Healthcare-Associated Viral Respiratory Infections
Julia S. Sammons
Lori K. Handy
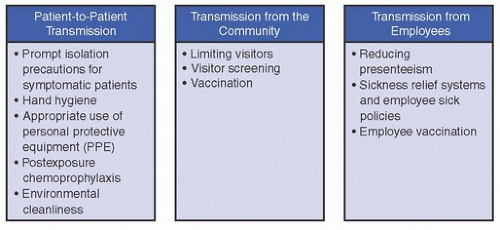
Healthcare-associated viral respiratory infections (HA-VRIs) are common and contribute to substantial morbidity, mortality, and healthcare costs. Effective strategies to prevent HA-VRI must be multifaceted in order to address the multiple avenues by which respiratory pathogens can spread in healthcare settings. Key prevention strategies address three major reservoirs/routes of transmission: (1) patient-to-patient transmission (eg, proper isolation precautions, emphasis on hand hygiene, environmental cleaning/disinfection); (2) transmission from the community (eg, visitor screening, limiting visitors); and (3) transmission from healthcare personnel (eg, reducing presenteeism, healthcare personnel [HCP] vaccination; Fig. 22-1). In this chapter, we summarize the epidemiology of HA-VRI, review risk factors and potential sources of transmission, and highlight key prevention strategies to reduce HA-VRI in children and adults.
EPIDEMIOLOGY OF HA-VRI
The epidemiology of HA-VRI includes numerous respiratory viral pathogens, namely, respiratory syncytial virus (RSV), influenza virus, adenovirus, parainfluenza virus, human metapneumovirus, and rhinovirus. Other emerging infections such as Middle East respiratory syndrome-related coronavirus (MERS-CoV) and severe acute respiratory syndrome-related coronavirus (SARS-CoV) that have been transmitted within healthcare facilities will not be addressed here but can be found in Chapter 49 Emergency Preparedness: Highly Communicable Respiratory Infections. Among pediatric patients, acute respiratory tract infection is the single most common cause for hospitalization. The majority of these infections are due to respiratory viruses, which are detected in nearly 20% of hospitalized infants and toddlers during peak respiratory viral season.1 Viruses are also frequently identified in adult patients admitted with acute respiratory tract infection.2,3,4 Among a cohort of critically ill adults hospitalized for pneumonia, 84% underwent respiratory viral testing, and, of those, approximately one-third had a respiratory virus detected.5 Thus, there is a substantial in-hospital reservoir of respiratory viruses in both pediatric and adult healthcare settings, which can lead to both sporadic healthcare transmission as well as hospital-based outbreaks of respiratory viral infections.6,7,8,9,10,11,12,13
In addition to the substantial in-hospital reservoir of respiratory viruses in pediatric healthcare facilities, there are also a number of features of pediatric patients and their care environments that make children particularly vulnerable to HA-VRI. For example, pediatric patients have less acquired immunity to viral pathogens as compared to adults; children often have increased numbers of visitors (including other children) as well as increased hours of visitation by parents and siblings, all of whom might be shedding respiratory viruses; and children often engage in developmentally appropriate behaviors that increase their likelihood of self-inoculation (eg, via hand-to-mouth behaviors) or patient-to-patient transmission of viral pathogens, through interactions in hospital playrooms and other group settings. In addition, viral shedding postrespiratory viral infection can be prolonged in children, contributing to increased exposure potential.14
Comprehensive data on the prevalence of HA-VRI in children’s hospitals are limited due to lack of national benchmarking data, but range from 0.68 to 1.91 infections per 1000 patient-days in published single-center studies.15,16 Recent data from a quaternary care children’s hospital performing house-wide surveillance for all healthcare-associated viral infections showed that HA-VRI constituted nearly two-thirds of all healthcare-associated viral infections: rhinovirus was the most commonly identified pathogen (n = 171), followed by RSV (n = 38), parainfluenza virus (n = 31), influenza virus (n = 29), adenovirus (n = 22), and human metapneumovirus (n = 13).16 The median hospital length of stay prior to infection was 22 days (IQR: 10-71 days). One-third of all infections occurred in intensive care units (ICUs); however, the average annual rate of infection was lower in ICUs (0.73) compared with oncology (1.08) and medical-surgical (0.81) units.16
Data on the prevalence of HA-VRI in adult patients are limited outside of outbreak settings; however, several studies have demonstrated a substantial contribution of respiratory viral pathogens among cases of both community and HA-respiratory infections.2,4,5,17,18 Among a cohort of adult patients with healthcare-associated pneumonia (HCAP) or community-associated pneumonia (CAP) at a tertiary hospital in Korea, of which 95% underwent respiratory viral testing, nearly 20% had a viral pathogen identified.
The prevalence of viral pathogens in HCAP was lower than in CAP (13.8% vs 24.6%, P = .004) in that study.4 However, up to one-third of cases of HCAP have a viral pathogen identified in published single-center studies.17,19,20
The prevalence of viral pathogens in HCAP was lower than in CAP (13.8% vs 24.6%, P = .004) in that study.4 However, up to one-third of cases of HCAP have a viral pathogen identified in published single-center studies.17,19,20
Transmission of Respiratory Viruses and Potential Risk Factors for HA-VRI
Respiratory viruses are transmitted from person to person by aerosols or through hand contact with the virus followed by self-inoculation. Aerosols are readily produced by coughing, sneezing, and nose-blowing.21,22 Some aerosolized particles have also been detected during normal speech. Transmission of aerosolized particles differs by particle size. Small-particle aerosols, composed of droplet nuclei ranging from 2 to 3 mm in diameter, remain suspended in the air and can be transmitted over extended distances.22 Once a virus suspended in a small-particle aerosol contaminates an airspace, only the circulation of the air and the ability of the virus to survive in the environment limit the time during which infection can be transmitted. Small-particle aerosols are not filtered by the nose and are inhaled into the lungs. Transmission by small-particle aerosol has implications for both the type of mask (surgical vs. respirator) and the type of isolation that might be required. In contrast, large-particle aerosols are composed of particles larger than 10 mm in diameter. These particles settle quickly and are transmitted only a few feet. For this reason, transmission of infection by a large-particle aerosol requires relatively close contact between infected and susceptible individuals. Large-particle aerosols are effectively filtered in the nose and do not reach the lower respiratory tract.
Prospective studies evaluating independent risk factors for HA-VRI are limited, but suggest that HA-VRI may be more common in patients with underlying chronic conditions or preterm birth.23,24,25 While HA-VRI can occur year-round, infections are typically detected in a seasonal pattern, consistent with the larger community burden of respiratory viruses, such as influenza and RSV, during peak respiratory viral season.16,26 Most data on transmission risk come from the evaluation of hospital-based outbreaks and reveal complex interactions between HCP, patients, and visitors, including lapses in infection prevention practices and inadequate personal protective equipment (PPE) use.6,7,8,10,25,27,28,29,30,31 Unfortunately, even under typical circumstances, compliance with PPE use is variable, posing substantial challenges to infection control and the potential for in-hospital transmission of viral pathogens. A systematic evaluation of HCP removal of PPE at a veterans’ hospital found that only 50% of HCP correctly remove their PPE and very few doff their PPE in the correct order or dispose of it in the proper location.32
There is a substantial body of literature supporting the role of HCP in the transmission of respiratory viruses and hospital-based outbreaks.33,34 Several studies have reported high frequency of HCP working while sick, a phenomenon termed “presenteeism”.34,35,36 A mixed-methods analysis of a cross-sectional, anonymous survey of >600 physicians and advanced practice clinicians at a large quaternary care children’s hospital found that 83% of respondents reported working while sick at least one time in the past year and nearly 10% reported working while sick at least five times.35 Reported reasons for working while ill include the perception that symptoms are mild, lack of sick leave (or combined sick leave with vacation time), not wanting to let colleagues or patients down, as well as a strong cultural norm to come to work unless remarkably ill.35,37 HCP working while sick poses substantial challenges to infection prevention and control: surveillance of a cohort of HCP for respiratory viruses via nasal swab over the 2009-2010 influenza season revealed viral shedding in nearly 30% of symptomatic HCP and that viral shedding was significantly associated with report of any symptom.36 Nasopharyngeal carriage of respiratory viruses among asymptomatic HCP has also been reported, although the frequency of detection varies between studies.26,36 Thus, HCP, primarily through presenteeism, contribute to a significant potential reservoir of respiratory viral transmission.
The healthcare environment serves as another important potential reservoir of respiratory viruses and has been implicated both in sporadic transmission events as well as hospital-based outbreaks.13,38 Environmental screening for a variety of clinically relevant viruses has revealed the presence of viral nucleic acid on numerous environmental surfaces, from high-touch surfaces such as door handles to pieces of equipment, such as mobile phones or handheld medical equipment.39,40 Symptomatic surveillance coupled with environmental sampling was performed at a skilled nursing facility and found evidence of environmental contamination in 20% of total room swabs among symptomatic residents.41 When community burden of respiratory viruses is high, leading to increased prevalence of respiratory viral infections among patients as well as visitors and HCP, it follows that the healthcare environment can play an important role in transmission.
Impact of HA-VRI on Hospitalized Patients
Perhaps most important to highlight is the impact of HA-VRI on patient outcomes. HA-VRI can be particularly harmful, leading to both increased morbidity as well as death, particularly in immunocompromised patients, such as those with hematologic malignancies or post-solid organ or hematopoietic stem cell transplant, as well as neonates and older adults.7,25,42,43 Studies comparing outcomes between those with community-associated and HA-VRI are limited and mixed, but some have shown worse outcomes, including increased rate of death, ICU admission, and need for mechanical ventilation, among patients with health-care-associated influenza or RSV compared to those admitted from the community, perhaps related to increased
severity of illness at time of infection onset.24,44 Hospitalized children who develop HA-VRI often experience worse outcomes, including prolonged hospital stays, escalation of respiratory support, and readmission.16,45,46,47 Because of this impact, the prevention of HA-VRI is even more critical, and we will devote the remainder of this chapter on key prevention strategies to reduce HA-VRI.
severity of illness at time of infection onset.24,44 Hospitalized children who develop HA-VRI often experience worse outcomes, including prolonged hospital stays, escalation of respiratory support, and readmission.16,45,46,47 Because of this impact, the prevention of HA-VRI is even more critical, and we will devote the remainder of this chapter on key prevention strategies to reduce HA-VRI.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree