In the pre-tyrosine kinase (TKI) era, allogeneic stem cell transplant (allo-SCT) was the front-line treatment of choice for young patients with chronic myelogenous leukemia (CML). Today, imatinib is well established as front-line therapy for CML, with excellent long-term outcomes. This has changed the role of allo-SCT and the number of patients undergoing allo-SCT has declined dramatically. Allo-SCT is currently recommended for patients in accelerated/blast phase disease, those who have failed a second-generation TKI and those with TKI-resistant mutations such as T315I. The role of allo-SCT in the management of CML will require continual reappraisal as medical therapies continue to evolve.
Chronic myelogenous leukemia (CML), characterized by the reciprocal translocation between chromosomes 9 and 22, t(9;22)(q34;q11), is the poster child for targeted therapies in human malignancies. Tyrosine kinase inhibitors (TKIs) such as imatinib have changed the natural history of the disease and are now well established as front-line therapy for CML patients. This fortunate change in treatment options has also significantly altered the role of allogeneic stem cell transplant (allo-SCT) for CML patients. Thus, the role of transplantation in CML can be divided into the pre-TKI era (before early 2000’s) and the current era where the use of TKIs is virtually ubiquitous in CML patients.
Role of allo-sct in cml—pre-tki era
The demonstration in the late 1970s that syngeneic (twin) donor transplant leads to the disappearance of Philadelphia chromosome established the paradigm of transplantation as curative therapy for chronic myelogenous leukemia (CML). Many studies in the early 1980s established the curative potential of allo-SCT for CML, and allo-SCT became the treatment of choice for young patients with CML and a human leukocyte antigen (HLA)-identical donor. Up to the early 1990s, chronic-phase (CP) CML was the most common indication for allo-SCT worldwide. It was also the most effective treatment for patients with advanced-stage CML, though the results in accelerated phase (AP) and blast phase (BP) CML were considerably worse than those in CP because of increased rates of recurrence and of treatment-related mortality (TRM). Practically all transplants used bone marrow (BM) as the stem cell source, and conditioning usually consisted of cyclophosphamide and total body irradiation (TBI). The initial studies used HLA-matched sibling donors (MSD); more recent ones also included unrelated donors (URD). Some of the most important studies are summarized in Table 1 . Relapse rates were low for patients transplanted in CP, but there was a considerable incidence of treatment-related complications and treatment-related deaths. Goldman and colleagues analyzed 450 patients with CP CML who received MSD allo-SCT and reported 3-year TRM, relapse rate, and overall survival (OS) ranging from 29% to 53%, 9% to 14%, and 45% to 67%, respectively, depending on the pretransplant treatment and interval from the diagnosis to the transplant. Robin and colleagues reported long-term outcomes of 102 CP patients who underwent myeloablative allo-SCT from an HLA-matched sibling using TBI/cyclophosphamide or busulfan/cyclophosphamide conditioning and cyclosporine-based graft versus host disease (GVHD) prophylaxis. The 15-year relapse rate, TRM, and OS were 8%, 46%, and 53%, respectively. Hansen and colleagues reported outcomes on 196 patients with CP CML who received URD allo-SCT between 1985 and 1994 with the use of cyclophosphamide/TBI myeloablative conditioning. At 5 years the relapse rate was 10%, nonrelapse mortality (NRM) 44%, and OS 57%. Acute grade II to IV GVHD occurred in 77% patients and chronic extensive GVHD in 67% patients.
| Authors | No. of Patients | Disease Phase | Donor Source | Conditioning Regimen | TRM | Relapse Rate | GVHD | OS |
|---|---|---|---|---|---|---|---|---|
| Thomas et al | 167 | CP1 67, CP2 12, AP 46, BP 42 | MSD | TBI/CY | 43% for CP1 | 18% for CP1 | 51% chronic GVHD among long-term survivors | At 3 y: CP 58%, AP 14% |
| Goldman et al | 450 | CP | MSD | TBI/CY or Bu/CY | 29%–53% | 9%–14% | 45%–67% at 3 y | |
| Hansen et al | 196 | CP | URD | TBI/CY | 43% | 10% at 5 y | Acute grade II–IV GVHD: 77%, extensive chronic GVHD: 67% | 57% at 5 y |
| Radich et al | 131 | CP | HLA-matched relative | Targeted Bu/CY | 14% | 8% at 3 y | Acute grade II–IV GVHD: 65%, extensive chronic GVHD: 60% | 86% at 3 y |
| Robin et al | 102 | CP | MSD | TBI/CY or Bu/CY | 46% at 15 y | 8% at 15 y | Chronic extensive GVHD 49% | 53% at 15 y |
While most of the deaths after the allo-SCT occur within the first 5 years, some patients also succumb to late sequelae. Goldman and colleagues, analyzing Center for International Blood and Marrow Transplant Research (CIBMTR) data, reported outcomes on 2444 patients who received myeloablative allo-SCT in CP1 and survived in continuous complete remission for 5 years or longer. The OS for the entire patient population was 94% at 10 years and 87% at 15 years. Compared with the age-, sex-, and race-matched general population, patients who had survived 5 years after allo-SCT still had a 2.5-times higher risk of death at 10 years, due to long-term complications of the allo-SCT. The most common causes of late deaths were organ failure (17%), infection (15%), GVHD (14%), disease relapse (7%), and secondary malignancies (7%). The mortality rates, however, for those surviving at 15-years after allo-SCT approached that of the general population. The cumulative incidence of relapse for the entire cohort was 4% at 10 years and 7% at 15 years after allo-SCT.
In the pre–tyrosine kinase inhibitor (TKI) era, there was a debate on appropriate age limits and timing of allo-SCT versus noncurative treatments with moderate efficacy, such as interferon (IFN) or IFN-cytarabine combination. The toxicity of myeloablative transplants was such that they were restricted to patients without significant comorbidities and mostly to those younger than 60 years. As the median age of diagnosis of CML is 65 years in the United States, the majority of patients were not eligible for the myeloablative transplant.
Many different approaches were tested to circumvent this limitation, including T-cell–depleted transplants, autologous transplants, and reduced-intensity conditioning (RIC). Experience with syngeneic transplants had shown that high-dose conditioning followed by the infusion of a tumor-free graft can induce durable remission. Based on this concept, several groups in the late 1980s explored the use of in vitro T-cell depletion to avoid both acute and chronic GVHD. These types of transplant were safer and were effective in preventing GVHD. Unfortunately they were also associated with high rates of graft failure, opportunistic infections, and increased rates of disease relapse, due to lack of induction of the graft versus leukemia (GvL) effect. In one such series, reporting on the outcomes of 405 patients in CP CML reported to the CIBMTR, patients with T-cell–depleted allo-SCT had 3-year probability of relapse of 48% compared with 9% with non–T-cell-depleted allo-SCT.
An alternative approach used autologous transplants rather than allo-SCT. Several studies evaluated induction of remission with chemotherapy followed by stem cell harvest in cytogenetic remission and autologous transplantation. These studies, which used unmanipulated grafts or a variety of ex vivo or in vivo purging methods to decrease the contamination by malignant clone, are now merely of historical interest, but they did show that duration of remissions correlates with the extent of residual stem cell contamination, which implies that autologous transplant with tumor-free grafts might be a curative procedure. McGlave and colleagues reported pooled data on 200 CML patients who underwent autologous transplant, with most patients receiving unmanipulated grafts. Median survival for the entire group was 42 months (CP: not reached; AP: 35.9 months; BP: 4.1 months). Most surviving patients, however, had evidence of persistent disease by cytogenetic or hematological parameters.
Kolb and colleagues were the first to show that donor lymphocyte infusion (DLI) could reliably induce remission in those relapsing with CML after allo-SCT, an observation that has been extensively reproduced. This finding, along with the observation that GvL effect plays an important role in disease control after allo-SCT and concerns over the potential toxicities of conventional myeloablation, led many groups in the last decade to use lower doses of the conditioning regimens (so-called RIC or nonmyeloablative conditioning) with the aim of decreasing NRM and using the GvL effect to maintain the disease control ( Table 2 ). Most of these studies were initiated in the pre-TKI era. After the introduction of imatinib, many of these studies had difficulty accruing patients, and few observations have been confirmed in large clinical trials.
| Authors | No. of Patients | Median Age | Time Period | Disease Phase | Donor Type | Donor Source | Median Follow-Up (mo) | Conditioning Regimen | Use of Prior Imatinib | TRM | Relapse Rate | GVHD | OS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Or et al | 24 | 35 | 1996–2001 | CP1 | MSD (n = 18), father (n = 1), URD (n = 5) | PB | 37 | Flu/Bu (n = 19), Flu/Bu/ATG (n = 5) | No | 0% at 100 d, 4% at 1 y | 0% | Acute (grade II–IV) 71%, Chronic 54% | 85% |
| Das et al | 17 | 34 | 1998–2000 | CP1 (n = 16), AP (n = 1) | MSD | BM (n = 3), PB (n = 14) | 30 | Flu/Bu/ATG (n = 11), Flu/Bu/TBI (n = 6) | No | 12% at 100 d | — | Acute (grade II–IV) 41%, Chronic 65% | 47% (1 y) |
| Bornhauser et al | 44 | 52 | NR | CP (n = 26), AP (n = 11), BP (n = 7) | MSD (n = 19), URD (n = 15) | BM (n = 15), PB (n = 29) | — | Flu/Bu (ATG in 34 patients) | No | NR | NR | Acute (grade II–IV) 43%, Chronic not reported | 14.8 mo |
| Weisser et al | 35 | 51 (included patients only ≥45 y) | — | CP1 (n = 26), CP2/AP (n = 9) | MSD (n = 19), URD (n = 16) | BM (n = 31), PB (n = 4) | 30 | TBI 8 Gy/Flu/CY/ATG | No | 11% at 100 d, 28.5% at 1 y | 8% CP1, 33% for CP2/AP | Acute (grade II–IV) 48%, Chronic extensive 23% | 69% at 2 y |
| Crawley et al | 186 | 50 | 1994–2002 | CP1 (63%), CP2 (14%), AP/BP (23%) | MSD (60%), URD (25%) | BM (28.5%), PB (71.5%) | 35 | Variety of regimens; Flu/Bu/ATG in 40% | 8% | 3.8% (100 d); 13.3% (1 y); 18.9% (2 y) | 47% at 3 y | Acute (grade II–IV) 30%, Chronic 42% | 54% at 3 y |
| Kerbauy et al | 24 | 57.5 | 1998–2003 | CP (n = 14), CP2 (n = 4), AP (n = 6) | Matched related donors | PB | 36 | TBI 2 Gy alone (n = 8); TBI/Flu (n = 16) | Yes (n = 7) | 4% at 100 d | For CP1: 22% at 2 y | Acute (grade II–IV) 46%, Chronic 57% | For CP1: 70% at 2 y |
| Kebriaei et al | 64 | 52 | 1996–2005 | CP1 20%, CP2 27%, AP/BP 53% | Matched related 47%, URD 47% | BM 59%, PB 41% | 84 | All regimens Flu-based; Flu/Mel 47% | 14% | TRM at 100 d 33%, at 1 y 38% | 34% | Acute (grade II–IV) 31%, Chronic 31% | For CP, OS at 2 and 5 y was 66% and 48% |
| Luo et al | 28 | 26 | 2005–2007 | CP1 | HLA-identical donors | BM (n = 7); PB (n = 21) | 23 | Flu/Bu/ATG; prophylactic imatinib from day 100 until 1 y | Yes, all patients | 3.6% at 100 d, 14.3% at 1 y | 32% | Acute (grade II–IV) 8% (no grade II–IV), Chronic 48% | 81% at 3 y |
| Olavarria et al | 22 | 49 | NR | CP1 | HLA-identical sibling | PB stem cells | 36 | Flu/Bu/alemtuzumab Patients received posttransplant imatinib | Yes (50% patients) | 0% at 100 d, 4% at 1 y | 68% (all after posttransplant imatinib discontinuation | Acute (grade II–IV) 5%, Chronic none | 87% at 3 y |
| Champlin et al | 33 | 41 | — | CP1 (n = 16), CP2/AP (n = 17) | — | — | 29 | Flu/Bu/ATG Patients with residual disease received planned imatinib and DLI | Yes, all patients | 0% at 100 d | 41% for AP/BP, none in CP | Acute (grade II–IV) 21% | 76% |
| Poire et al | 9 | 61 | 2002–2007 | CP1 (n = 3), CP2 (n = 2), AP (n = 1), BP (n = 3) | MSD (n = 7), URD 8/8 match (n = 1), URD 7/8 match (n = 1) | PB | — | Flu/Bu/alemtuzumab (n = 4), Flu/Mel/alemtuzumab (n = 4), Bu/CY/alemtuzumab (n = 1) | Yes, all patients (4 patients prior dasatinib also) | 0% at 100 d, 33% at 1 y | — | Grade II–IV acute GVHD (45%) (none grade III–IV) | — |
Stem cell transplant in the era of tyrosine kinase inhibitors
The Changing Paradigm
Given its superior efficacy, ease of administration, and the lack of significant side effects, imatinib was rapidly adopted as the first-line medical therapy in CP CML patients. However, clinicians across the globe also rapidly adopted the oral TKI therapy as a preferred alternative to transplant. Imatinib was approved by the United States Food and Drug Administration (FDA) in October 2001. The decline in the number of allo-SCT for CML started before FDA approval, and certainly before the availability of long-term follow-up data. Gratwohl and colleagues reported on behalf of the European Group for Blood and Marrow Transplantation (EBMT) outcomes of all CML patients reported to the EBMT between 1980 and 2004. The number of allo-SCT in Europe peaked in 1999 with 1396 transplants, with a subsequent decrease to 791 allo-SCT in 2003 and 434 allo-SCT in 2007, a 69% decrease. These investigators also reported a significant increase in the proportion of URD transplants (7% in 1980–1990 to 36% in 2000–2003), increasing use of peripheral blood (PB) as a source of stem cells (21% in 1990-2000 vs 53% in 2000-2003) and the use of RIC regimens. Bacher and colleagues reported 72% decrease in number of CML transplants from 1998 to 2004 in Germany, with a larger percentage decrease in patients undergoing allo-SCT in CP1. A similar pattern was reported by CIBMTR whereby the reported number of allo-SCT decreased from 617 in 1998 to 223 in 2003. The majority of the decrease in the transplant numbers is for CP1 patients, for whom TKIs have become the front-line standard treatment. The number of patients transplanted in AP/BP has remained relatively stable.
Economic factors may also have an impact on the choice between allo-SCT and TKI therapy, especially in the developing countries. Ruiz-Argüelles and colleagues reported data on 24 patients transplanted in CP1 using MSD and RIC in 4 Latin American countries; they mentioned the cost of allo-SCT in Mexico would be equal to approximately 200 days of imatinib 400 mg daily, thus making the case for allo-SCT. Similar observations were made by Gratwohl and colleagues when they analyzed the EBMT survey data; they noticed that the rates of allo-SCT fell after the introduction of imatinib in high-income countries in Europe but remained relatively stable for the lower-income countries, indicating persistent reliance on the “one-time” cost of allo-SCT as compared with expensive potentially “life-long” TKI therapy.
Stem cell transplant in the era of tyrosine kinase inhibitors
The Changing Paradigm
Given its superior efficacy, ease of administration, and the lack of significant side effects, imatinib was rapidly adopted as the first-line medical therapy in CP CML patients. However, clinicians across the globe also rapidly adopted the oral TKI therapy as a preferred alternative to transplant. Imatinib was approved by the United States Food and Drug Administration (FDA) in October 2001. The decline in the number of allo-SCT for CML started before FDA approval, and certainly before the availability of long-term follow-up data. Gratwohl and colleagues reported on behalf of the European Group for Blood and Marrow Transplantation (EBMT) outcomes of all CML patients reported to the EBMT between 1980 and 2004. The number of allo-SCT in Europe peaked in 1999 with 1396 transplants, with a subsequent decrease to 791 allo-SCT in 2003 and 434 allo-SCT in 2007, a 69% decrease. These investigators also reported a significant increase in the proportion of URD transplants (7% in 1980–1990 to 36% in 2000–2003), increasing use of peripheral blood (PB) as a source of stem cells (21% in 1990-2000 vs 53% in 2000-2003) and the use of RIC regimens. Bacher and colleagues reported 72% decrease in number of CML transplants from 1998 to 2004 in Germany, with a larger percentage decrease in patients undergoing allo-SCT in CP1. A similar pattern was reported by CIBMTR whereby the reported number of allo-SCT decreased from 617 in 1998 to 223 in 2003. The majority of the decrease in the transplant numbers is for CP1 patients, for whom TKIs have become the front-line standard treatment. The number of patients transplanted in AP/BP has remained relatively stable.
Economic factors may also have an impact on the choice between allo-SCT and TKI therapy, especially in the developing countries. Ruiz-Argüelles and colleagues reported data on 24 patients transplanted in CP1 using MSD and RIC in 4 Latin American countries; they mentioned the cost of allo-SCT in Mexico would be equal to approximately 200 days of imatinib 400 mg daily, thus making the case for allo-SCT. Similar observations were made by Gratwohl and colleagues when they analyzed the EBMT survey data; they noticed that the rates of allo-SCT fell after the introduction of imatinib in high-income countries in Europe but remained relatively stable for the lower-income countries, indicating persistent reliance on the “one-time” cost of allo-SCT as compared with expensive potentially “life-long” TKI therapy.
Allo-SCT compared with imatinib as front-line therapy in chronic phase
As imatinib was rapidly adopted as a standard front-line therapy for CP CML, with excellent results, randomized clinical trials to prove the superiority of imatinib to allo-SCT were considered unjustifiable. As a result, only indirect evidence exists with which to compare the two modalities. In a study by the German CML group, newly diagnosed CP CML patients with a matched siblings were offered matched-related donor allo-SCT; all others were given the best available drug therapy (IFN plus hydroxyurea at the time of initiation of this study). This type of treatment assignment based on donor availability is sometimes called genetic randomization. With a median observation time of 8.9 years, the survival was superior for patients in the drug treatment arm ( P = .049), particularly in the low-risk patients. The survival difference was most pronounced at 3 years with the two curves merging only at approximately 8 years, given steady disease progression in the drug treatment arm. Of note, two-thirds of the patients in the drug therapy arm were over time switched to imatinib because of failure of or intolerance to IFN/hydroxyurea. These data, though debated at the time, were the first to indicate superiority of drug treatment over allo-SCT in CP CML patients and to provide a compelling argument for superiority of TKI therapy as front-line treatment for CP CML patients in comparison with allo-SCT. In another retrospective analysis reported by Bittencourt and colleagues, CP1 CML patients who failed or were intolerant to IFN received either imatinib as second-line therapy (n = 174) or allo-SCT (n = 90) based on availability of donor and Sokal score. The imatinib group had significantly improved event-free survival (EFS) and OS at 5 years compared with the allo-SCT group, again indicating superiority of the imatinib in this setting.
A third more recent study from the German CML Study IV group took a slightly different approach. This group reported interim outcomes on 1242 CML patients who underwent 5-arm treatment randomization (imatinib 400 mg daily vs imatinib plus IFN vs imatinib plus cytarabine vs imatinib post-IFN failure vs imatinib 800 mg daily). Based on predefined transplantation criteria (high Hasford score and/or low EBMT score, imatinib-failure or AP/BP), 84 of these 1242 patients underwent allo-SCT between 2003 and 2008 (23% in CP1 as an elective option, 44% after imatinib failure in CP1, and 33% in AP/BP). The median age at the time of transplantation was 37 years (range, 16–62 years). All except 3 patients received imatinib pretransplantation. Seventy percent of CP patients and 42% of AP/BP patients had achieved cytogenetic remission at the time of transplantation. Three-year OS was 91% for patients in CP and 59% for patients in AP/BP. In the matched-pair analysis, survival at 3 years was similar for the patients who underwent allo-SCT in CP1 versus those who were treated with imatinib alone, though those assigned to transplant had a higher risk score. The investigators draw attention to the very low TRM of 8% in this study, which they attribute to improvements in supportive care, and also speculate that imatinib-induced reduction of tumor burden prior to transplant may have favorably influenced outcomes.
Although the debate over front-line therapy for adults with CP CML has been settled in favor of TKI therapy, whether the same could be said for pediatric patients with CML is not entirely clear. Cwynarski and colleagues reported retrospective EBMT data on the outcomes of 314 children (median age 14 years) who underwent allo-SCT for CML in the pre-imatinib era (between 1985 and 2001). The donor was an HLA-matched sibling in 58%, and 81% were in CP1 at the time of allo-SCT. The source of stem cells was BM for all patients. For CP1 patients, 3-year OS, TRM, and relapse rate for those receiving sibling donor versus URD was 75%, 20%, 17%, and 65%, 31%, 13%, respectively. Grade II to IV acute GVHD occurred significantly more in the URD group (52% vs 37% for sibling donors). Suttorp and colleagues reported 10-year prospective follow-up data on 176 children who underwent allo-SCT for CML from year 1995 to 2004 (median age 12.4 years). At the time of allo-SCT, 82% were in CP1 and 66% underwent matched sibling allo-SCT. For CP1 patients, OS was 64% with significantly better OS in the matched sibling group (OS 87% compared with 52% in matched URD transplant group, P = .002). Thus the outcomes in pediatric patients appear superior to those in the adults. Data with use of imatinib in children is limited and again, as is true for adults, very long-term data on imatinib use is unavailable. However, as is the case with adults, the current consensus is to use imatinib as front-line therapy for CML in children.
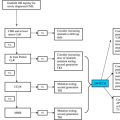
At present, 3 broad groups of patients are considered to benefit from allo-SCT:
- 1.
Those in AP or BP
- 2.
Those failing/intolerant to TKI therapy
- 3.
Those with TKI-resistant mutations such as T315I mutation.
Patients in AP/BP
Long-term outcomes of patients treated with single-agent imatinib for advanced-phase disease range from an OS of 37 to 47 months for AP, and of approximately 7 months for BP. Second-generation TKIs are superior to imatinib, but are not considered curative in advanced-phase CML. Allo-SCT provides the only potential for long-term survival for these individuals. However, the outcomes after allo-SCT for this group of patients remain suboptimal, especially for BP disease. Gratwohl and colleagues reported 2-year OS, TRM, and relapse rate for AP and BP disease as 47%, 37%, 28%, and 16%, 50%, 38%, respectively. Jiang and colleagues reported on the outcomes of AP patients who were assigned to treatment with imatinib versus an upfront allo-SCT. The treatment assignment was non-randomized and based on patient’s preference. The allo-SCT group had superior outcomes (6-year EFS and OS rates of 71.8% and 83.3%, respectively compared to 39.2% and 51.4%, respectively for the imatinib group). A multivariate analysis revealed that a CML duration greater than or equal to 12 months, hemoglobin less than 100 g/L, and peripheral blood blasts greater than or equal to 5% were independent poor prognostic factors for both OS and progression-free survival (PFS). When stratified by these 3 risk factors, the superiority of allo-SCT was seen in only intermediate (presence of one factor) and high-risk (presence of ≥2 factors) groups. Patients without any risk factor (low-risk) did equally well with imatinib or allo-SCT. Patients with advanced-phase CML remain a challenging group of patients with poor outcomes with currently available therapies, and every effort should be made to induce a second CP before allo-SCT.
Patients Who Fail TKI Therapy
As TKIs have become standard therapy for newly diagnosed CML patients, almost all patients with CML will have received imatinib and/or second-generation TKIs before proceeding with an allo-SCT. Even for patients with advanced disease, as the plans for allo-SCT are being formulated patients will usually be initiated on a TKI. Patients who have failed TKI represent the majority of those undergoing transplantation. So an important question is whether prior use of TKIs affects the transplant outcomes.
Effect of prior imatinib or second-generation TKIs on transplant outcomes
Deininger and colleagues reported retrospective data on 70 patients with CML who underwent allo-SCT after imatinib treatment, and compared the outcomes with a historical control group from EBMT registry. Eighty- four percent of the patients were in AP/BP prior to imatinib, and this was reduced to 44% prior to allo-SCT. Median duration of imatinib therapy was 97 days, and the median interval from diagnosis to transplantation was 22.6 months. When compared with the historical controls, the investigators found no influence of prior imatinib use on OS, PFS, and NRM. Another study from CIBMTR evaluated 409 patients (185 patients CP1; the rest advanced disease) who received imatinib before allo-SCT in comparison with 900 patients who did not receive imatinib before allo-SCT. In the multivariate model, exposure to imatinib prior to allo-SCT was associated with better OS. Leukemia-free survival (LFS) and rates of acute and chronic GVHD were similar in the two groups. For the patients beyond CP1 there was no difference in the OS, LFS, TRM, or relapse rate. Other groups have also reported that pretransplant use of imatinib is safe and is not associated with increased TRM.
In addition, response to imatinib prior to allo-SCT has been shown to improve clinical outcomes for CP patients. Oehler and colleagues reported allo-SCT outcomes of 69 patients who had received imatinib before allo-SCT. Those who had achieved major cytogenetic remission (MCyR) prior to allo-SCT had better outcome than those who did not (or who lost their cytogenetic response). The latter group had a statistically significantly higher hazard of mortality (hazard ratio 5.31, P = .03).
In the initial few years of the TKI era, allo-SCT was recommended after imatinib failure/intolerance. At present, second-generation TKIs (dasatinib and nilotinib) have shown excellent results in imatinib failure/intolerant patients with MCyR in up to 40% to 45% patients, and as such have been approved by the FDA for this indication. In addition, both dasatinib and nilotinib have shown excellent results as front-line therapy for CP CML, and have been recently approved by the FDA for front-line therapy. It is therefore not surprising that most patients now will have received a second-generation TKI at the time of consideration for an allo-SCT. Second-generation TKIs have also been reported to be safe when used prior to allo-SCT. In accordance with the aforementioned data, the recent European LeukemiaNet guidelines advocate allo-SCT after failure of a second-generation TKI (and not after only imatinib failure).
Patients with TKI-Resistant Mutations
Although many mutations have been identified in the Abl kinase domain, contributing to the resistance to TKIs, threonine-to-isoleucine substitution at position 315 of Bcr-Abl fusion protein (T315I mutation) leads to resistance to all first-generation and second-generation TKIs. Given the lack of effectiveness of the TKIs, early allo-SCT is recommended for such patients. Velev and colleagues reported outcomes of 8 patients with T315I mutation who underwent allo-SCT. At the time of allo-SCT 2 patients were in CP1, 3 patients were in AP, and 3 patients were in CP2. The best responses after allo-SCT were complete molecular remission (CMR) in 3 patients, complete cytogenetic response (CCyR) in 4 patients, and complete hematologic response (CHR) in 1 patient. The 2 patients in CP1 at the time of allo-SCT had the best outcomes. There have been other anecdotal reports of patients undergoing allo-SCT for T315I mutation, with good outcomes. Jabbour and colleagues reported allo-SCT outcomes of 19 patients who underwent allo-SCT due imatinib failure secondary to a BCR-ABL kinase domain mutation. The 2-year EFS and OS were 36% and 44%, respectively. They also reported that these outcomes were inferior (though encouraging) compared to outcomes of 28 other patients treated at the same institution with an allo-SCT for a non-mutation related imatinib failure. Nicolini and colleagues reported on 64 patients with T315I mutation who underwent allo-SCT. At the time of allo-SCT, 51% of patients were in CP and 26% were in blast crisis. The 2-yr OS rate for CP, AP, and BP disease were 59%, 67% and 30%, respectively. These results are better than that achieved by non-transplant strategies, though emerging data on the efficacy of third-generation TKIs such as ponatinib in T315I mutated patients may alter this argument in near future.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree