Radionuclide
T1/2 (h)
Emission
Energy deposited (MeV maximum)a
Source
Ra-223
273.6
4 Alpha
2 Beta
4 Gamma
2 X-ray
Alpha 5.6, 6.8, 7.4, (6.6 or 7.50)
Beta 0.48, 0.50
Gamma, 0.154, and 0.270, 0.351
X-ray 0.81, 0.84
Th-227 decay chain
At-211
7.21
1 Alpha
0 Beta
0Gamma
2 X-ray
Alpha 5.86 or 7.45
Beta none
Gamma none
X-ray 0.077–0.079
Cyclotron Bi-209 (alpha, 2n)
Bi-212
1.01
1 Alpha
1or 3 Beta
3 Gamma
Alpha 6.05 or 8.8
Beta 0.83 or (0.44, 0.53, 0.65)
Gamma (0.51, 0.58, 2.60, 0.86)
Th-228 decay chain. Ra-224 generator
Bi-213
0.76
1 Alpha
3 Beta
1 Gamma
Alpha 8.4 or 5.9
Beta 0.49, 0.32, 0.2
Gamma 0.440
Ac-225 decay chain
Ac-225
238
4 Alpha
3 Beta
2 Gamma
Alpha 5.8, 6.3, 7.1, (8.4 or 5.9)
Beta 0.49, 0.32, 0.2
Gamma, 0.218, 0. 440
Th-229 decay chain or Ra-226 (p,2n) reaction
Pb-212
10.6
1 Alpha
2or 4 Beta
3 Gamma
Alpha 6.05 or 8.8
Beta 0.177 and 0.83 or (0.44,0.53, 0.65)
Gamma (0.51, 0.58, 2.60, 0.86)
Th-228 decay chain
Several clinical examples of therapeutic applications of alpha emitters exist, predominantly using antibodies as the targeting vehicle [13–16]. Radiolabeling methods to attach the various alpha emitters to radiopharmaceuticals have been described [17]. Ra-223 dichloride (formerly known as Alpharadin and more recently known as Xofigo™) is the only approved alpha emitter radiopharmaceutical to date [18]. While Ra-223 is only approved in the setting of metastatic prostate cancer, promising results have been reported in breast cancer [19, 20], and a single case report for a patient with metastatic paraganglioma has been published [21].
Although the clinical applications of alpha emitters in NETs are limited thus far, their feasibility and safety have been evaluated in diverse clinical studies. In this chapter, we review the rationale for alpha emitters (see above); touch on some of the clinical trials in the non-NET literature, as they provide some preliminary feasibility and safety data for alpha-based radiopharmaceuticals that may be applicable to NETs; and discuss preclinical and, where available, clinical work with alpha emitters in NETs.
21.2 Application of Alpha Emitters in Cancer (Preclinical and Clinical)
21.2.1 Ac-225 and Bi-213
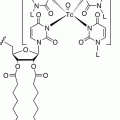
Ac-225 for clinical use is typically produced from Th-229 [22], although accelerator-based methods for Ac-225 production have been described. Ra-226 (p,2n) and Ac-225 are yet to be used clinically [23]. Bi-213 is normally obtained from an Ac-225/Bi-213 generator [24]. Scheinberg et al. from Memorial Sloan Kettering have been pioneers in the field of alpha therapy, performing the first radioimmunotherapy studies in humans using Bi-213 huM195 directed against CD33 in patients with acute myelocytic leukemia (AML) [25]. Bi-213 has predominantly been attached to antibodies via a chelator such as 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA), the same chelate used for attaching metals to somatostatin analogs. In clinical studies, activities ranging from 0.6 to 1.6 GBq were administered intravenously [15]. A clinical trial combining Bi-213 huM195 with cytarabine in patients with AML delivering activity ranging from 18.5 to 46.25 MBq/kg showed a maximum tolerated dose (MTD) of 37 MBq/kg due to myelosuppression. That study concluded that the combination of alpha-emitting radionuclide therapy with cytarabine was tolerable and could induce remission in patients with AML [25].
