ovaries, and/or may be locally generated, since hair follicles are equipped with all the necessary enzymes for biosynthesis and metabolism of androgens [23]. DHT is almost entirely synthesized locally in a step catalyzed by the enzyme 5α-reductase [24]. Therefore, circulating androgen levels do not quantify the real exposure of hair follicle to androgens, since a quota is locally generated. Furthermore, cutaneous androgen effects also depend on the local expression and activity of the androgen receptor [22]. This justifies why the severity of hirsutism is always not correlated with circulating androgen concentrations.
Similarly, virilization results from the interaction between androgen concentration and local sensitivity to androgens. However, virilization also results from an excessive widespread exposure to androgens (mainly T and DHT), involving not only hair follicles but also sebaceous glands, muscle mass, adipose tissue, and external genitalia. Similarly to hirsutism, also for virilization the source of androgens is mainly represented by the adrenals (from prenatal to prepubertal periods) and ovaries (after puberty until menopause), but a quota is peripherally or locally synthesized [25, 26]. The most deleterious effect of hyperandrogenism in females is certainly when it appears in the prenatal period, because it may cause ambiguous external genitalia [27].
At the start of life, in fact, the external genitalia are identical, regardless of the genetic or gonadal sex and consist of the genital tubercle, the urogenital folds, and the genital swellings (Fig. 13.1a). In a normal female fetus, when androgenic effects are lacking, the genital tubercle forms a clitoris, and the urethral folds and the genital swellings develop into the labia minora and majora, respectively (Fig. 13.1a, left side) [28]. On the contrary, when hyperandrogenism takes place in utero, such as in a 46,XX fetus affected by CCAH, male genital differentiation occurs due to the elevated amounts of adrenal testosterone, which is produced from the seventh to eighth week of gestation. In this case the genital tubercle grows into a penis, the urethral folds fuse to create a tubular penile urethra with the tendency of the meatus to locate at the tip of the penis, and the genital swellings join to form a scrotum (Fig. 13.1a; right side). In cases of milder androgen exposure (before the fourteenth week of gestational age), the posterior labial fusion results in an increased anal–genital distance (Fig. 13.1b). Since internal genitalia are not sensitive to androgens, the uterus, fallopian tubes, and the upper part of the vagina develop normally.
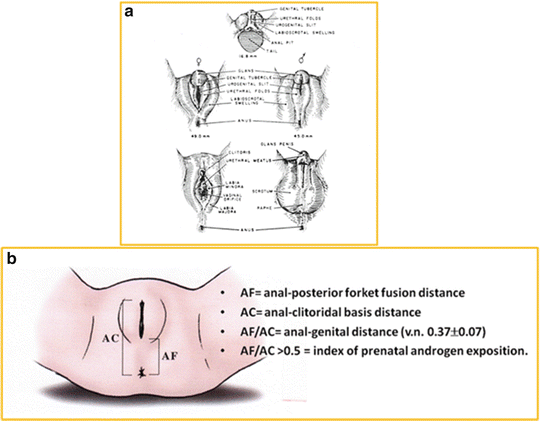

Fig. 13.1
(a) External Genitalia development in females and males; (b) Anal–genital distance
Key Points to the Diagnosis of Hirsutism and Virilization
The diagnosis of hirsutism and virilization is based on the quantification of the problem and on the definition of the etiology. The quantification of hirsutism or virilization is obtained by a physical exam through the use of subjective and objective methods. The establishment of the most probable etiology is based on clinical history (age of onset and rapidity of progression), hormone profile, and, in some cases, genetic analysis.
Physical Examination
The quantification of hirsutism in women can be obtained through objective and subjective methods. Objective methods, such as photographic evaluations, weighing of shaved or plucked hairs and microscopic measurements are reliable. However, the complexity and high cost of these methods limit their use in clinical practice [29]. Subjective methods mainly refer to visually scoring terminal hairs in specified areas. These methods have the advantage of being easy, convenient, cheap, and fast; however, they are subject to some inter-observer variation. This limitation can be drastically reduced if the visual score is applied by trained physicians at least 3 months after the use of laser or electrolysis, at least 4 weeks after depilation or waxing, and at least 5 days after shaving, and if the number of examiners is minimized [29]. Of the visual scores available, the modified Ferriman–Gallwey score (mFG) has now become the gold-standard for the evaluation of hirsutism [30]. This method applies a similar 0–4 scale to nine body areas (upper lip, chin, chest, upper and lower back, upper and lower abdomen, arm, forearm, thigh, and lower leg) that appear to be the best areas indicative of the action of androgens on the female hair follicle [29]. There are actually two open questions related to the interpretation of the mFG score. The first is the cutoff value to be used to diagnose the presence of hirsutism. The second is what interpretation to give to hirsutism predominantly localized on the face with respect to hirsutism predominantly localized on the trunk or arms, and what interpretation to give to the presence of terminal hairs selectively on the face in the absence of an mFG score indicative of hirsutism. The Androgen Excess and Polycystic Ovary Syndrome Society recently issued recommendations regarding the cutoff value of the mFG score to be applied. The Society recommends adapting the cutoff to the race and ethnicity of the population to which it is applied and, if this value is unavailable, using a cutoff value of 8 or above for White, Black and South-East Asian women, and a cutoff of 3 or above for Far-East Asian women [21]. Given the importance of the topic in both clinical practice and scientific research, how to define hirsutism requires more intensive research, possibly involving dermatologists and with the help of new technologies. In addition, other factors should be considered in defining cutoff points, including age (i.e., pediatric versus adult age) and, as reported above, how the presence of terminal hairs selectively on the face should be evaluated with respect to body hair. In fact, this often represents a major complaint in women with borderline hirsutism mFG scores, irrespective of age, social condition, and health problems.
The clinical evaluation of virilization is based on genital Prader Staging (Fig. 13.2) and anogenital distance (Fig. 13.1b) in newborns and on a complete physical examination (evaluation of the presence or absence of palpable gonads, measurement of phallus length, urethral opening identification, presence or absence of a vagina, presence or absence of clitoral hypertrophy) thereafter. When virilization is clinically suspected, the diagnosis usually needs to be confirmed by imaging such as abdominal/pelvic ultrasound, genitourethrogram, and, if necessary, MRI.
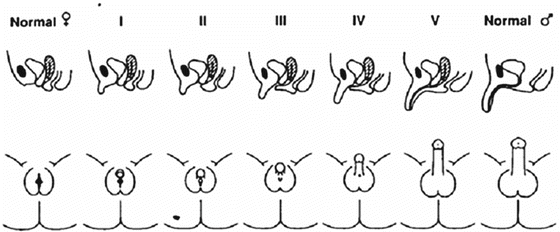

Fig. 13.2
Prader stages of genital androgenization
Hormone Profile
As previously mentioned, hirsutism and virilization are markers of excessive tissue exposure to androgen action, which results from the interaction between local androgen concentration and tissue sensitivity to androgens. Therefore, the diagnosis of hirsutism and virilization do not necessarily reflect high circulating androgen levels. This means that serum androgen measurements are often not sufficient to diagnose the presence of hirsutism or virilization, or to establish their severity, although they are extremely useful to define the etiology. However, the assay performance of the analytical methods used to measure androgens in the circulation has to be seriously taken into account for the correct interpretation of the results. The performance of modern immunoassay methods in terms of specificity and accuracy is poor, particularly for some androgens (17OH-progesterone, androstenedione, DHEA) and for low circulating concentrations, such as those enriched by testosterone in prepuberty and in the normal adult female range (<1 ng/mL) [31]. Liquid chromatography combined with tandem mass spectrometry (LC-MS/MS) is an innovative technique displaying good precision, sensitivity, and high accuracy for measuring female androgen levels throughout all stages of life, with the advantages of the high throughput and the low cost required for the analysis [32, 33]. Therefore, it represents a convenient and reliable assay when androgen measurement in females is required, provided that a reference normal range is determined in house, by using a carefully selected healthy non-hyperandrogenic control female population [21]. In addition, LC-MS/MS makes it possible to measure important steroids such as 11deoxycortisol, and deoxycorticosterone (DOC) that cannot be measured with immunoassay methods and that are triggers for the diagnosis of uncommon forms of CCAH and NCCAH. Due to the numerous advantages of this technique, LC-MS/MS has recently been introduced in laboratories in critical fields that strongly rely on the reliability and rapidity of the measurement, such as newborn screening for congenital diseases including CCAH [34].
Genetic Analysis
Genetic analyses are warranted when NCCAH or CCAH are strongly suspected as causes of hirsutism or virilization, because they are genetic disorders with autosomal recessive inheritance. The genes involved in all defects have been isolated and characterized, and specific mutations have been identified (Table 13.2). Genetic analysis for identification of mutations of the genes involved is informative for the index case (type and severity of the disorder) and for future prenatal diagnosis.
Gene/protein type | Chromosome location | Inheritance | Gonads | Müllerian structures | External genitalia | Other associated features |
|---|---|---|---|---|---|---|
HSD3B2/enzyme | 1p13.1 | AR | Ovary | Yes | Female or ambiguous | CAH, adrenal insufficiency |
CYP21A2/enzyme | 6p21.23 | AR | Ovary | Yes | Female, ambiguous or male | CAH, +/− adrenal insufficiency |
CYP11B1/enzyme | 8q21.22 | AR | Ovary | Yes | Female, ambiguous or male | CAH, +/− hypertension |
POR/CYP electron donor | 7q11.2 | AR | Ovary | Yes | Female or ambiguous | Mixed features of 21-OHDef, 17-OHDef/17-20-lyaseDef, aromatase Def; +/− Antley–Bixler’s Syndrome |
CYP19/enzyme | 15q21 | AR | Ovary | Yes | Ambiguous | Maternal virilization during pregnancy, absent breast development at puberty, except partial cases |
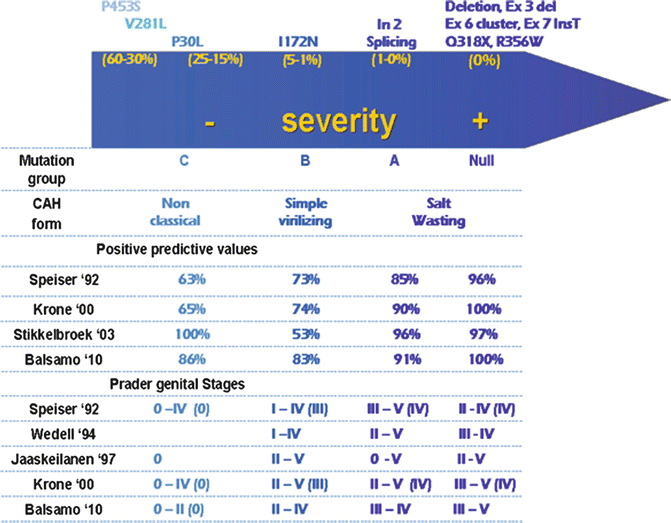
21-Hydroxylase deficiency, the most frequent cause of NCCAH and CCAH, is caused by cytochrome P450c21 enzyme impairment. The CYP21A2 gene, encoding for 21-hydroxylase, is homologous at 98 % in exons and at 96 % in introns to the non-functional CYP21A1P pseudogene. The high degree of sequence homology and tandem repeating order of RCCX module sequences is the cause of sequence misalignments during meiosis, which result in frequent unequal meiotic crossovers, as these modules are located in the HLA region of the genome (where recombination events occur at a particularly high level to ensure high immunological diversity) [35]. This mechanism generates gene deletions as well as the commonly called “large gene conversions”, both consisting in chromosomes with a chimeric gene (with a 5′ part of pseudogenic origin fused with a 3′ CYP21A2 region), but without a CYP21A2 gene [36]. The majority of disease-causing mutations are small mutations that arise from micro-conversions (transfer of small DNA sequences) from CYP21A1P to CYP21A2: P30L, IVS2-13A/C>G (I2 splice), Del 8 bp E3, I172N, Cluster E6, V281L, 1762_1763insT (L307 Frameshift), Q318X, R356W; the P453S mutation, which is not pseudogene-derived, is usually added to this list. The remaining 10 % of alleles have new/rare mutations due to random events [37]; over 90 rare pseudogene-independent mutations are actually listed in the Human Cytochrome P450 (CYP) Allele Nomenclature Committee at http://www.imm.ki.se/CYPalleles/cyp21.htm. The phenotype derives from the type/types of genetic mutations with a final influence on the total residual enzymatic activity [38] (Fig. 13.3). The CCAH forms result from deletions/conversions or point mutations associated with <10 % residual enzymatic activity (Null, A, B mutation groups of Fig. 13.3); the NCCAH forms result from point mutations with residual enzymatic activity comprised between 15 and 60 % (C mutation groups of Fig. 13.3). As for genital appearance, while the presence of “group C” mutations is highly predictive of forms without external genital virilization, a clear predicting capacity does not exist for stages of virilization within the Null, A and B mutation groups. In fact, although more severe virilization tends somehow to be present in patients with the most severe genotypes [39], Prader stages from II to V have, however, been reported in each of the previous mutation groups [40] (Fig. 13.3). When patients are compound heterozygotes, with two or more different mutations on the two alleles, the severity of the phenotype depends principally on the mutation with the less impaired residual activity [38].