The chronic myeloproliferative neoplasms (MPNs) are clonal disorders characterized by overproduction of mature myeloid cells. They share associations with molecular abnormalities such as the JAK2V617F mutation but are distinguished by important phenotypic differences. This review first considers the factors that may influence phenotype in JAK2 -mutated MPNs, especially polycythemia vera (PV) and essential thrombocythemia (ET), and then discusses the mutations implicated in JAK2 -negative MPNs such as in MPL and epigenetic regulators. Current evidence supports a model where ET and PV are disorders of relatively low genetic complexity, whereas evolution to myelofibrosis or blast-phase disease reflects accumulation of a higher mutation burden.
- •
JAK2V617F -positive polycythemia vera (PV) and essential thrombocythemia (ET) share certain clinical characteristics and may be distinguished by factors that include JAK2V617F homozygosity and disease-specific differences in JAK2 -related signaling.
- •
JAK2 exon 12–mutated PV is characterized by a specific phenotype of isolated and marked erythrocytosis, which may reflect increased signaling strength through JAK2.
- •
MPL mutations, found in ET and primary myelofibrosis (PMF), do not define distinct subsets of these diseases but show certain clinical associations that vary with the specific mutation.
- •
Mutations in TET2, other epigenetic regulators, and other regulators of cytokine signaling are not specific to the classic myeloproliferative neoplasms (MPNs) but may influence prognosis and play roles in hematopoietic stem cell (HSC) dysregulation and progression to accelerated or blast-phase disease.
Introduction
The MPNs comprise a set of clonal HSC disorders, characterized by the overproduction of 1 or more mature myeloid cell types. The 3 classic Philadelphia-negative MPNs, which are discussed in this review, are PV, ET, and PMF. Although these disorders share certain clinical features—including bone marrow hypercellularity, frequent splenomegaly, and risks of thrombosis, hemorrhage, and transformation to acute myeloid leukemia (AML)—they also show important phenotypic differences. In PV, bone marrow panmyelosis is associated with increased numbers of red blood cells and, in some patients, neutrophilia and/or thrombocytosis. In ET, there is thrombocytosis with a normal red cell mass. PMF is characterized by proliferation predominantly of bone marrow megakaryocytes and granulocytes, resulting in deposition of fibrous connective tissue, and often associated with peripheral blood leukoerythroblastosis, cytopenias, and constitutional symptoms.
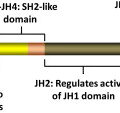
Although a relationship between these disorders was originally suggested by Dameshek in 1951, it was not until 2005 that a molecular basis for this was identified, in the form of an acquired activating mutation in JAK2 ( JAK2V617F ). With sensitive detection techniques, this mutation is detectable in more than 95% of patients with PV and in 50% to 60% of those with ET or PMF. These early studies demonstrated that expression of JAK2V617F in retroviral bone marrow transplantation models caused erythrocytosis, confirming its relevance to human MPNs. Moreover, in human studies, JAK2V617F has been identified in the stem cell–enriched CD34 + CD38 − CD90 + Lin − compartment, common myeloid progenitors, granulocyte-monocyte precursors, and megakaryocyte-erythroid precursors as well as natural killer cells, B cells, and T cells in some patients, confirming its origin in an early hematopoietic progenitor. The activating effects of the mutation are thought to reflect disruption of the normal autoinhibitory function of the JH2 domain, within which JAK2V617F is found, and have been shown to affect downstream pathways, including STATs (especially STAT5), phosphatidylinositol 3-kinase, and MAP kinase pathways in cell lines and animal models. STAT5 activation seems particularly important for JAK2V617F -induced cytokine independence in vitro and erythrocytosis in vivo and is in itself sufficient to support formation of endogenous erythroid colonies, a cardinal feature of PV.
The JAK2V617F mutation is particularly common in the classic MPNs, although it is also found in approximately half of patients with the uncommon myelodysplastic (MDS)/MPN, refractory anemia with ringed sideroblasts and marked thrombocytosis, and at lower frequencies in AML, other myeloproliferative, and myelodysplastic disorders. The reason for the myeloid bias in these diseases is unclear, given the role of JAK2 downstream of numerous cytokine receptors, but could reflect qualitative differences in the consequences of JAK2V617F in the context of different receptors. An important question however, given the high prevalence of JAK2V617F in MPNs, is how it can be associated with several diseases with distinct clinical phenotypes (PV, ET, and PMF)? Conversely, the development of MPNs in the absence of JAK2V617F must be explained and is likely to reflect the presence of other mutations in JAK2V617F -negative patients. This review discusses specific factors that may contribute to disease phenotype in MPNs. Discussed first are JAK2 -mutated MPNs, particularly focusing on the factors determining the differences between JAK2 -mutated PV and ET. Then the mutations found in JAK2V617F -negative MPNs, especially MPL mutations in ET and PMF, together with the other mutations identified in a broader range of myeloid malignancies are discussed.
JAK2 -mutated MPNs
Relationships Between Genotype and Phenotype in JAK2V617F -positive PV and ET
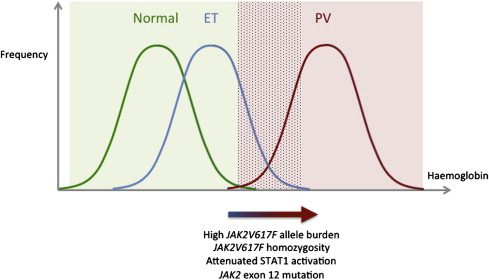
In 2005, a study of more than 800 ET patients demonstrated that in comparison to JAK2V617F -negative patients, JAK2V617F -positive patients were older; showed higher hemoglobin levels, neutrophil counts, bone marrow erythropoiesis, and granulopoiesis; showed lower platelet counts, mean corpuscular volume, serum erythropoietin, and ferritin; and showed a higher rate of PV transformation. Subsequent studies have confirmed many of these associations, supporting the view that JAK2V617F -positive ET resembles a mild form of PV and that the 2 disorders form a phenotypic continuum. Although patients with PV have hemoglobin levels above the normal range ( Fig. 1 ), patients with JAK2V617F -positive ET show a rightward shift in the normal Gaussian curve, such that some patients have hemoglobin levels in the normal range but others overlap with PV. Recent studies have identified factors that may cause this curve to shift rightwards from ET to PV. As discussed later, these include homozygosity for JAK2V617F and disease-specific differences in the signaling consequences of the JAK2V617F mutation.
Studies of JAK2V617F allele burden and clinical phenotype
A homozygous JAK2V617F sequencing pattern (>50% mutant) in granulocyte DNA was originally identified in 25% to 30% of those with PV, 9% to 20% with PMF, and 0% to 3% with ET and is a result of mitotic recombination. Several clinical studies then investigated how JAK2V617F gene dosage may influence clinical phenotype by analyzing the associations between JAK2V617F allele burden, typically measured in granulocyte DNA, and clinical parameters. Initially these studies utilised sequencing, allele-specific polymerase chain reaction (PCR) and/or restriction enzyme digestion to divide patients into “heterozygous” (<50% mutant allele) and “homozygous” (>50% mutant allele) groups. In PV, “homozygous” PV was associated with higher hemoglobin and white cell counts and lower platelet counts at diagnosis, increased pruritus, more splenomegaly, and more need for cytoreduction compared with “heterozygous” PV. Subsequent studies analyzed the associations between mutant allele burden, a continuous variable measured by real-time quantitative PCR (qPCR), and clinical features in PV and ET ( Tables 1 and 2 ). There is some discordance between studies in the associations found. This probably reflects methodologic differences between the studies, including in the use of retrospective or prospective data collection; different diagnostic and inclusion criteria; variable use of peripheral blood, granulocyte, or bone marrow DNA to determine allele burden ; and technical aspects of the qPCR assay, such as the precise probes and standards used. Higher JAK2V617F allele burdens, however, have been reproducibly associated with higher hemoglobin levels, higher white cell counts, and lower platelet counts in PV, together with other features suggestive of more extreme PV (lower mean corpuscular volume, lower serum ferritin and erythropoietin levels, more splenomegaly, and more pruritus). These data are in keeping with the concept that a higher JAK2V617F allele burden promotes a PV-like rather than ET-like phenotype.
| Association (References) | No Association (References) | |
|---|---|---|
| Higher hemoglobin/hematocrit | ||
| Higher white cell/neutrophil count | ||
| Lower platelet count | ||
| Lower mean corpuscular volume | ||
| Lower serum ferritin | ||
| Lower serum erythropoietin | ||
| More splenomegaly | ||
| Increased pruritus | ||
| Higher risk of thrombosis | ||
| Higher serum lactate dehydrogenase | ||
| Higher leukocyte alkaline phosphatase and PRV-1 expression | ||
| Higher risk of myelofibrotic transformation |
| Association (References) | No Association (References) | |
|---|---|---|
| Higher white cell/neutrophil count | ||
| Higher platelet count | ||
| More splenomegaly | ||
| Higher risk of thrombosis |
Higher JAK2V617F allele burdens in ET have been associated with some features in common with PV (higher white cell counts and more splenomegaly), but in contrast to PV, higher mutant allele burdens were associated with higher platelet counts (see Tables 1 and 2 ). This may reflect the fact that qPCR assays in these studies have not discriminated between JAK2V617F heterozygous and homozygous cells. It is possible that a higher burden of JAK2V617F -heterozygous cells is associated with more pronounced thrombocytosis but that a higher burden of homozygous-mutant cells results in a lower platelet count, for example, as a result of increased signaling strength through JAK2 impairing megakaryocytic differentiation.
Studies of JAK2V617F homozygosity in hematopoietic precursors from patients
A limitation of allele burden studies is that even in a patient with a JAK2V617F allele burden less than 50% in pooled granulocyte DNA, JAK2V617F -homozygous cells may still be present. To circumvent this issue, individual erythroid colonies from MPN patients have been genotyped to investigate the prevalence of homozygosity at the level of single precursors. Although virtually all PV and ET patients produced both wild-type and mutant colonies, colonies with homozygosity for JAK2V617F were found in almost all of those with PV but none of 17 patients with ET. This contrasts to allele burden studies in which JAK2V617F burdens greater than 50% were identified in up to 55% of patients with PV by qPCR. Subsequent studies have largely supported these observations from colony assays. JAK2V617F -homozygous erythroid colonies have been reported, however, in a few patients with ET, and granulocyte DNA JAK2V617F allele burdens over 50% have also been identified in 1% to 6% of ET patients. Small homozygous clones can be identified in approximately half of ET patients when large numbers of colonies are grown in low erythropoietin conditions. PV patients lacking JAK2V617F- homozygous colonies have also been reported. Homozygosity for JAK2V617F in hematopoietic precursors is, therefore, preferentially associated with a clinical phenotype of PV rather than ET, but the association is not absolute and it has not been clear whether this relationship is causal. The authors have recently found that in PV, a higher proportion of homozygous colonies, relative to heterozygous colonies, is associated with higher hemoglobin levels, higher white cell counts, and lower platelet counts at the time of diagnosis but not at the time of colony assay. These data support a causal role for JAK2V617F homozygosity in the development of a PV phenotype.
Little is known about the differential effects of JAK2V617F heterozygosity and homozygosity on hematopoietic cell function. The effect of JAK2V617F on expansion of hematopoietic progenitors has been assessed by measuring JAK2V617F allele burden in patient samples, either in immature and mature fractions isolated directly from bone marrow or blood or in CD34 + cells and their progeny before and after a period of liquid culture. The observation that JAK2V617F allele burden may increase from more immature to mature fractions has suggested that any selective advantage of JAK2V617F may predominantly affect more mature cell types, although some in vitro culture methods have given conflicting results. A more recent study of MPN bone marrow progenitor fractions also distinguished between heterozygous and homozygous JAK2V617F compartments and demonstrated that JAK2V617F -homozygous clones expanded from the HSC-enriched compartment to the differentiated erythroid and granulocytic compartments, at the expense of wild-type cells and in some cases JAK2V617F -heterozygous cells. The effect of JAK2V617F homozygosity on erythroid cell growth has also been studied in cultures of erythroid colonies from patient samples, in which lowering the erythropoietin concentration had the most inhibitory effect on wild-type cells, a lesser effect on JAK2V617F -heterozygous cells, and least inhibition of JAK2V617F -homozygous cells. There is, therefore, evidence that JAK2V617F exerts its selective effect preferentially in the later stages of at least some hematopoietic lineages, rather than at the HSC level, and moreover that this late advantage may be greatest for JAK2V617F -homozygous cells, especially in low erythropoietin conditions. This late expansion, together with the selective effects of relative erythropoietin deficiency in vivo, may help explain the observation that some patients with clear clinical PV seem to harbor small JAK2V617F -homozygous subclones, as assessed by erythroid colony assays. Nonetheless, it remains unclear to what extent these effects of JAK2V617F homozygosity contribute to the clinical differences between PV and ET.
Mouse models of JAK2V617F -positive MPNs
Mouse models of JAK2V617F , which are reviewed comprehensively by Li and colleagues elsewhere, have provided additional insights into the factors influencing phenotype. Where a murine JAK2V617F gene has been introduced into bone marrow cells that are transplanted into irradiated mice, the phenotype has varied at least in part with genetic background, supporting the concept that constitutional genetic differences may alter the nature of the myeloproliferative phenotype between individuals. Nonetheless, these models with high, dysregulated JAK2V617F expression levels generally showed phenotypes resembling PV, with erythrocytosis, leukocytosis, and normal or near-normal platelet counts. The effect of JAK2V617F gene dosage has been examined further in transgenic models. In one model derived from a mutated murine Jak2 gene, mice with lower transgene expression levels developed variable erythrocytosis, leukocytosis, and thrombocytosis, whereas higher levels were associated with more severe leukocytosis, thrombocytosis, anemia, and bone marrow fibrosis. In contrast, a transgenic model using human JAK2V617F showed erythrocytosis, leukocytosis, and thrombocytosis, all of which were more marked in mice with higher transgene copy numbers. In a third model, human JAK2V617F was expressed at variable levels, depending on whether a MxCre or VavCre system was used. Mice with lower JAK2V617F expression levels mice showed marked thrombocytosis, mild neutrophilia, and normal hemoglobin, reminiscent of human ET, whereas higher expression levels were associated with erythrocytosis, neutrophilia, and thrombocytosis, resembling human PV. The concept that increasing dosage may contribute to the differences between PV and ET is also supported by the observation that in one retroviral bone marrow transplantation model, secondary recipients of bone marrow with lower JAK2V617F expression levels showed a transient thrombocytosis that was not seen in recipients of marrow with higher expression levels. Overall, these studies support the concept that JAK2V617F dosage can affect phenotype, although dysregulated JAK2V617F expression makes interpretation of these effects difficult.
In view of the likely significance of appropriate levels and patterns of JAK2V617F expression in determining phenotype, several groups have developed knock-in models in which JAK2V617F is expressed at physiologic levels under the normal regulatory elements of the mouse Jak2 gene. Three knock-in models, all of which utilized a murine mutant Jak2 gene, resembled PV with erythrocytosis, leukocytosis, splenomegaly, and variable thrombocytosis. In one of these studies, mice homozygous for Jak2V617F were also generated but showed no further increase in hemoglobin levels. In contrast, a fourth model, which carried a human mutant JAK2 gene, showed thrombocytosis with minimal erythrocytosis, similar to the mild phenotypes seen in human ET, with a proportion of mice transforming to a PV-like or MF-like disease. The phenotypic variation between these models may reflect technical issues associated with the different targeting strategies or inherent differences in the mutant human and mouse proteins.
In summary, mouse models indicate that JAK2V617F gene dosage and genetic background may influence disease phenotype, but the mechanisms responsible for the variable phenotypes seen between the heterozygous knock-in models remain unclear.
Different signaling consequences of JAK2V617F in PV and ET
Although there is evidence that the gene dosage of JAK2V617F , in particular homozygosity, may influence disease phenotype, there are differences in JAK2V617F -dependent signaling between PV and ET patients, which precede the acquisition of JAK2V617F homozygosity. Although expression arrays were previously used to investigate the consequences of JAK2V617F on gene expression, these studies generally compared peripheral blood leukocytes from MPN patients with normal individuals and are, therefore, complicated by the effects of interindividual variation in gene expression and cellular heterogeneity within samples containing variable mixtures of normal and mutant cells. To circumvent these issues, a recent study analyzed differences in gene expression between clonally derived erythroid cells from patients with PV and ET. For each patient, a JAK2V617F -heterozygous cell pool was compared with a corresponding wild-type cell sample from the same patient, thus controlling for interindividual variation in gene expression. A notable difference between PV and ET was that in ET, JAK2V617F -heterozygous cells showed strong up-regulation of genes related to interferon-γ (IFN-γ) signaling compared with wild-type cells, whereas PV mutant cells showed minimal up-regulation of this pathway. Levels of phosphorylated STAT1, the major downstream mediator of IFN-γ receptor signaling, were increased in mutant erythroblasts from ET patients compared with wild-type cells, consistent with the known role of JAK2 downstream of the IFN-γ receptor. In contrast, PV patients showed no STAT1 activation in mutant cells, suggesting that JAK2V617F -induced STAT1 activation is attenuated or absent in patients with PV. Moreover, in K562 cells and CD34 + cells from MPN patients, expression of a constitutively active STAT1 mutant caused increased megakaryocytic and reduced erythroid differentiation, whereas a dominant negative form of STAT1 had the opposite effect, indicating that these signaling differences could account for aspects of the PV and ET phenotypes.
This study demonstrated that there are qualitative differences in signaling in JAK2V617F -heterozygous erythroid cells between PV and ET. The molecular mechanism for these differences remains unclear and could be due to either constitutional or acquired factors. It is also unclear whether differences in STAT1 signaling contribute to phenotype in PV and ET solely through direct effects on erythroid and megakaryocytic differentiation and whether these effects interact with JAK2V617F homozygosity.
JAK2 Exon 12 Mutations in PV
Mutations in exon 12 of JAK2 were first identified in PV patients negative for JAK2V617F in 2007 and revealed a distinct subtype of PV. Compared with PV patients with JAK2V617F , these patients were younger and had higher hemoglobin concentrations, lower white cell and platelet counts, and isolated bone marrow erythroid hyperplasia without granulocytic or megakaryocytic morphologic abnormalities. At least 20 different exon 12 mutations have been reported ( Table 3 ) and, in contrast to JAK2V617F in PV, these are usually heterozygous, with homozygosity described in only a handful of patients and in one case confirmed due to mitotic recombination. The clinical features associated with exon 12 mutations were summarized in a recent series of 106 patients: 64% presented with isolated erythrocytosis, with 15% having additional leukocytosis, 12% additional thrombocytosis, and 9% both, with no significant differences between the different exon 12 mutations. This study also confirmed the clinical associations described in the original report and demonstrated that PV patients with JAK2V617F and exon 12 mutations exhibit a similar incidence of thrombosis, myelofibrosis, acute leukemia, and death. The prevalence of these mutations in patients with erythrocytosis varies, depending on whether patients meeting criteria for PV or idiopathic erythrocytosis (absolute erythrocytosis in the absence of secondary causes and not fulfilling criteria for PV) are included. In one study of 114 patients with PV, all had either JAK2V617F (either in granulocyte DNA or erythroid colonies) or an exon 12 mutation, suggesting that almost all patients with true PV carry an activating mutation in JAK2 . In another series of 58 patients, however, with idiopathic erythrocytosis and low serum erythropoietin, 27% were found to have a JAK2 exon 12 mutation, highlighting that other mechanisms can cause isolated erythrocytosis.
| Type | Mutation | Approximate Number of Patients | References |
|---|---|---|---|
| K539L -type | F537-K539delinsL | 12 | |
| K539L | 6 | ||
| H538QK539L | 4 | ||
| H538-K539delinsL | 4 | ||
| H538-K539del | 1 | ||
| H538DK539LI540S | 1 | ||
| E543del -type | N542-E543del | 35 | |
| E543-D544del | 14 | ||
| R451-E543delinsK | 8 | ||
| I540-E543delinsMK | 4 | ||
| I540-E543delinsKK | 1 | ||
| E543del | 1 | ||
| Duplications | V536-I546dup11 | 1 | |
| V536-F547dup | 1 | ||
| F537-F547dup | 1 | ||
| F537-I546dup10+F547L | 1 | ||
| 547insL+I540-F547dup8 | 1 | ||
| F547V | 1 | ||
| Others | I540-N542delinsS | 1 | |
| D544-L545del | 1 |
a Patients in a recent large case series with non-novel mutations are not included because many had been previously reported.
Similarly to JAK2V617F , exon 12 mutations have activating effects on the kinase: in Ba/F3 cells, the mutations can confer cytokine independence, with constitutively increased levels of activated JAK2, STAT5, and ERK1/2 in the absence of erythropoietin. Use of the JAK2K539L allele in this assay resulted in more marked phosphorylation of JAK2 and ERK1/2 than the V617F allele, and the use of JAK2K539L in a retroviral murine bone marrow transplantation model caused more erythrocytosis but less marked leukocytosis and thrombocytosis than V617F , with expansion of erythroid and granulocytic but not megakaryocytic bone marrow compartments. These findings suggest that the activating effect of exon 12 mutations may be stronger than V617F and that this activation is specifically associated with a more pronounced PV phenotype.
JAK2 exon 12 mutations, therefore, promote a phenotype characterized by an isolated and marked erythrocytosis, suggestive of a further rightward shift of the hemoglobin curve, depicted in Fig. 1 . Consistent with this, exon 12 mutations have never been reported in ET. It remains unclear why the effects of JAK2 exon 12 mutations should be more selective for erythroid progenitors than JAK2V617F . The phenotype may partly reflect the stronger activating effects of these mutations, particularly because certain aspects (higher hemoglobin and lower platelet counts) have also been associated with higher mutant allele burdens in JAK2V617F -positive PV. Conversely, JAK2V617F -positive PV is frequently associated with bone marrow panmyelosis, peripheral blood neutrophilia, and sometimes thrombocytosis. It is unclear why these features are less frequent with exon 12 mutations, but this may reflect qualitative differences in the signaling consequences of the 2 types of mutation.
JAK2V617F and Accelerated-Phase Disease: Myelofibrosis and Acute Myeloid Leukemia
Although JAK2V617F is also found in 50% to 60% of those with PMF, the additional factors determining the phenotype of this clinically heterogeneous disorder are less clear. It is likely that additional acquired mutations are important: most of the other mutations identified in MPNs are more common in PMF than in PV or ET and frequently coexist with JAK2V617F (discussed later). Moreover, the spectrum of genetic lesions in PMF shows increasing overlap both with secondary AML and with poor prognostic subgroups of other myeloid malignancies. These observations, together with fact that transformations of ET and PV to secondary myelofibrosis are well recognized, support the concept that PMF represents an accelerated phase of disease. Consistent with this idea, patients diagnosed with PMF may show prior evidence of an undiagnosed chronic-phase MPN.
The role of JAK2V617F in progression of chronic-phase MPNs to AML is particularly complex. In individuals in whom JAK2V617F is detectable in the leukemic blasts, the mutation may have contributed to disease evolution through causing increased DNA damage together with an impaired apoptotic response to DNA damage. Other patients with JAK2V617F -positive chronic MPNs may, however, develop JAK2V617F -negative AML. This may reflect evolution within a shared pre-JAK2 founder clone or independent transformation of a clonally unrelated HSC. Other mutations, such as in TET2 , are likely to play a role in such transformations (discussed later).
JAK2 -mutated MPNs
Relationships Between Genotype and Phenotype in JAK2V617F -positive PV and ET
In 2005, a study of more than 800 ET patients demonstrated that in comparison to JAK2V617F -negative patients, JAK2V617F -positive patients were older; showed higher hemoglobin levels, neutrophil counts, bone marrow erythropoiesis, and granulopoiesis; showed lower platelet counts, mean corpuscular volume, serum erythropoietin, and ferritin; and showed a higher rate of PV transformation. Subsequent studies have confirmed many of these associations, supporting the view that JAK2V617F -positive ET resembles a mild form of PV and that the 2 disorders form a phenotypic continuum. Although patients with PV have hemoglobin levels above the normal range ( Fig. 1 ), patients with JAK2V617F -positive ET show a rightward shift in the normal Gaussian curve, such that some patients have hemoglobin levels in the normal range but others overlap with PV. Recent studies have identified factors that may cause this curve to shift rightwards from ET to PV. As discussed later, these include homozygosity for JAK2V617F and disease-specific differences in the signaling consequences of the JAK2V617F mutation.
Studies of JAK2V617F allele burden and clinical phenotype
A homozygous JAK2V617F sequencing pattern (>50% mutant) in granulocyte DNA was originally identified in 25% to 30% of those with PV, 9% to 20% with PMF, and 0% to 3% with ET and is a result of mitotic recombination. Several clinical studies then investigated how JAK2V617F gene dosage may influence clinical phenotype by analyzing the associations between JAK2V617F allele burden, typically measured in granulocyte DNA, and clinical parameters. Initially these studies utilised sequencing, allele-specific polymerase chain reaction (PCR) and/or restriction enzyme digestion to divide patients into “heterozygous” (<50% mutant allele) and “homozygous” (>50% mutant allele) groups. In PV, “homozygous” PV was associated with higher hemoglobin and white cell counts and lower platelet counts at diagnosis, increased pruritus, more splenomegaly, and more need for cytoreduction compared with “heterozygous” PV. Subsequent studies analyzed the associations between mutant allele burden, a continuous variable measured by real-time quantitative PCR (qPCR), and clinical features in PV and ET ( Tables 1 and 2 ). There is some discordance between studies in the associations found. This probably reflects methodologic differences between the studies, including in the use of retrospective or prospective data collection; different diagnostic and inclusion criteria; variable use of peripheral blood, granulocyte, or bone marrow DNA to determine allele burden ; and technical aspects of the qPCR assay, such as the precise probes and standards used. Higher JAK2V617F allele burdens, however, have been reproducibly associated with higher hemoglobin levels, higher white cell counts, and lower platelet counts in PV, together with other features suggestive of more extreme PV (lower mean corpuscular volume, lower serum ferritin and erythropoietin levels, more splenomegaly, and more pruritus). These data are in keeping with the concept that a higher JAK2V617F allele burden promotes a PV-like rather than ET-like phenotype.
| Association (References) | No Association (References) | |
|---|---|---|
| Higher hemoglobin/hematocrit | ||
| Higher white cell/neutrophil count | ||
| Lower platelet count | ||
| Lower mean corpuscular volume | ||
| Lower serum ferritin | ||
| Lower serum erythropoietin | ||
| More splenomegaly | ||
| Increased pruritus | ||
| Higher risk of thrombosis | ||
| Higher serum lactate dehydrogenase | ||
| Higher leukocyte alkaline phosphatase and PRV-1 expression | ||
| Higher risk of myelofibrotic transformation |
| Association (References) | No Association (References) | |
|---|---|---|
| Higher white cell/neutrophil count | ||
| Higher platelet count | ||
| More splenomegaly | ||
| Higher risk of thrombosis |
Higher JAK2V617F allele burdens in ET have been associated with some features in common with PV (higher white cell counts and more splenomegaly), but in contrast to PV, higher mutant allele burdens were associated with higher platelet counts (see Tables 1 and 2 ). This may reflect the fact that qPCR assays in these studies have not discriminated between JAK2V617F heterozygous and homozygous cells. It is possible that a higher burden of JAK2V617F -heterozygous cells is associated with more pronounced thrombocytosis but that a higher burden of homozygous-mutant cells results in a lower platelet count, for example, as a result of increased signaling strength through JAK2 impairing megakaryocytic differentiation.
Studies of JAK2V617F homozygosity in hematopoietic precursors from patients
A limitation of allele burden studies is that even in a patient with a JAK2V617F allele burden less than 50% in pooled granulocyte DNA, JAK2V617F -homozygous cells may still be present. To circumvent this issue, individual erythroid colonies from MPN patients have been genotyped to investigate the prevalence of homozygosity at the level of single precursors. Although virtually all PV and ET patients produced both wild-type and mutant colonies, colonies with homozygosity for JAK2V617F were found in almost all of those with PV but none of 17 patients with ET. This contrasts to allele burden studies in which JAK2V617F burdens greater than 50% were identified in up to 55% of patients with PV by qPCR. Subsequent studies have largely supported these observations from colony assays. JAK2V617F -homozygous erythroid colonies have been reported, however, in a few patients with ET, and granulocyte DNA JAK2V617F allele burdens over 50% have also been identified in 1% to 6% of ET patients. Small homozygous clones can be identified in approximately half of ET patients when large numbers of colonies are grown in low erythropoietin conditions. PV patients lacking JAK2V617F- homozygous colonies have also been reported. Homozygosity for JAK2V617F in hematopoietic precursors is, therefore, preferentially associated with a clinical phenotype of PV rather than ET, but the association is not absolute and it has not been clear whether this relationship is causal. The authors have recently found that in PV, a higher proportion of homozygous colonies, relative to heterozygous colonies, is associated with higher hemoglobin levels, higher white cell counts, and lower platelet counts at the time of diagnosis but not at the time of colony assay. These data support a causal role for JAK2V617F homozygosity in the development of a PV phenotype.
Little is known about the differential effects of JAK2V617F heterozygosity and homozygosity on hematopoietic cell function. The effect of JAK2V617F on expansion of hematopoietic progenitors has been assessed by measuring JAK2V617F allele burden in patient samples, either in immature and mature fractions isolated directly from bone marrow or blood or in CD34 + cells and their progeny before and after a period of liquid culture. The observation that JAK2V617F allele burden may increase from more immature to mature fractions has suggested that any selective advantage of JAK2V617F may predominantly affect more mature cell types, although some in vitro culture methods have given conflicting results. A more recent study of MPN bone marrow progenitor fractions also distinguished between heterozygous and homozygous JAK2V617F compartments and demonstrated that JAK2V617F -homozygous clones expanded from the HSC-enriched compartment to the differentiated erythroid and granulocytic compartments, at the expense of wild-type cells and in some cases JAK2V617F -heterozygous cells. The effect of JAK2V617F homozygosity on erythroid cell growth has also been studied in cultures of erythroid colonies from patient samples, in which lowering the erythropoietin concentration had the most inhibitory effect on wild-type cells, a lesser effect on JAK2V617F -heterozygous cells, and least inhibition of JAK2V617F -homozygous cells. There is, therefore, evidence that JAK2V617F exerts its selective effect preferentially in the later stages of at least some hematopoietic lineages, rather than at the HSC level, and moreover that this late advantage may be greatest for JAK2V617F -homozygous cells, especially in low erythropoietin conditions. This late expansion, together with the selective effects of relative erythropoietin deficiency in vivo, may help explain the observation that some patients with clear clinical PV seem to harbor small JAK2V617F -homozygous subclones, as assessed by erythroid colony assays. Nonetheless, it remains unclear to what extent these effects of JAK2V617F homozygosity contribute to the clinical differences between PV and ET.
Mouse models of JAK2V617F -positive MPNs
Mouse models of JAK2V617F , which are reviewed comprehensively by Li and colleagues elsewhere, have provided additional insights into the factors influencing phenotype. Where a murine JAK2V617F gene has been introduced into bone marrow cells that are transplanted into irradiated mice, the phenotype has varied at least in part with genetic background, supporting the concept that constitutional genetic differences may alter the nature of the myeloproliferative phenotype between individuals. Nonetheless, these models with high, dysregulated JAK2V617F expression levels generally showed phenotypes resembling PV, with erythrocytosis, leukocytosis, and normal or near-normal platelet counts. The effect of JAK2V617F gene dosage has been examined further in transgenic models. In one model derived from a mutated murine Jak2 gene, mice with lower transgene expression levels developed variable erythrocytosis, leukocytosis, and thrombocytosis, whereas higher levels were associated with more severe leukocytosis, thrombocytosis, anemia, and bone marrow fibrosis. In contrast, a transgenic model using human JAK2V617F showed erythrocytosis, leukocytosis, and thrombocytosis, all of which were more marked in mice with higher transgene copy numbers. In a third model, human JAK2V617F was expressed at variable levels, depending on whether a MxCre or VavCre system was used. Mice with lower JAK2V617F expression levels mice showed marked thrombocytosis, mild neutrophilia, and normal hemoglobin, reminiscent of human ET, whereas higher expression levels were associated with erythrocytosis, neutrophilia, and thrombocytosis, resembling human PV. The concept that increasing dosage may contribute to the differences between PV and ET is also supported by the observation that in one retroviral bone marrow transplantation model, secondary recipients of bone marrow with lower JAK2V617F expression levels showed a transient thrombocytosis that was not seen in recipients of marrow with higher expression levels. Overall, these studies support the concept that JAK2V617F dosage can affect phenotype, although dysregulated JAK2V617F expression makes interpretation of these effects difficult.
In view of the likely significance of appropriate levels and patterns of JAK2V617F expression in determining phenotype, several groups have developed knock-in models in which JAK2V617F is expressed at physiologic levels under the normal regulatory elements of the mouse Jak2 gene. Three knock-in models, all of which utilized a murine mutant Jak2 gene, resembled PV with erythrocytosis, leukocytosis, splenomegaly, and variable thrombocytosis. In one of these studies, mice homozygous for Jak2V617F were also generated but showed no further increase in hemoglobin levels. In contrast, a fourth model, which carried a human mutant JAK2 gene, showed thrombocytosis with minimal erythrocytosis, similar to the mild phenotypes seen in human ET, with a proportion of mice transforming to a PV-like or MF-like disease. The phenotypic variation between these models may reflect technical issues associated with the different targeting strategies or inherent differences in the mutant human and mouse proteins.
In summary, mouse models indicate that JAK2V617F gene dosage and genetic background may influence disease phenotype, but the mechanisms responsible for the variable phenotypes seen between the heterozygous knock-in models remain unclear.
Different signaling consequences of JAK2V617F in PV and ET
Although there is evidence that the gene dosage of JAK2V617F , in particular homozygosity, may influence disease phenotype, there are differences in JAK2V617F -dependent signaling between PV and ET patients, which precede the acquisition of JAK2V617F homozygosity. Although expression arrays were previously used to investigate the consequences of JAK2V617F on gene expression, these studies generally compared peripheral blood leukocytes from MPN patients with normal individuals and are, therefore, complicated by the effects of interindividual variation in gene expression and cellular heterogeneity within samples containing variable mixtures of normal and mutant cells. To circumvent these issues, a recent study analyzed differences in gene expression between clonally derived erythroid cells from patients with PV and ET. For each patient, a JAK2V617F -heterozygous cell pool was compared with a corresponding wild-type cell sample from the same patient, thus controlling for interindividual variation in gene expression. A notable difference between PV and ET was that in ET, JAK2V617F -heterozygous cells showed strong up-regulation of genes related to interferon-γ (IFN-γ) signaling compared with wild-type cells, whereas PV mutant cells showed minimal up-regulation of this pathway. Levels of phosphorylated STAT1, the major downstream mediator of IFN-γ receptor signaling, were increased in mutant erythroblasts from ET patients compared with wild-type cells, consistent with the known role of JAK2 downstream of the IFN-γ receptor. In contrast, PV patients showed no STAT1 activation in mutant cells, suggesting that JAK2V617F -induced STAT1 activation is attenuated or absent in patients with PV. Moreover, in K562 cells and CD34 + cells from MPN patients, expression of a constitutively active STAT1 mutant caused increased megakaryocytic and reduced erythroid differentiation, whereas a dominant negative form of STAT1 had the opposite effect, indicating that these signaling differences could account for aspects of the PV and ET phenotypes.
This study demonstrated that there are qualitative differences in signaling in JAK2V617F -heterozygous erythroid cells between PV and ET. The molecular mechanism for these differences remains unclear and could be due to either constitutional or acquired factors. It is also unclear whether differences in STAT1 signaling contribute to phenotype in PV and ET solely through direct effects on erythroid and megakaryocytic differentiation and whether these effects interact with JAK2V617F homozygosity.
JAK2 Exon 12 Mutations in PV
Mutations in exon 12 of JAK2 were first identified in PV patients negative for JAK2V617F in 2007 and revealed a distinct subtype of PV. Compared with PV patients with JAK2V617F , these patients were younger and had higher hemoglobin concentrations, lower white cell and platelet counts, and isolated bone marrow erythroid hyperplasia without granulocytic or megakaryocytic morphologic abnormalities. At least 20 different exon 12 mutations have been reported ( Table 3 ) and, in contrast to JAK2V617F in PV, these are usually heterozygous, with homozygosity described in only a handful of patients and in one case confirmed due to mitotic recombination. The clinical features associated with exon 12 mutations were summarized in a recent series of 106 patients: 64% presented with isolated erythrocytosis, with 15% having additional leukocytosis, 12% additional thrombocytosis, and 9% both, with no significant differences between the different exon 12 mutations. This study also confirmed the clinical associations described in the original report and demonstrated that PV patients with JAK2V617F and exon 12 mutations exhibit a similar incidence of thrombosis, myelofibrosis, acute leukemia, and death. The prevalence of these mutations in patients with erythrocytosis varies, depending on whether patients meeting criteria for PV or idiopathic erythrocytosis (absolute erythrocytosis in the absence of secondary causes and not fulfilling criteria for PV) are included. In one study of 114 patients with PV, all had either JAK2V617F (either in granulocyte DNA or erythroid colonies) or an exon 12 mutation, suggesting that almost all patients with true PV carry an activating mutation in JAK2 . In another series of 58 patients, however, with idiopathic erythrocytosis and low serum erythropoietin, 27% were found to have a JAK2 exon 12 mutation, highlighting that other mechanisms can cause isolated erythrocytosis.