IMPORTANCE OF ANTIBIOTIC STEWARDSHIP
America’s 15 600 NHs house an estimated 1.3 million complex, frail residents, in whom infections can evolve rapidly and present atypically.
1,2 It is, therefore, not surprising that NHs are the healthcare setting, other than acute care hospitals, with the highest rates of antibiotic use. According to one study, at any given day, 10.8 percent of NH residents were taking an antibiotic
3; another found that, on average, NH residents received an antibiotic prescription every 77 days.
4 Furthermore, in contrast to hospital patients, NH residents live there for months and sometimes years, and so the antimicrobial resistance produced by high levels of antibiotic exposure remains and compounds itself, to the point the NHs have some of the highest rates of antibiotic resistance found anywhere.
NH residents are frequently colonized with multidrugresistant organisms (MDROs), with the prevalence varying across countries and regions. In one study, in which the nares, oropharynx, groin, and perianal area were cultured from a sample of 82 residents of NHs in Michigan, among persons without an indwelling device, 56% carried methicillin-resistant
Staphylococcus aureus (MRSA), 15% carried vancomycin-resistant
Enterococcus (VRE), 25% carried ceftazidime-resistant Gram-negative bacteria, and 69% carried ciprofloxacin-resistant Gram-negative bacteria. Another study, of 605 NHs in California, identified the prevalence of MDROs as 45%.
5 In European NHs, the reported prevalence of MRSA ranged from 0.8% in Sweden to 57% in Spain; among invasive clinical isolates, the prevalence was 54% for MRSA and 23% for VRE.
6,7 Rates were higher among persons with indwelling devices, and new acquisition of resistant organisms occurred frequently after moving into the NH.
8 Additionally, due in large measure to the residential, less medicalized nature of NH settings, the U.S. Centers for Disease Control and Prevention’s (CDC) Emerging Infections Program (EIP) surveillance study of invasive MRSA infections reported a higher onset of invasive MRSA colonization in NHs than in hospitals.
9Urinary tract colonization with antibiotic-resistant organisms has been demonstrated worldwide. In France, of 19 471 culture-positive urine samples identified, Enterobacteriaceae were the predominant bacteria isolated in residents aged 65+ years, and that NH residents had a 40% greater risk of getting antibiotic-resistant to Enterobacteriaceae than persons living in the community.
10 Again, regional variation is present in resistance rates and in distribution of specific Enterobacteriaceae bacterial types.
11 Bacterial colonization is more often associated with indwelling devices, most importantly the urinary catheter. In Michigan, a study of 100 residents noted that, when compared to those without indwelling devices, those with any devices are 2 times more likely be colonized with MRSA, 1.1 times with VRE, and 5.6 times with ceftazidime-resistant Gramnegative bacteria.
12 Among persons with a urinary catheter, the probability of becoming colonized with antibiotic-resistant bacteria in the groin or perineal area, compared to that of persons without a catheter, is 4.5 times greater for MRSA, 2.7 times greater for VRE, and 7.8 times greater for ceftazidime-resistant Gram-negative bacilli (GNB).
5,12Since colonization rates with resistant organisms are high in NHs, it is not surprising that outbreaks of infections with MDROs are also common in these settings. Some important examples are MRSA, VRE, and multidrug-resistant (MDR) GNB infections. MRSA outbreaks in NHs have been reported since the 1980s
13,14 and VRE outbreaks since the 1990s.
15 MDR Gram-negative infections have also been reported since the late 1980s.
16,17 Since the turn of the century, such outbreaks have become so commonplace as to not merit case reports. NHs with ventilator units, including LTACs, are particularly prone to developing MDRO outbreaks and, in fact, merit especially stringent infection control and antibiotic stewardship policies and practices.
18Recent literature has focused more on
Clostridioides difficile infections (CDI), the burden of which has increased markedly in recent decades and is associated with substantial morbidity and mortality in NHs. The incidence
of CDI is 8.6 times higher in people age 65 years and above compared to younger adults, possibly due to changes in the intestinal microbiota with population aging. Furthermore, 80% of CDI deaths occur in persons aged 65 years and older.
19 CDI is often associated with antibiotic use, in the United States, of the 262 NH-onset CDI cases in 2012, 76% had a history of antibiotic therapy and 75% of a recent hospitalization.
20 CDI infections occasionally occur as outbreaks; for example, a 1985 outbreak in a NH in Baltimore resulted in 49 CDI cases and 9 deaths.
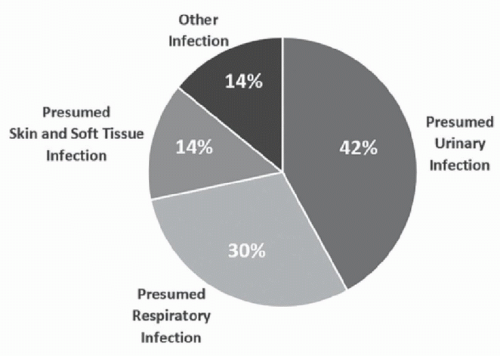
21The most common reasons antibiotics are prescribed for NH residents are suspected urinary tract infections (UTIs), respiratory infections, suspected skin and soft tissue infections (SSTIs), and other infections. Urinary tract and respiratory conditions are by far the most common reasons antibiotics are prescribed and are responsible for over 2/3 of prescriptions. Within the “other” category, the most common diagnosis is now
C difficile infection (
Fig. 46-1).
Between 25% and 75% of the antibiotics prescribed in NHs are thought to be unnecessary.
22,23 One factor supporting this contention is the wide variability in the antibiotic use across countries and regions.
6,24 In Europe, the prevalence of NH residents on at least one antibiotic is <2% in Croatia, Germany, and Hungary and more than 10% in the Czech Republic, Denmark, United Kingdom (Northern Ireland), and Greece.
6 In the United States, antibiotic use is quite high in NH, with a point prevalence of ˜10% and 87% receiving at least one antibiotic in a year.
25 Likewise, there is also variation in antimicrobial use between NHs.
24Furthermore, prescribing often (>50% of the time) fails to meet expert guidelines.
26 In Wisconsin, 56% of the antibiotic prescribed in NH did not meet the Loeb “minimum criteria” for instituting antibiotics in NH residents,
27 including 52% of prescriptions for lower UTI and 22% for skin and soft tissue infections.
28 Similarly, 46% of antibiotics prescribed are not consistent with NH prescribing guidelines in Ireland.
29 Additionally, prophylactic antibiotics are commonly used in NHs, which is contrary to most antibiotic prescribing guidelines.
30The overuse of antibiotics is directly linked to the occurrence of antibiotic resistance.
31,32 Studies have shown that rates of antibiotic prescribing and thus resistance can be reduced by antibiotic stewardship.
33,34,35,36 Therefore, in an attempt to decrease the rates of antibiotic resistance among NH residents, antibiotic stewardship programs have been introduced.
REGULATORY REQUIREMENT FOR ANTIBIOTIC STEWARDSHIP IN U.S. NURSING HOMES
In 2016, the U.S. Centers for Medicare and Medicaid Services (CMS) mandated that all U.S. nursing homes develop an infection prevention and control program that included antibiotic stewardship; written standards, policies, and procedures; a system to monitor antibiotic use; and at least one staff member designated as an infection preventionist. The implementation timetable mandated that the antibiotic stewardship program be initiated by November 2017 and that the infection preventionist should be in place and serving on the facility’s quality assessment and assurance committee by November 2019.
37Enforcement of these new regulations became another function of the CMS-certified NH survey teams, who on average visit each NH once a year. Federal Tags (citation types) that relate to the new CMS infection prevention and control requirements include F-Tag 441 (infection control), F-Tag 329 (unnecessary drugs), F-Tag 332/333 (medication errors), and F-Tag 428 (drug regimen review). Even prior to the new (2016) CMS requirements, 32% of NHs were reported to receive a deficiency citation for lapses in infection control
38; so, it is likely that the proportion now is even higher.
To assist NHs seeking to establish antibiotic stewardship in response to the CMS mandate, a number of national efforts have been implemented. Among the most prominent are the following:
The CDC developed a Web site that described six “core elements” of antibiotic stewardship in NHs—leadership commitment, accountability, drug expertise, policy and practice change, tracking and reporting antibiotic use and outcomes, and education—and provided educational materials.
39
The U.S. Agency for Healthcare Research and Quality (AHRQ) developed a NH antimicrobial stewardship guide, which assembled and made publicly available a variety of toolkits, including ones on (1) implementing, monitoring, and sustaining an antibiotic stewardship program, (2) when to initiate antibiotics, (3) choosing the right antibiotic, and (4) engaging residents and families in stewardship activities.
40
AMDA—the Society for Post-Acute and Long-Term Care Medicine published a model antibiotic stewardship policy that NHs could adapt and modify instead of having to start
de novo.
41
The University of Nebraska Antibiotic Stewardship Assessment and Prevention Program developed and placed on line a variety of resources, including templates for activity reports, antibiograms, antimicrobial stewardship meetings, infection and antibiotic stewardship monthly logs, and tools for staff to use in documenting patient status and determining whether antibiotics are warranted.
42
The Cecil G. Sheps Center for Health Services Research of the University of North Carolina at Chapel Hill, in collaboration with the North Carolina Statewide Program for Infection Control and Epidemiology (SPICE), developed and marketed a start-up package of materials that NHs could use to implement infection tracking and education of nursing staff, medical providers, and patients/families.
43
The CDC developed a 12-hour, online training program for NH staff who will function as CMS-mandated infection preventionists.
44
HOW AND WHY NH ANTIBIOTIC STEWARDSHIP MUST DIFFER FROM HOSPITAL PRACTICE
NHs differ from hospitals in many important ways that create challenges for antibiotic stewardship. Among the major differences are the presence of fewer medical professionals on site, decision-making that is frequently done on the telephone and is less readily supported by laboratory testing, and a relative lack of resources for infection control.
The typical U.S. nursing home has ˜120 beds and is part of a for-profit chain. Most of the day-to-day care is provided by certified nursing assistants, who have modest training (the federal requirement is 75 hours initial training plus 10 hours annually of continuing education).
45 Licensed nurses are on site 24 hours per day, usually at a ratio of between 20 and 40 patients per nurse, and so their main
functions are medication administration, communication with families and medical providers, and supervision of the certified nursing assistants. Medical care is usually provided by a small group of physicians, one of whom is the medical director and visits the facility at least once a week; a nurse practitioner or physician assistant, who often splits his or her time between several NHs; and a contingent of community physicians, each of whom has a small number of patients and may visit the NH as infrequently as once a month. According to one report, the average NH physician spends 2 hours a week seeing patients in that setting, with only a small (but growing) minority committing a substantial portion of their workload to NH care.
46Because medical providers are rarely on site, a substantial proportion of treatment decisions are made over the telephone, without a physician, nurse practitioner, or physician assistant examining the patient.
47 This means that the opinions and attitudes of nursing staff, many of whom are licensed practical nurses or licensed vocational nurses, are more prominent than in hospitals. Families play a substantial role in decision-making as well; so, the influence of physicians is diluted by others, and attitudes toward infection and antibiotic use in the general population and the nursing community are important influences on antibiotic prescribing.
48 Furthermore, clinical pharmacists, who are readily accessible as consultants to hospital physicians, typically visit a NH only once a month; so, their input is also largely unavailable.
Adding to the challenge of decision-making around suspected infections is the fact that all laboratory studies, except point-of-care glucose testing and, in some settings, a urine dipstick, are sent out to commercial laboratories, often returning 24 hours or more after a sample was obtained. Chest radiograph results tend to come more quickly, as services bring portable machines to NHs and the reports are often available within hours; however, the quality of the portable images is not as good as hospital studies, resulting in radiologists more frequently expressing uncertainty about findings (eg, whether or not an infiltrate is present).
49Another issue in NH decision-making around suspected infections is the fact that a high proportion of residents have Alzheimer disease or another dementia.
50 This can make it difficult to obtain a reliable history and, therefore, leads to staff relying on subtle behavioral changes, such as fatigue, agitation, or lack of appetite to assess for possible infection. As a result, these symptoms can sometimes be overinterpreted as infection and result in prescribing an antibiotic “just to be safe.”
48NH resources for infection prevention and antibiotic stewardship are similarly limited, particularly in contrast to the acute care hospital.
51
DEVELOPING AN ANTIBIOTIC STEWARDSHIP PROGRAM IN A NURSING HOME
According to the CDC, a NH antibiotic stewardship program should have six core elements: leadership commitment, accountability, drug expertise, policy and practice change, tracking and reporting of antibiotic use, and education.
39 Each of these elements is discussed below and summarized in
Table 46-1.
Leadership Commitment
Given the magnitude and growth of antimicrobial resistance globally and particularly in NHs, antibiotic stewardship should be a priority. However, changing established ways of doing things is difficult and requires committed leadership. In the NH, this should largely be from four individuals, who together should comprise the antibiotic stewardship team: the medical director, the director of nursing, the consultant pharmacist, and the infection preventionist nurse (who oversees all infection control for the facility). In the background, supporting these efforts should be the NH administrator and, if the facility is part of a chain, the administrative structure of the larger organization.
Accountability
The leadership commitment must have teeth in the form of ability to set goals, create policies, and monitor infectionrelated prescribing. The medical director should be empowered to set standards and create policies regarding antibiotic prescribing; the director of nursing should have the authority to implement standards and policies related to monitoring and reporting changes in the condition of patients; and the infection preventionist should be responsible for tracking and reporting antibiotic use, as well as effectively implementing policies related to infection control and surveillance. Additionally, the consultant pharmacist should have the authority to review antibiotic prescribing and make recommendations to the medical director and medical staff.
Drug Expertise
Professional standards and guidelines for antibiotic use in postacute and long-term care settings are changing, and the antibiotic stewardship team needs to stay abreast of these standards and recommendations. The point person for this should be the consultant pharmacist, who should have specific training in infectious diseases and/or antibiotic stewardship. Additionally, the medical director should have a designated infectious diseases specialist that he she can consult with informally about antimicrobial-related questions.
Policy and Practice Change
At the heart of the antibiotic stewardship program should be policies and procedures aimed at standardizing care, meeting professional guidelines and addressing specific targets set by the antibiotic stewardship leadership team. An example might be policies on empiric antibiotic selection for suspected urinary tract and respiratory infections, which are implemented as a means of achieving the goal of reduced fluoroquinolone use.
A sample overall facility antibiotic stewardship policy has been published and is available on line from AMDA—the Society of Post-Acute and Long-Term Care Medicine at http://www.jamda.com/article/S1525-8610(17)30430-9/fulltext.
41
Tracking and Reporting Antibiotic Use
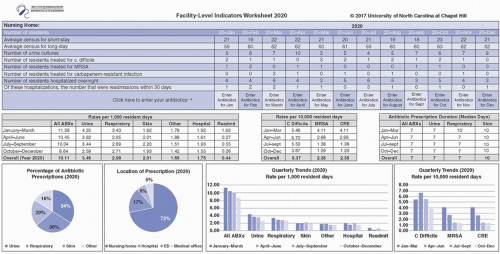
Perhaps the most important first step after assembling the antibiotic stewardship team is to begin effectively tracking antibiotic use and infection incidence. In most cases, this should be done manually on at minimum a weekly basis by the infection preventionist, by auditing all medication orders and the charts related to them and then entering the data onto a spreadsheet such as the one displayed in
Figure 46-2. With increased sophistication of electronic
health records, portions of this process or the entire process will in the future be largely automatic, and then the role of the infection preventionist will evolve to focus on evaluating and reporting these results rather than tracking.
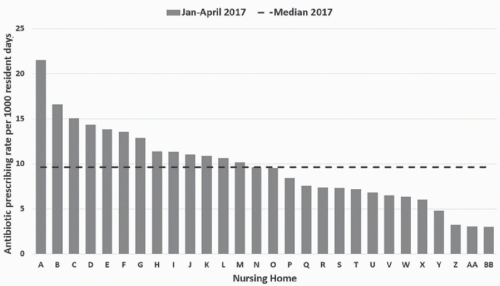
Provision of benchmarks and comparative data is important in allowing NHs to see how they are performing compared with their peers. Such comparison data can be created by NH chains for member facilities or by regional organizations such as the CMS Quality Improvement Organization (QIO) Program.
Figure 46-3 displays an example of such a comparative data graphic.
In addition, the CDC National Healthcare Safety Network (NHSN) has created several long-term care facility component modules that NHs can use to report data; as of 2019, the available module topics included MDR infections, UTIs, and prevention process measures.
52 The biggest challenge for NHs in NHSN participation is that the person reporting the data (ie, the preventionist) must individually enroll in NHSN, a process that takes considerable time and effort, so, whenever the preventionist position turns over, the need for the new person to enroll can create lapses in reporting.
Education
Changing practice requires education, and antibiotic stewardship requires frequent educational outreach to medical staff, nursing staff, patients, and families. This is especially true in the area of antibiotic stewardship, because professional standards often conflict with traditional attitudes. An example is the common disconnect between nurses and physicians or between family members and medical providers, around the significance of urine odor and color, described later in this chapter.
53