Bi-213 9.2.27 anti-melanoma monoclonal antibody has been used in patients with metastatic melanoma. Following intravenous administration of 46–925 MBq of the Bi-213 9.2.27 antibody, 38 patients were followed for response and toxicity. The MTD was not achieved and no adverse events occurred. An objective partial response rate of 10 % was observed, with 40 % stable disease at 8 weeks and a median survival of 8.9 months [26].
The utilization of intralesional delivery of Bi-213 with anti-melanoma 9.2.27 monoclonal antibody was well tolerated and induced tumor kill [27]. Furthermore, intra-tumoral injection of Bi-213 substance P in patients with brain gliomas (1.1–7.36 GBq) demonstrated proof of principle, with pathological evidence of tumor necrosis on resection and no relevant toxicity [28].
One disadvantage of Bi-213 is its short physical half-life of 0.76 h, making it logistically difficult to elute and label enough activity in a timely manner, thus requiring production on-site with an Ac-225/Bi-213 generator. To solve this problem, McDevitt and Scheinberg’s group at MSKCC developed the concept of an in vivo generator using Ac-225, a longer-lived alpha emitter that could be conjugated to antibody and results in the generation of multiple alpha particles in situ [29]. This longer-lived Ac-225 with a T1/2 of 240 h allows for the production of a radiopharmaceutical that can be shipped from a local manufacturing site to end users. Its use has been validated in preliminary phase I multicenter trials [13, 30] with Ac-225 lintuzumab (huM195) provided by a commercial manufacturer, Actinium Pharmaceuticals, Inc (www.actiniumpharma.com).
The use of Bi-213 DOTATOC in metastatic NETs to the liver has been reported [31]. The rationale for intra-arterial (IA) delivery is the superiority of IA hepatic administration of Ga-68 DOTATOC that resulted in a 3.7-fold higher intrahepatic tumor accumulation compared with IV infusion [32], consistent with significant first-pass extraction. Given the short T1/2 of Ga-68 DOTATOC, it was not clear how translatable this would be when a longer-lived radionuclide such as In-111, Lu-177, or Y-90 was utilized. Thus, in a subsequent study of a longer-lived In-111 DOTATOC, this group also showed tumor uptake immediately after IA that was four times greater than systemic administration, decreasing to 1.3 times greater at 72 h [33]. In 15 patients treated with IA somatostatin analogs of Lu-177 or Y-90 DOTATOC, a 7 % complete response (CR), 53 % partial response (PR), and 40 % stable liver metastasis were noted [33]. However, the pharmacokinetics of longer-lived isotopes showing a decrease in tumor uptake at 72 h compared to immediately post-infusion [33] suggest that short-lived radionuclides such as Bi-213-labeled somatostatin analogs are preferential compared to longer-lived isotopes when locally delivered to tumor, given that they would deliver their payload before significant release from tumor.
Animal studies with Bi-213 or Ac-225 somatostatin analogs have shown promising results in preclinical tumor models [34, 35]. Given the favorable preclinical data with Bi-213 DOTATOC, seven patients with NETs refractory to cold octreotide and Y-90/Lu-177 DOTATOC received compassionate use of Bi-213 DOTATOC IA [31]. An Ac-225/Bi-213 generator was used to obtain the Bi-213 [36]. The Bi-213 DOTATOC had radiochemical purity of 99.7 ± 0.3 %, and the specific activity was typically 80 MBq/nmol peptide at the time of injection. Injections were performed 15 ± 2 min after generator elution. Tumor concentration was demonstrated by planar and SPECT imaging of the 414 keV gamma ray of Bi-213. These patients received 1–5 treatment cycles (median 4.5) with single doses ranging from 3.3 to 10.5 GBq (median 7.1 GBq) and cumulative activities of 3.3 to 20.8 GBq (median 17.65 GBq). Enduring responses in liver metastasis were seen in all patients, and no evidence of tumor progression in the liver was noted by RECIST criteria during a follow-up period of 24–34 months, resulting in 1 CR, 2PR, and 3 SD. Response of extrahepatic disease was also noted in several patients. Hematopoietic toxicity was less than that observed in patients who had received Lu-177 or Y-90 DOTATOC. Grade 2 thrombocytopenia was seen in one patient, and no greater than Grade 1 thrombocytopenia was noted in other patients. Other hematologic toxicities were mild. One patient was diagnosed with MDS and AML at 2-year follow-up. The mean GFRs determined with Cr-51 EDTA were 115 ml/min, 101 ml/min, and 83 ml/min at baseline, 1 year post-therapy, and 2 years post-therapy, respectively. Only one of seven patients had a serum creatinine of 1.5 times higher than the upper limit of normal (ULN). Notably, these patients had received prior peptide receptor radionuclide therapy, which on its own can contribute to a decrease in GFR. These preliminary results, which demonstrated tumor response at therapeutically effective doses with limited toxicity, are encouraging [31].
A preliminary dose escalation trial of Ac-225 DOTATOC has been reported in abstract form on the treatment of patients with progressive NETs. Thirty-six patients were administered 48 treatments of Ac-225 DOTATOC intravenously, with 17 patients receiving 2-year follow-up. The single MTD was 40 MBq. Alternative multiple fractions with 25 MBq every 4 months or 18.5 MBq every 2 months were delivered. A cumulative activity of 75 MBq was found tolerable with respect to delayed toxicity. Detailed response data was not provided, although radiologic treatment responses were observed in some patients [37].
21.2.2 Lead-212 (Pb-212)
Pb-212 is another alpha-emitting radionuclide that has undergone extensive preclinical work by Brechbiel et al. at the NIH [38] and some clinical evaluation. It can be obtained from a Ra-224 generator [39]. The clinical development of Pb-212 has been promoted by AREVA Med (http://www.arevamed.areva.com). Chelators utilized for binding Pb-212 include DOTA or 2-(4-isothiocyanotobenzyl)-1,4,7,10-tetraaza-1,4,7,10-tetra-(2-carbamoyl methyl)-cyclododecane (TCMC) chelates, with some advantage in the stability of the complex of Pb with TCMC [40]. Pb-212 has been conjugated to trastuzumab and used for intraperitoneal administration in patients with metastatic HER2-positive malignancy [41]. For clinical trials, the TCMC-chelated trastuzumab was radiolabeled on-site because of the short 10.6 h T1/2 of Pb-212. Patients were administered escalating doses of Pb-212 TCMC trastuzumab intraperitoneally at 7.4, 9.6, 12.6, 16.3, and 21.1 MBq/m2 after pre-dosing with intravenous administration of trastuzumab at 4 mg/kg. This proof of principal study showed little toxicity, as expected, based on dosimetric estimates. Assessment of tumor response was difficult to evaluate in these patients with intraperitoneal disease [42].
21.2.3 Bi-212
Bismuth-212 is obtained from the decay chain of Th-228 using a generator system [39]. It has a half-life of 60.6 min and emits one of two alpha particles (Table 21.1). Part of its decay scheme also involves emission of a 2.6 MeV gamma ray that is difficult to shield and may pose radiation safety issues. Although some preclinical work has been performed with Bi-212-radiolabeled antibodies [43, 44], no preclinical studies in NET models or clinical studies have been performed. Preclinical work with Bi-212 DOTPM, a bone-seeking radiopharmaceutical, has also been performed [45]. It appears that rather than Bi-212 development, the approach with the Bismuth radionuclide has gravitated to Bi-213 as previously discussed.
21.2.4 At-211
At-211 is a cyclotron-produced alpha emitter with a 7.2 h T1/2 [46]. Various preclinical trials have been performed with At-211-radiolabeled (1) antibodies [47–52], (2) MIBG analogs [53–55], and (3) octreotate analogs [56]. Zalutsky and his group have been pioneers in the use of At-211-labeled pharmaceuticals and methods for radiolabeling proteins [46, 50, 57].
At-211-labeled ch81C6 anti-tenascin antibody has been used clinically via intracavitary delivery in patients with primary recurrent supratentorial tumors that were candidates for resection. After gross resection of the tumor, a Rickham reservoir and catheter were placed within the surgical resection cavity (SCRC). The SCRC was then directly injected with 71–347 MBq of the At-211 ch81C6. Some Grade 2 neurologic toxicity was noted. The 2-year survival probability was 61 % and the median overall survival was 54.1 weeks [14]. Because At-211 is a halogen and accumulates in the thyroid, thyroid blocking is required due to the possible release of free At-211. Minimal toxicity was observed in these patients [14].
Another study with At-211-labeled MX35 F (ab) 2 in patients with ovarian cancer used activity of 83–355 MBq delivered intraperitoneally. This study concluded that therapeutically effective amounts of i.p. therapy could be administered without causing any acute or deterministic radiation toxicities [52]. In this and other studies with At-211, gamma camera imaging has been performed to assess biodistribution and targeting [58].
Preclinical work has been done with octreotate derivatives that have been modified with two prosthetic groups: one containing a carbohydrate unit and the other, a tin group-containing aryl moiety, both amenable to At-211 labeling [56]. Unfortunately, these derivatives have less than ideal tumor targeting and internalization, and clinical trials have not been performed.
Preclinical studies have also been performed with At-211-labeled analogs of MIBG. Norepinephrine transporters are overexpressed in neuroblastoma, PHEO/PARA, carcinoids, and other NETs. Iodine-labeled MIBG uptake is based on binding to the norepinephrine transporter and has been used extensively for the imaging and therapy of these NETs [59–61]. Based on MIBG, Zalutsky et al. have developed various At-211-labeled analogs of MIBG with high affinity. The first reagent labeled was meta-[At-211] astatobenzylguanidine [49]. Following a technique used for the preparation of no-carrier-added MIBG, At-211 MABG has been prepared in good yields [49], and it behaves in vitro like MIBG with respect to its in vitro binding to SK-N-SH human neuroblastoma cells. Its tissue distribution in normal mice has been determined [62]. In clonogenic assays with the SK-N-SH human neuroblastoma cell line, the inherent radiosensitivity, or Do value, of [At-211] MABG was 0.2 kBq/ml compared with 384 kBq/ml for no-carrier-added I-131 MIBG, indicating a more than 1,000-fold greater cytotoxicity for the alpha particle-emitting analog compared to I-131 MIBG under single-cell conditions [63]. A kit method for the synthesis of relatively high amounts of At-211 MABG in good radiochemical yields has been developed; this method is adaptable to a kit formulation, and the quality control characteristics of the final dose are consistent with those appropriate for clinical studies [54]. To date, however, no clinical trials have been performed with At-211-labeled MABG analogs.
21.3 Bone Metastasis: NETs and PHEO/PARA
Many cancers present or develop bone metastases, which can lead to adverse clinical sequelae including pain, fractures, spinal cord compression, bone marrow dysfunction, and hypercalcemia, known as skeletal-related events (SRE). These lead to significant morbidity, which impact performance status and patient quality of life.
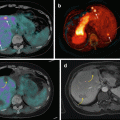
Various NETs also frequently metastasize to the bone. In a report of 86 consecutive patients with carcinoid tumors that had metastasized to the liver, the frequency of bone metastasis was 10 % (9/86 patients) [64]. The median interval between diagnosis of metastatic carcinoid and bone metastasis was 37 months. In that report, they evaluated the use of bone scan, In-111 pentetreotide, and I-131 MIBG for detection. In all patients with metastatic bone disease, the disease was noted on the bone scan, whereas I-131 MIBG detected only two of nine patients with bone metastasis and In-111 pentetreotide detected only four of eight patients. Other reports have shown sensitivity for bone metastasis of 70 % for In-111 pentetreotide scintigraphy and 90 % for bone scintigraphy [65]. In contrast to In-111 pentetreotide, the sensitivity of newer Ga-68 somatostatin analogs has been much higher. In one study, Ga-68 DOTANOC PET/CT was able to identify more lesions than CT (246 vs. 194), resulting in higher sensitivity (100 % vs 80 %) and specificity (100 % vs. 98 %) [66]. Similarly, using Ga-68 DOTATOC PET/CT, the sensitivity for detection of skeletal metastases was 100 % compared to 58 % for CT [67]. This provides an opportunity for the use of somatostatin analogs such as DOTANOC, DOTATOC, and DOTATATE radiolabeled with an alpha emitter for targeting bone and soft tissue disease in NETs.
In an autopsy series of NETs, the incidence of bone metastases was much higher than recognized clinically, with an incidence of 42 % [68]. NETs and PHEO/PARA involving bone had a high incidence of SRE [69]. The most common SRE in PHEO/PARA was severe pain (33 %), followed by pathological fractures (27 %) and spinal cord compression (25 %). Median time from diagnosis to first bone metastasis to first SRE was 4.3 months. These SREs usually happened after the bone metastases were diagnosed, and SRE were often the first manifestation of malignant disease as well (31 %) [70]. FDG PET imaging was able to identify 95 % of skeletal metastases, allowing the use of this reagent for assessment of tumor response. In contrast, MIBG identified only 70 %; it is important to identify this group, which can then be directed toward MIBG therapy. The exact incidence of bone-only metastatic disease has not been well determined. In a series of 15 patients with metastatic PHEO treated with I-131 MIBG, four patients had soft tissue disease only, and 4 had bone disease only [71].
Overall, most NET bone metastases are found to be osteoblastic [72, 73], which has implications for bone-seeking radionuclides, as they are more likely to localize in blastic bone lesions than in exclusively lytic lesions and in bone scan-positive rather than bone scan-negative disease. Given the incidence of metastatic bone disease that accumulates bone-seeking radiopharmaceutical in NETs, this should allow the use Ra-223 dichloride, which requires having a positive bone scan. In addition, PHEO/PARA have a high incidence of metastatic disease to the bone with up to 71 % incidence reported in a series of 137 malignant PHEO [70]. Most of the PHEO/PARA patients with metastatic disease have widespread bone metastases, which tend to be lytic. Nonetheless, PHEO/PARA bone metastases accumulate bone-seeking agents and thus are likely amenable to Ra-223 dichloride therapy [74].
21.3.1 Ra-223 Dichloride (Xofigo or Alpharadin)
Radium-223 dichloride (Ra-223) is a novel, bone-seeking calcium mimetic alpha emitter that accumulates in areas of increased bone turnover. It has been approved for the treatment of metastatic prostate cancer to the bone. Among its advantages are its availability from a long-lived actinium-227/thorium-227 generator [75] and a physical half-life of 11.4 days, which allows for easy shipping to end users with a long time between manufacturing and expiration. Similar to other alkaline earth elements such as calcium, Ra-223 is absorbed into bone matrix at sites of active mineralization [76]. The path length of the alpha particles emitted (<100 μm) is much shorter than the beta particles of Sm-153 and Sr-89, resulting in less hematologic toxicity [77].
Following the promising results in the Alpharadin in Symptomatic Prostate Cancer (ALSYMPCA) Phase III Ra-223 multinational, placebo-controlled trial in 921 subjects with prostate cancer, the FDA granted full approval in May 2013 [18, 78], with subsequent approval in the European Union. The trade name of Ra-223 dichloride is Xofigo, previously known as Alpharadin.
21.4 Pharmacokinetics and Biodistribution
Two studies have evaluated the blood pharmacokinetics of Ra-223 [79, 80]. Ra-223 clears rapidly from blood and less than 1 % of initial activity is present at 24 h. Gamma camera imaging is suboptimal but feasible, based on the gamma rays emitted in conjunction with the alpha emissions [79–81]. Elimination is mainly intestinal. Specifically, early excretion is seen into the small bowel, presumably transluminal, with a median of 52 % in bowel at 24 h and up to 2–82 % removed from the body at approximately 1 week [80]. Very little urinary excretion is noted (typically <5 %).
21.5 Clinical Studies with Ra-223
Ra-223 dosing is on an activity/kg basis. The initial Phase I study used single escalating doses ranging from 46 to 250 kBq/kg [79]. Several Phase II trials used single injections of ≤ 200 kBq/kg or cumulative activity of up to 240 kBq/kg, with demonstrated benefit and limited toxicity. The clinically approved dose is 50 kBq/kg with repeat injections × 6, based on the ALSYMPCA Phase III trial [78]. It should be noted that recently and subsequent to these studies National Institutes of Standards (NIST) have identified that the previous calibration for Ra-223 was 10 % low indicating that the 50kBq really represents 55kBq, thus the new treatment dose corresponds to 55kBq/kg.
Clinical trials with Ra-223 have consistently shown a decrease in alkaline phosphatase [79, 80, 82]. Markers of bone turnover such as uNTX-1, CTX-1, and procollagen I N-terminal propeptide also have shown significant decreases in patients treated with Ra-223 vs. control [19, 82]. Variable changes in PSA have been observed; in patients treated with four doses of Ra-223 at 50 kBq/kg every 4 weeks, a 23.8 % PSA drop was noted, compared to a 44.9 % increase in the placebo control group. Furthermore, the median time to PSA progression was 26 weeks vs. 8 weeks in the control group [82]. A dose response was seen in patients receiving three injections of 25, 50, or 80 kBq/kg, with ≥ 30 % drop in PSA found in 5 %, 17 %, and 26 % of the patients treated, respectively [83]. Note that a consistent drop in PSA was not observed in the ALSYMPCA trial.
A more important outcome is a decrease in SREs, including pain, a significant problem in patients with prostate cancer. In a group of 64 patients, with XRT plus 50 kBq/kg × 4 doses of Ra-223 compared to XRT plus placebo, the median time for the first SRE was 14 weeks in the Ra-223 group compared to 11 weeks in the placebo group [82]. In the ALSYMPCA trial, the time to first SRE was significantly delayed, with a median time to SRE of 15.6 months vs. 9.4 months for the control group and hazard ration of 0.66 [78]. With respect to pain, several of the Ra-223 clinical trials demonstrated improvement following Ra-223 [83].
Furthermore, in contrast to other bone-seeking radiopharmaceuticals, Ra-223 therapy has demonstrated significant improvements in survival. The median overall survival (OS) at 18 months was 14.9 months compared to 11.3 months for the control group [78]. A second look at the data at 24 months confirmed this effect, with overall survival of 16.3 months compared to 11.6 for the placebo group in favor of the Ra-223 arm [84]. Furthermore, at 2 years, 30 % of the Ra-223 group was alive, compared to only 13 % of the placebo group. Interestingly, patients receiving the four intended injections had longer OS than those receiving placebo (93 weeks vs. 49 weeks, respectively) [84].
21.6 Toxicity
Dosimetry estimates using standard approaches have been compared to cell-level dosimetry using a model that accounts for the expected localization of Ra-223 relative to marrow cavity architecture by Hobbs et al. [77]. These investigators estimated the percentage of cells that received a potentially toxic-absorbed dose (2 or 4 Gy). Based on their model, it was estimated that the majority of the radiation dose will be deposited near the trabecular bone, in which case increasing the activity of Ra-223 may not substantially increase the risk of marrow toxicity [77]. Organ-based dosimetry based on patient studies and utilizing the MIRD method and OLINDA software are provided in the product monograph (http://www.bayer.ca/omr/online/xofigo-pm-en-10mar2015-161312.pdf).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree