B Cell Activation and Signaling
Learning Objectives
• Describe the structure of the B cell receptor (BCR) complex
• Differentiate between the signaling and nonsignaling components of the BCR
• Define the role of CD2 in intracellular signaling
• Explain the usefulness of BCR cross-linking in the intracellular signaling process
• Identify the intracellular signaling pathways activated by cross-linked BCRs
• Recognize the definition of thymus-dependent (TD) antigen
• Identify the definition of a thymus-independent (TI) antigen
• Compare and contrast the molecular structure of TD and TI antigens
• Explain the interactions between TI types I and II antigens and cellular ligands
• Compare and contrast the antibody responses elicited by TI types I and II antigens
• Explain the role(s) of the B lymphocyte stimulator (BLyS) in B cell differentiation to plasma cells
• Identify the role of CD22 in B cell regulation
• Examine the role of atacicept in blocking B cell differentiation
• Explain how belimumab and epratuzumab inhibit B cell differentiation
Key Terms
B cell receptor (BCR)
B lymphocyte stimulator (BLyS)
Brutons tyrosine kinase (BTK)
Plasma cell
Thymus-dependent (TD) antigens
Thymus-independent (TI) antigens
Type I thymus-independent antigens
Type II thymus-independent antigens
X-linked agammaglobulinemia
Introduction
B cells are the major effector cells in an antibody-mediated immune response. The production of antibodies to protein antigens often requires interactions among T cells, B cells, and monocytes. The response is termed thymus-dependent (TD) antibody production. B cells are activated by the engagement of two receptors on the cell surface. However, some antigens can initiate antibody production without antigen processing or the help of T cells. These antigens are termed thymus-independent (TI) antigens. The activation of B cells may require the engagement of one or more receptors. As a consequence of intracellular signaling, B cells differentiate into antibody-producing plasma cells.
B Cell Receptor Complex
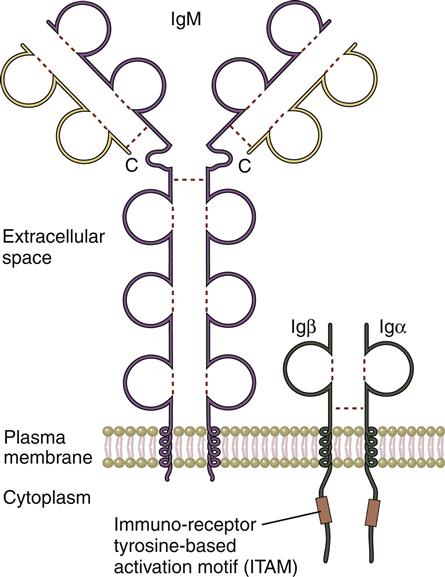
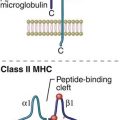
In the initial stage of B cell activation, antigens react with the membrane B cell receptor (BCR), which is a monomeric form of an antibody called immunoglobulin M (IgM; see Chapter 9). Monomeric IgM consists of two mu (μ)–heavy chains and two kappa (κ)– or lambda (λ)–light chains. M–heavy chains have four constant regions (CH1–CH4) and one variable domain (VH). Light chains are connected to the μ-chains and have one constant CL domain and one VL domain. Within the variable regions of both heavy and light chains are hypervariable regions. The combination of hypervariable regions in the heavy and light chains creates a three-dimensional pocket that determines antigen specificity (Figure 8-1). Each B cell expresses a homogeneous population of BCRs that recognize only one antigen. Therefore, 1 × 1013 B cell clones are needed to respond to all known antigens.
The BCR has a short intracytoplasmic tail and cannot transduce signals to the nucleus. To transduce signals, the BCR forms complexes with invariant immunoglobulin-alpha (Ig-α) and immunoglobulin-beta (Ig-β) molecules, which contain multiple immunoreceptor tyrosine-based activation motifs (ITAMs).
B Cell Receptor Signaling
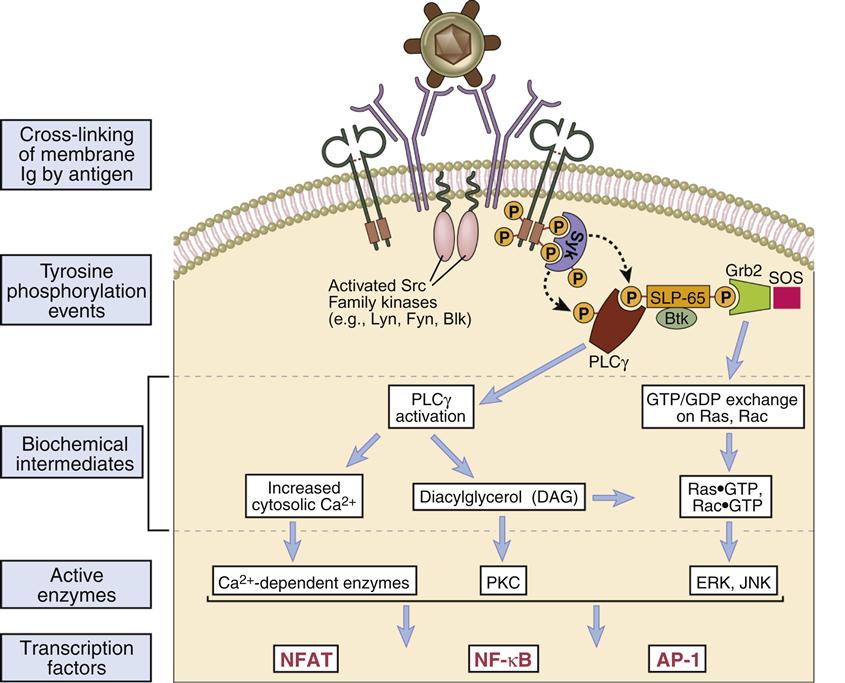
Activating signals are initiated by the cross-linkage of two BCRs by multi-valent antigens. Cross-linkage of two BCRs brings activated a sarcoma (v-src) kinase into the proper position for the activation of cellular signaling. The v-src enzyme phosphorylates the tyrosine residues on the Ig-α and Ig-β ITAMs (Figure 8-2). Phosphorylated ITAMs also act as “docking sites” for syk, an enzyme analogous to ZAP-70 in T cells. Syk activates two downstream signaling pathways whose products promote the synthesis of antibodies.
In one pathway, phosphorylation of phospholipase Cγ2 (the B cell isoform of phospholipase 1) activates the PLC calcium-dependent and DAG pathways. Activation results in the translocation of nuclear factor of activated T cells (NFAT) and nuclear factor–kappaB (NF-κB) to the nucleus. In a second pathway, phosphorlyation of an adaptor protein called SLP-65 (SH-2 binding leukocyte protein) allows interaction with Btk and Grb kinases. The Grb–SLP-65 complex recruits SOS (son of sevenless), which acts as a guanosine triphosphate–guanosine diphosphate (GTP–GDP) exchange protein that activates Ras and Rac. In turn, the MAP (mitogen-activated protein) kinase and the JNK (c-Jun N terminal kinase) pathways are activated with translocation of AP-1 to the nucleus. In B cells, AP-1 accelerates for cell division, which creates B memory cells and B cell differentiation into plasma cells.
Amplification of the B Cell Signal
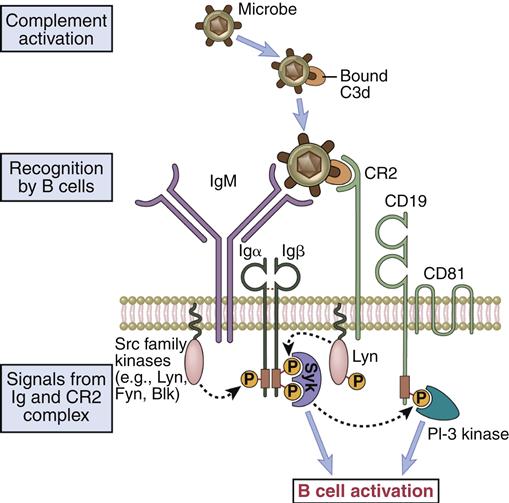
Like T cells, B cells require a second signal for activation. The second signal is provided by a B cell co-receptor complex that consists of CR2, CD19, and CD81 (TAPA-1). The CR2 molecule recognizes a decay product of complement called C3d that is bound to large-molecular-weight antigens or bacteria. Complement is a series of serum proteins that can be activated by polysaccharides (innate immunity) or antigen–antibody complexes (acquired immunity). Complement is discussed in detail in Chapter 11.
Amplification occurs when the multi-valent antigen binds to CD2 and cross-links multiple BCRs. Engagement of the BCR and CR2 activates a lyn kinase that phosphorylates the Ig-α and Ig-β ITAMs. Binding also reorients CD19 and CD81 to bring them into proximity with Ig-α and Ig-β ITAMs (Figure 8-3).
Phosphorylation of CD19 activates a PI-3 kinase, which initiates the Ras–MAP and Jak–STAT signaling pathways that stimulate B cell differentiation, survival, and growth.
Thymus-Dependent Antigens
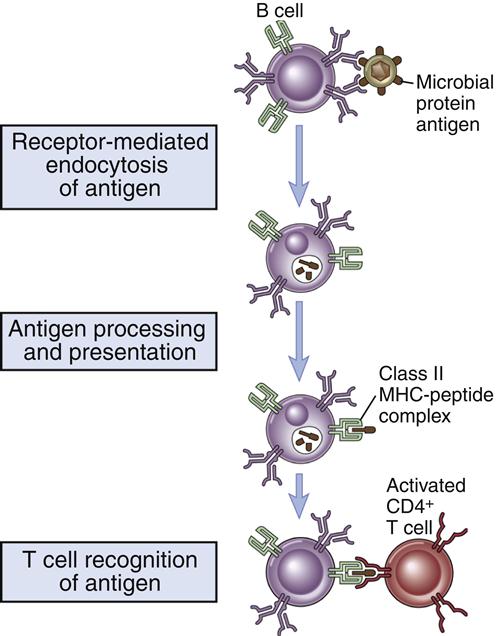
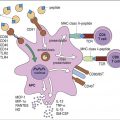
Some antigens require the help of T cells for the stimulation of B cells to produce antibodies. In the parafollicular cortex (T cell zone) of the lymph node, dendritic cells process and present antigens in the context of class II molecules. Antigens are presented to CD4Th2 cells. At this point, the T cell is activated but does not know which B cell needs help to produce antibodies.
B cell activation occurs in the lymph node B cell zone. The unprocessed antigens that activated the T cell bind to antigen-specific BCRs. After interaction between the BCR and antigens, B cells internalize, process, and present epitopes in the context of class II molecules (Figure 8-4).