Waldenström macroglobulinemia (WM) is an IgM-secreting B-cell lymphoproliferative disorder, with strong familial predisposition. MYD88 L265P and CXCR4 WHIM mutations are common in WM and support the growth and survival of WM cells. Clinical manifestations of disease are related to both tumor cell infiltration and paraprotein production. Current treatment includes monoclonal antibodies, alkylating agents, nucleoside analogs, proteasome inhibitors, immunomodulatory drugs, and signal inhibitors. Short- and long-term toxicities should be weighed in treatment decisions with use of these agents. Elucidation of the signaling pathways involved in WM is helping to advance targeted therapeutics for WM and includes efforts directed at MYD88 and CXCR4 signaling.
Key points
- •
Waldenström macroglobulinemia (WM) is an IgM-secreting B-cell lymphoproliferative disorder, with strong familial predisposition.
- •
Clinical manifestations of disease are related to both tumor cell infiltration and paraprotein production.
- •
Current treatment options include monoclonal antibodies, alkylating agents, nucleoside analogs, proteasome inhibitors, immunomodulatory drugs, and signal inhibitors.
- •
Both short- and long-term toxicities should be weighed in treatment decisions with use of these agents.
- •
Elucidation of the signaling pathways involved in WM are helping to advance targeted therapeutics for WM and include efforts directed at MYD88 and CXCR4 signaling.
Introduction
WM is a distinct clinicopathologic entity resulting from the accumulation, predominantly in the bone marrow, of clonally related lymphocytes, lymphoplasmacytic cells, and plasma cells that secrete a monoclonal IgM protein. This condition is considered to correspond to the lymphoplasmacytic lymphoma (LPL) as defined by the World Health Organization classification system. Most cases of LPL are WM, with less than 5% of cases made up of IgA, IgG, and nonsecreting LPL.
Introduction
WM is a distinct clinicopathologic entity resulting from the accumulation, predominantly in the bone marrow, of clonally related lymphocytes, lymphoplasmacytic cells, and plasma cells that secrete a monoclonal IgM protein. This condition is considered to correspond to the lymphoplasmacytic lymphoma (LPL) as defined by the World Health Organization classification system. Most cases of LPL are WM, with less than 5% of cases made up of IgA, IgG, and nonsecreting LPL.
Epidemiology
WM is an uncommon disease, with a reported age-adjusted incidence rate of 3.4 per million among men and 1.7 per million among women in the United States, and a geometric increase with age. The incidence rate for WM is higher among whites, with African descendants representing only 5% of all patients. The incidence of WM may be higher for individuals of Ashkenazi Jewish descent. Genetic factors seem important to the pathogenesis of WM. A common predisposition for WM with other malignancies has been raised, with numerous reports of familiar clustering of individuals with WM alone and with other B-cell lymphoproliferative diseases. In a large single-center experience, 26% of 924 consecutive patients with WM had a first- or second-degree relative with either WM or another B-cell disorder.
Biology
Cytogenetics
Chromosome 6q deletions encompassing 6q21-25 have been observed in up to half of WM patients and at a comparable frequency among patients with and without a familial history. The presence of 6q deletions has been suggested to discern patients with WM from those with IgM monoclonal gammopathy of unknown significance (MGUS) and to have potential prognostic significance, including impact on progression-free survival (PFS) after treatment response, although other investigators have reported no prognostic significance to the presence of 6q deletions in WM. Other abnormalities by cytogenetic or fluorescence in situ hybridization analyses include deletions in 13q14, TP53, and ATM, and trisomy 4, 12, and 18. IgH rearrangements are uncommon in WM and may be helpful in discerning cases of WM from IgM myeloma, wherein IgH switch region rearrangements are a prominant feature.
Mutation in MYD88
A highly recurrent somatic mutation (MYD88 L265P) was first identified in WM patients by paired tumor/normal whole-genome sequencing and subsequently confirmed by multiple groups using Sanger sequencing and allele-specific (AS)–polymerase chain reaction (PCR) assays. MYD88 L265P is expressed in 90% to 100% of WM cases when more sensitive AS-PCR has been used using both CD19 sorted and unsorted bone marrow cells. By comparison, MYD88 L265P was absent in myeloma samples, including IgM myeloma, and was expressed in a small subset (6.5%) of marginal zone lymphoma patients, who surprisingly had many WM-related features. By PCR assays, 50% to 80% of IgM MGUS patients also express MYD88 L265P, and expression of this mutation was associated with increased risk of malignant progression. The presence of MYD88 L265P in IgM MGUS patient suggests that this somatic mutation is likely an early oncogenic driver and other mutations or copy number alterations that affect critical regulatory genes are likely to play a role in disease progression to WM.
The role of MYD88 L265P in supporting growth and survival signaling in WM cells has been addressed in several studies ( Fig. 1 ). Knockdown of MYD88 decreased survival of MYD88 L265P expressing WM cells, whereas survival was more enhanced by knock-in of MYD88 L265P versus wild-type MYD88. The discovery of a mutation in MYD88 is of significance given its role as an adaptor molecule in Toll-like receptor (TLR) and interleukin 1 receptor (IL-1R) signaling. All TLRs except TLR3 use MYD88 to facilitate their signaling. After TLR or IL-1R stimulation, MYD88 is recruited to the activated receptor complex as a homodimer, which then complexes with IRAK4 and activates IRAK1 and IRAK2. Tumor necrosis factor receptor–associated factor 6 is then activated by IRAK1, leading to nuclear factor κB (NF-κB) activation via IκBα phosphorylation. Use of inhibitors of MYD88 pathway led to decreased IRAK1 and IκBα phosphorylation as well as survival of MYD88 L265P expressing WM cells. These observations are of particular relevance to WM because NF-κB signaling is important for WM growth and survival. Recently, Yang and colleagues showed that Bruton tyrosine kinase (BTK) was also activated by MYD88 L265P. Activated BTK coimmunoprecipated with MYD88 that could be abrogated by use of the BTK kinase inhibitor and overexpression of MYD88 L265P but not MYD88 wild-type triggered BTK activation. Moreover, knockdown of MYD88 by lentiviral tranfection or use of a MYD88 homodimer inhibitor in MYD88 L265P mutated WM cells abrogated BTK activation.
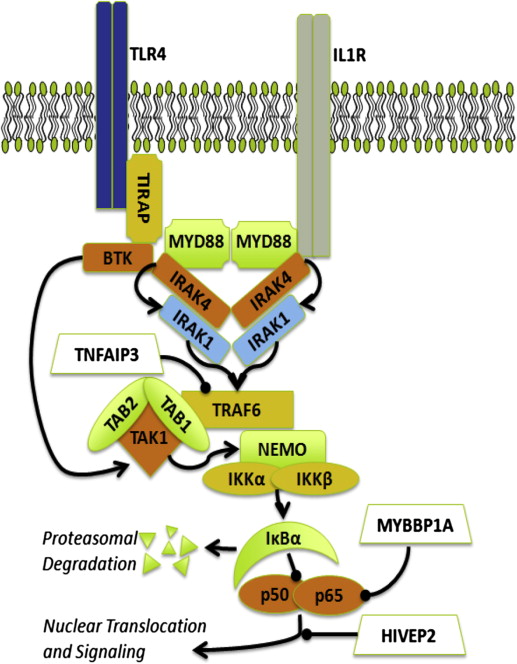
CXCR4 WHIM Mutations
Recently Hunter and colleagues identified the first ever reported somatic mutations in human cancer involving CXCR4. These mutations were present in 30% of WM patients, and involve the C-terminus that contain serine phosphorylation sites which regulate signaling of CXCR4 by its only known ligand, stromal derived factor-1a (SDF-1a) (CXCL12). The location of somatic mutations in the C-terminal domain of WM patients are similar to those observed in the germline of patients with warts, hypogammaglobulinemia, infections, and myelokathexis (WHIM) syndrome, a congenital immunodeficiency disorder characterized by chronic noncyclic neutropenia. Germline mutations in the C-terminus of CXCR4 in WHIM patients block receptor internalization after SDF-1a stimulation in myeloid cells that results in persistent CXCR4 activation and bone marrow (BM) myeloid cell trafficking.
In WM patients, 2 classes of CXCR4 mutations occur in the C-terminus. These include nonsense (CXCR4 WHIM/NS ) mutations that truncate the distal 15 to 20 amino acid region, and frameshift (CXCR4 WHIM/FS ) mutations that compromise a region of up to 40 amino acids in the C- terminal domain. Nonsense and frameshift mutations are almost equally divided among WM patients with CXCR4 somatic mutations, and more than 30 different types of CXCR4 WHIM mutations have been identified in WM patients. Preclinical studies with the most common CXCR4 WHIM/NS mutation in WM (S338X) have shown enhanced and sustained AKT, ERK, and BTK signaling after SDF-1a relative to CXCR4 WT as well increased cell migration, adhesion, growth, and survival of WM cells.
Other recurrent somatic mutations described in WM include ARID1A, TRAF3, CD79B, TP53, and MYBBP1A as well as monoallelic deletions of PRDM2, BTG1, TNFAIP3, and HIVEP2. Many of these mutations and/or losses have an impact on NF-κB regulation and may further enhance NF-κB signaling in response to MYD88 L265P (see Fig. 1 ).
Morbidity mediated by the effects of IgM
Hyperviscosity Syndrome
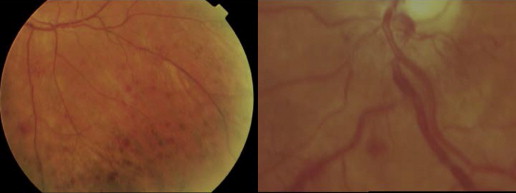
Serum hyperviscosity is effected by increased serum IgM levels leading to hyperviscosity-related complications. The mechanisms behind the marked increase in the resistance to blood flow and the resulting impaired transit through the microcirculatory system are complex. The main determinants are (1) a high concentration of monoclonal IgMs, which may form aggregates and may bind water through their carbohydrate component, and (2) their interaction with blood cells. Monoclonal IgMs increase red cell aggregation (rouleaux formation) and red cell internal viscosity while also reducing deformability. The possible presence of cryoglobulins can contribute to increasing blood viscosity as well as to the tendency to induce erythrocyte aggregation. Serum viscosity is proportional to IgM concentration up to 30 g/L, then increases sharply at higher levels. Clinical manifestations are related to circulatory disturbances that are best appreciated by ophthalmoscopy, which result in distended and tortuous retinal veins, hemorrhages, and papilledema ( Fig. 2 ). Symptoms usually occur when the monoclonal IgM concentration exceeds 50 g/L or when serum viscosity is greater than 4.0 cP, but there is a great individual variability, with some patients showing no evidence of hyperviscosity even at 10 cp. The most common symptoms are oronasal bleeding, visual disturbances due to retinal bleeding, and dizziness that may rarely lead to coma. Heart failure can be aggravated, particularly in the elderly, owing to increased blood viscosity, expanded plasma volume, and anemia. Inappropriate transfusion can exacerbate hyperviscosity and may precipitate cardiac failure.
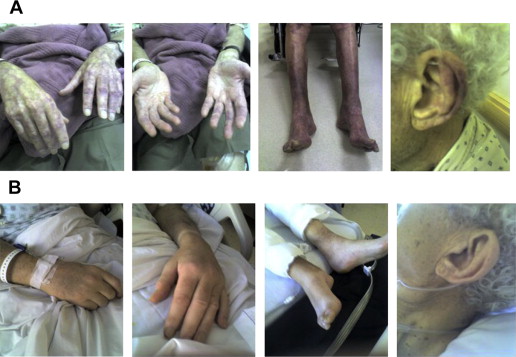
Cryoglobulinemia
In up to 20% of WM patients, the monoclonal IgM can behave as a cryoglobulin (type I), but it is symptomatic in 5% or less of the cases. Cryoprecipitation is mainly dependent on the concentration of monoclonal IgM; for this reason, plasmapheresis or plasma exchange is commonly effective in this condition. Symptoms result from impaired blood flow in small vessels and include Raynaud phenomenon; acrocyanosis; necrosis of the regions most exposed to cold, such as the tip of the nose, ears, fingers, and toes ( Fig. 3 ); malleolar ulcers; purpura; and cold urticaria. Renal manifestations may occur but are infrequent.
IgM-Related Neuropathy
The presence of peripheral neuropathy has been estimated to range from 5% to 38% in WM patients. The nerve damage is mediated by diverse pathogenetic mechanisms: IgM antibody activity toward nerve constituents causing demyelinating polyneuropathies; endoneurial granulofibrillar deposits of IgM without antibody activity, associated with axonal polyneuropathy; occasionally by tubular deposits in the endoneurium associated with IgM cryoglobulin; and, rarely, by amyloid deposits or by neoplastic cell infiltration of nerve structures. Half of patients with IgM neuropathy have a distinctive clinical syndrome that is associated with antibodies against a minor 100-kDa glycoprotein component of nerve, myelin-associated glycoprotein (MAG). Anti-MAG antibodies are generally monoclonal IgMκ, and usually also exhibit reactivity with other glycoproteins or glycolipids that share antigenic determinants with MAG. The anti-MAG–related neuropathy is typically distal and symmetric, affecting both motor and sensory functions; it is slowly progressive with a long period of stability. Most patients present with sensory complaints (paresthesias, aching discomfort, dysesthesias, or lancinating pains); imbalance and gait ataxia, owing to lack proprioception; and leg muscle atrophy in advanced stage.
Cold Agglutinin Hemolytic Anemia
Monoclonal IgM may present with cold agglutinin activity (ie, it can recognize specific red cell antigens at temperatures below physiologic, producing chronic hemolytic anemia). This disorder occurs in less than 10% of WM patients and is associated with cold agglutinin titers greater than 1:1000 in most cases. The monoclonal component is usually an IgMκ and reacts most commonly with I/i antigens, with complement fixation and activation. Mild chronic hemolytic anemia can be exacerbated after cold exposure but rarely does hemoglobin drop below 70 g/L. The hemolysis is usually extravascular (removal of C3b opsonized cells by the reticuloendotelial system, primarily in the liver) and rarely intravascular from complement destruction of red blood cell (RBC) membrane. The agglutination of RBCs in the cooler peripheral circulation also causes Raynaud syndrome, acrocyanosis, and livedo reticularis. Macroglobulins with the properties of both cryoglobulins and cold agglutinins with anti-Pr specificity have been reported. These properties may have as a common basis the immune binding of the sialic acid–containing carbohydrate present on RBC glycophorins and on Ig molecules. Several other macroglobulins with various antibody activities toward autologous antigens (phospholipids, tissue and plasma proteins and so forth) and foreign ligands have also been reported.
Amyloidosis and Other Deposition Disease
In addition to the precipitation of monoclonal IgM in tissues, as observed after cold exposure in cryoglobulinemia, the monoclonal protein or its fragments (light chains) can deposit in several organs determining their dysfunction. For instance, the monoclonal IgM can produce kidney injury characterized by a mesangiocapillary glomerulonephritis. IgM-associated AL amyloidosis is an uncommon complication of WM and presents distinctive clinical characteristics. Patients are older than non-IgM patients, have a higher frequency of lymph node involvement, and have significantly lower median proteinuria and less frequent and severe heart involvement.
Laboratory investigations and findings
Hematological Abnormalities
Anemia is the most common finding in patients with symptomatic WM and is caused by a combination of factors: myelosuppression due to bone marrow infiltration, hemolysis due to warm and cold antibodies, splenic entrapment, and hepcidin production. Leukocyte and platelet counts are usually within the reference range at presentation, although patients may occasionally present with severe thrombocytopenia. Monoclonal B-lymphocytes are uncommonly recognized by peripheral blood flow cytometry, although circulating clonal cells can be detected by AS-PCR testing for the MYD88 L265P mutation.
Biochemical Investigations
High-resolution electrophoresis combined with immunofixation of serum and urine is recommended for identification and characterization of the IgM monoclonal protein. The light chain of the monoclonal IgM is κ in 75% to 80% of patients. A few WM patients have more than one M component. The concentration of the serum monoclonal protein is variable but in most cases lies within the range of 15 to 45 g/L. Densitometry should be adopted to determine IgM levels for serial evaluations because nephelometry is unreliable and shows large intralaboratory as well as interlaboratory variation. The presence of cold agglutinins or cryoglobulins may affect determination of IgM levels and, therefore, testing for cold agglutinins and cryoglobulins should be performed at diagnosis. If present, subsequent serum samples should be analyzed under warm conditions for determination of serum monoclonal IgM level. Although Bence Jones proteinuria is frequently present, it exceeds 1 g/24 h in only 3% of cases. Although IgM levels are elevated in WM patients, IgA and IgG levels are most often depressed and do not demonstrate recovery even after successful treatment, suggesting that patients with WM harbor a defect that prevents normal plasma cell development and/or Ig heavy chain rearrangements.
Serum Viscosity
Because of its large size (approximately 1,000,000 Da), most IgM molecules are retained within the intravascular compartment and can exert an undue effect on serum viscosity. Therefore, serum viscosity should be measured if the patient has signs or symptoms of hyperviscosity syndrome. Fundoscopy remains an excellent indicator of clinically relevant hyperviscosity. Among the first clinical signs of hyperviscosity, the appearance of peripheral and midperipheral dot and blot hemorrhages in the retina, which are best appreciated with indirect ophthalmoscopy and scleral depression. In more severe cases of hyperviscosity, dot, blot, and flame-shaped hemorrhages can appear in the macular area along with markedly dilated and tortuous veins with focal constrictions resulting in venous sausaging, as well as papilledema.
Bone Marrow Findings
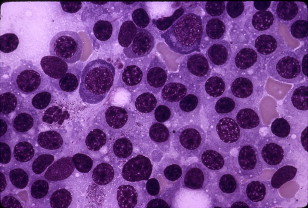
The bone marrow is always involved in WM. Central to the diagnosis of WM is the demonstration, by trephine biopsy, of bone marrow infiltration by a lymphoplasmacytic cell population constituted by small lymphocytes with evidence of plasmacytoid/plasma cell differentiation ( Fig. 4 ). The pattern of bone marrow infiltration may be diffuse, interstitial, or nodular, usually showing an intertrabecular pattern of infiltration. A solely paratrabecular pattern of infiltration is unusual and should raise the possibility of follicular lymphoma. The bone marrow infiltration should routinely be confirmed by immunophenotypic studies (flow cytometry and/or immunohistochemistry) showing the following profile: sIgM + CD19 + CD20 + CD22 + CD79 + . Up to 20% of cases may express CD5, CD10, or CD23. In these cases, care should be taken to satisfactorily exclude chronic lymphocytic leukemia and mantle cell lymphoma. Intranuclear periodic acid–Schiff–positive inclusions (Dutcher-Fahey bodies) consisting of IgM deposits in the perinuclear space and sometimes, in intranuclear vacuoles, may be seen occasionally in lymphoid cells in WM. An increased number of mast cells, usually in association with the lymphoid aggregates, are commonly found in WM, and their presence may help in differentiating WM from other B-cell lymphomas. AS-PCR testing for MYD88 L265P is particularly helpful in discerning WM from other overlapping IgM-secreting B-cell malignancies, including marginal zone lymphomas, IgM myeloma, and chronic lymphocytic leukemia, which rarely express this somatic mutation.
Prognosis and risk stratification
WM typically presents as an indolent disease though considerable variability in prognosis can be seen. Castillo and colleagues recently examined trends in relative survival (RS) and overall survival (OS) in patients with WM from the Surveillance, Epidemiology, and End Results database. A total of 6231 patients diagnosed with WM between 1980 and 2010 were included in their analysis. The median OS times were 5.6 and 7.3 years for the 1980 to 2000 and the 2001 to 2010 cohorts, respectively. The 5-year RS rates for the 1980 to 2000 and 2001 to 2010 cohorts were 67% and 78%, and the 5-year OS rates were 56% and 65%, respectively. In the multivariate analysis, survival benefits were identified for the 2001 to 2010 cohort in almost every stratum analyzed, with exception of patients less than 40 years and greater than or equal to 80 years. These results are consistent with a 1555-patient Swedish population-based study that also showed that outcome of patients with WM has improved over the period 1980 to 2005.
Age has been consistently an important prognostic factor (>60–70 years), although unrelated morbidities often have an impact. Anemia, which can be multifactorial, is an adverse prognostic factor in WM, with hemoglobin levels of less than 9 to 12 g/dL associated with decreased survival in several series. Cytopenias have also been regularly identified as a significant predictor of survival. The number of cytopenias in a given patient may predict survival. Serum albumin levels have correlated with survival in WM patients in certain but not all studies using multivariate analyses. High-serum β 2 -microglobulin (>3–3.5 g/dL) levels, high-serum IgM M-protein (>7 g/dL), low-serum IgM M-protein (<4 g/dL), the presence of cryoglobulins, and the presence of a familial disease background have also been reported to confer adverse outcomes. The presence of 6q deletion as an adverse marker remains controversial. In one study, the presence of MYD88 wild-type correlated with poor survival, whereas the presence of nonsense CXCR4 WHIM mutations was associated with more aggressive presentation but did not have an impact on survival relative to those patients with wild-type CXCR4.
There have been several attempts to organize prognostic factors into prognostic systems for the risk stratification of patients with WM and as a tool for the comparison between studies. A multicenter collaborative project analyzed a large number of previously untreated, symptomatic patients and 5 adverse covariates (age >65 years, hemoglobin ≤11.5 g/dL, platelet count ≤100 × 109/L, β 2 -microglobulin >3 mg/L, and serum monoclonal protein concentration >70 g/L) was used to define 3 risk groups (low-, intermediate-, and high-risk, respectively). This International Prognostic Scoring System for WM (ISSWM) has been externally validated in independent cohorts. Although results per ISSWM risk category are increasingly reported in phase II studies and are used for the stratification of patients in randomized clinical trials, its use in making treatment decisions remains to be delineated.
Treatment of Waldenström macroglobulinemia
Treatment Indications
Consensus guidelines on indications for treatment intitiation were formulated as part of the Second International Workshop on Waldenström’s Macroglobulinemia. Initiation of therapy should not be based only on the IgM levels because they may not correlate with either disease burden nor symptomatic status. Initiation of therapy is appropriate for patients with constitutional symptoms, such as recurrent fever, night sweats, fatigue due to anemia, or weight loss. The presence of progressive, symptomatic lymphadenopathy or splenomegaly provides additional reasons to begin therapy. The presence of anemia with a hemoglobin value of less than or equal to 10 g/dL or a platelet count less than or equal to 100 × 10 9 /L on this basis of disease is also a reasonable indication for treatment initiation. Certain complications of WM, such as hyperviscosity syndrome, symptomatic sensorimotor peripheral neuropathy, systemic amyloidosis, renal insufficiency, and symptomatic cryoglobulinemia, are also indications for therapy.
Treatment Options
A precise therapeutic algorithm for therapy for WM remains to be defined given the paucity of randomized clinical trials. Active agents include monoclonal antibodies (rituximab and ofatumumab), alkylators (chlorambucil, cyclophosphamide, and bendamustine), nucleoside analogs (cladribine and fludarabine), proteasome inhibitors (bortezomib and carfilzomib), immunomodulatory drugs (thalidomide, lenalidomide, and pomalidomide), and signal inhibitors (everolimus and ibrutinib). Combination regimens particularly with rituximab commonly have been used to treat WM. Individual patient considerations, including the presence of cytopenias, need for more rapid disease control, age, and candidacy for autologous transplant therapy, should be taken into account in making the choice of a first-line agent. For patients who are candidates for autologous transplant therapy, exposure to continuous chlorambucil or nucleoside analog therapy should be limited given potential for stem cell damage. The use of nucleoside analogs may also increase risk for histologic transformation to diffuse large B-cell lymphoma as well as myelodysplasia and acute myelogenous leukemia.
Monoclonal antibodies
Rituximab is a chimeric monoclonal antibody that targets CD20, a widely expressed antigen on lymphoplasmacytic cells in WM. The use of rituximab at standard dosimetry (ie, 4 weekly infusions at 375 mg/m 2 ) induces partial or better responses in approximately 27% to 35% of previously treated and untreated patients. Patients who achieved even minor responses (MRs), however, benefited from rituximab as evidenced by improved hemoglobin and platelet counts and reduction of lymphadenopathy and/or splenomegaly. The median time to treatment failure in these studies was found to range from 8 to 27+ months. Studies evaluating an extended rituximab schedule consisting of 4 weekly courses at 375 mg/m 2 /wk, repeated 3 months later by another 4-week course, have demonstrated higher major response rates of 44% to 48%, with time to progression estimates exceeding 29 months.
In many WM patients, a transient increase of serum IgM (IgM flare) may be noted immediately after initiation of rituximab treatment. The IgM flare in response to rituximab does not herald treatment failure, and although most patients return to their baseline serum IgM level by 12 weeks, some patients may flare for months despite having tumor responses in their bone marrow. Patients with baseline serum IgM levels of greater than 40 g/dL or serum viscosity of greater than 3.5 cp may be particularly at risk for a hyperviscosity-related event and in such patients plasmapheresis should be considered or rituximab omitted for the first few cycles of therapy until IgM levels decline to safer levels. Because of the decreased likelihood of response in patients with higher IgM levels as well as the possibility that serum IgM and viscosity levels may abruptly rise, rituximab monotherapy should not be used as sole therapy for the treatment of patients at risk for hyperviscosity symptoms. Intolerance to rituximab is common in WM patients, with an estimated frequency of 10%. Ofatumumab is a fully humanized CD20-directed monoclonal antibody that targets the small loop of CD20, an epitope that is different from rituximab. An overall response rate (ORR) of 59% was observed in a series of 37 symptomatic WM patients after ofatumumab administration, which included untreated and previously treated patients. Responses were higher among rituximab-naïve patients. An IgM flare with symptomatic hyperviscosity was also observed in 2 (5%) patients in this series who required plasmapheresis. Ofatumumab has also been successfully administered to WM patients who demonstrated intolerance to rituximab.
Alkylating agents
Oral alkylating drugs, alone and in combination therapy with steroids, have been extensively evaluated in the upfront treatment of WM. The greatest experience with oral alkylator therapy has been with chlorambucil, which has been administered on both a continuous (ie, daily dose) as well as an intermittent schedule. Patients receiving chlorambucil on a continuous schedule typically receive 0.1 mg/kg per day, whereas on the intermittent schedule patients typically receive 0.3 mg/kg for 7 days, every 6 weeks. In a prospective randomized study, Kyle and colleagues reported no significant difference in the ORR between these schedules, although the median response duration was greater for patients receiving intermittent versus continuously dosed chlorambucil (46 vs 26 months). Despite the favorable median response duration in this study for use of the intermittent schedule, no difference in the median OS was observed. Moreover, an increased incidence for development of myelodysplasia and acute myelogenous leukemia with the intermittent (3 of 22 patients) versus the continuous (0 of 24 patients) chlorambucil schedule prompted the investigators of this study to express preference for use of continuous chlorambucil dosing. In a study of 414 patients, including 339 with WM, 37 with non–mucosa-associated lymphoid tissue marginal zone lymphoma, and 38 with LPL, patients were randomized to receive chlorambucil or fludarabine. On the basis of intent-to-treat analysis, the ORR was 47.8% in the fludarabine arm versus 38.6% in the chlorambucil arm. With a median follow-up of 36 months, median PFS was significantly higher in those patients who received fludarabine (36.3 months) compared with chlorambucil arm (27.1 months). Moreover, in patients with WM, median OS was not reached in the fludarabine arm versus 69.8 months in the chlorambucil arm. Grade 3 to 4 neutropenia was significantly higher among patients treated with fludarabine (36%) compared with patients treated with chlorambucil (18%). However, secondary malignancies were significantly more frequent with chlorambucil versus fludarabine, with a 6-year cumulative incidence rate of 21% and 4%, respectively. Additional factors to be taken into account in considering chlorambucil therapy for patients with WM include necessity for more rapid disease control given the slow nature of response to this agent as well as consideration for preserving stem cells in patients who are candidates for autologous transplant therapy. Chlorambucil should, therefore, be reserved for nontransplant candidates with more indolent disease.
The combination of cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) with rituximab (CHOP-R) was investigated in a randomized frontline study by the German Low Grade Lymphoma Study Group involving 69 patients, most of whom had WM. The addition of rituximab to CHOP resulted in a higher ORR (94% vs 67%) and median time to progression (63 vs 22 months) in comparison to patients treated with CHOP alone. Dimopoulos and colleagues investigated the combination of rituximab, dexamethasone, and oral cyclophosphamide as primary therapy in 72 patients with WM. At least a major response was observed in 74% of patients in this study, and the 2-year PFS was 67%. Therapy was well tolerated, although 1 patient died of interstitial pneumonia. In the salvage setting, the use of CHOP-R has been investigated in relapsed/refractory WM patients. Among 13 evaluable patients, 10 patients achieved a major response (77%), including 3 CR and 7 PR, and 2 patients achieved an MR. In a retrospective study, Ioakimidis and colleagues examined the outcomes of symptomatic WM patients who received CHOP-R, Cyclophosphamide, Vincristine, Prednisone, Rituximab (CVP-R), or Cyclophosphamide, Prednisone, Rituximab (CP-R). Baseline characteristics for all 3 cohorts were similar for age, prior therapies, bone marrow involvement, hematocrit, platelet count, and serum β 2 -microglobulin, although serum IgM levels were higher in patients treated with CHOP-R. The ORRs to therapy were comparable for all 3 treatments: CHOP-R (96%), CVP-R (88%), and CP-R (95%), although more CRs were observed among patients treated with either CVP-R or CHOP-R. Comparison of adverse events for these regimens showed a higher incidence for neutropenic fever as well as treatment-related neuropathy in patients receiving CHOP-R and CVP-R versus CPR. These studies suggest that in WM, the use of doxorubicin and vincristine may be omitted to minimize treatment-related complications. Therefore, more intense cyclophosphamide-based regimens, such as CHOP-R or CVP-R, should be avoided.
Bendamustine is a recently approved agent for the treatment of relapsed/refractory indolent non-Hodgkin lymphoma. Bendamustine has structural similarities to both alkylating agents and purine analogs. The use of bendamustine in combination with rituximab was explored by Rummel and colleagues in the frontline therapy for WM. As part of a randomized study, patients received 6 cycles of bendamustine plus rituximab (Benda-R) or CHOP-R. A total of 546 patients were enrolled in this study for indolent non-Hodgkin lymphoma patients and included 40 patients with WM. Patients in the Benda-R arm received bendamustine at 90 mg/m 2 on days 1 and 2 and rituximab at 375 mg/m 2 on day 1 with the frequency of 4 weeks for each cycle. The ORR was 96% for Benda-R and 94% for CHOP-R treated patients. With a median observation period of 26 months, 20/23 (87%) Benda-R versus 9/17 (53%) CHOP-R–treated WM patients remain free of progression. Benda-R was associated with a lower incidence of grade 3 or 4 neutropenia, infectious complications, and alopecia. In the salvage setting, the outcome of 30 WM patients with relapsed/refractory disease who received bendamustine alone or with a CD20-directed antibody showed an ORR of 83.3% and a median PFS of 13.2 months. Overall, therapy was well tolerated in this study although prolonged myelosuppression occurred in patients who received prior nucleoside analog therapy. Cytoreduction, including extramedullary disease, is particulary good with bendamustine-based therapy and can be considered in patients presenting with bulky adenopathy or splenomegaly or who have other symptomatic extramedullary disease.
Nucleoside analogs
Both cladribine and fludarabine have been evaluated in untreated as well as previously treated WM patients. Cladribine administered as a single agent by continuous intravenous (IV) infusion, by 2-hour daily infusion or by subcutaneous bolus injections for 5 to 7 days has resulted in major responses in 40% to 90% of patients who received primary therapy, whereas in the salvage setting responses have ranged from 38% to 54%. The ORR with daily infusional fludarabine therapy administered mainly on 5-day schedules in previously untreated and treated WM patients has ranged from 38% to 100% and 30% to 40%, respectively, on par with the response data for single-agent cladribine. In general, response rates and durations of responses have been greater for patients receiving nucleoside analogs as first-line agents, although in one large study that included both untreated and previously treated patients, no substantial difference in the overall response to fludarabine was observed. Myelosuppression commonly occurred after prolonged exposure to either of the nucleoside analogs, as did lymphopenia with sustained depletion of both CD4 + and CD8 + T lymphocytes observed in WM patients 1 year after initiation of therapy. Treatment-related mortality due to myelosuppression and/or opportunistic infections attributable to immunosuppression occurred in up to 5% of all treated patients in some series with either nucleoside analog.
Combination therapy with nucleoside analogs has been investigated as both first-line and salvage therapy in WM. Weber and colleagues administered rituximab along with cladribine and cyclophosphamide to 17 previously untreated patients with WM. At least a partial response was documented in 94% of WM patients, including a complete response (CR) in 18%. With a median follow-up of 21 months, no patient has relapsed. Laszlo and colleagues evaluated the combination of subcutaneous cladribine with rituximab in 29 WM patients with either untreated or previously treated disease. Intended therapy consisted of rituximab on day 1 followed by subcutaneous cladribine, 0.1 mg/kg, for 5 consecutive days, administered monthly for 4 cycles. With a median follow-up of 43 months, the ORR observed was 89.6%, with 7 CRs, 16 partial responses, and 3 MRs. Response activity was similar between untreated and previously treated patients. No major infections were observed despite the lack of antimicrobial prophylaxis. In a study by the Waldenström’s Macroglobulinemia Clinical Trials Group (WMCTG), a combination of rituximab and fludarabine was administered to 43 WM patients, 32 (75%) of whom were previously untreated. The ORR was 95.3%, and 83% of patients achieved a major response. The median time to progression was 51.2 months in this series and was longer for those patients who were previously untreated and for those achieving at least a very good partial response (VGPR). Hematological toxicity was common, in particular neutropenia and thrombocytopenia. Two deaths occurred in this study due to non– Pneumocystis carinii pneumonia. Secondary malignanices, including transformation to aggressive lymphoma and development of myelodysplasia or acute myeloid leukemia, were observed in 6 patients in this series. The addition of rituximab to fludarabine and cyclophosphamide has also been explored in the salvage setting by Tam and colleagues wherein 4 of 5 patients demonstrated a response. Hensel and colleagues administered rituximab along with pentostatin and cyclophosphamide to 13 patients with untreated and previously treated WM or LPL. A major response was observed in 77% or patients. The addition of alkylating agents to nucleoside analogs has also been explored in WM. Dimopoulos and colleagues examined fludarabine in combination with IV cyclophosphamide and observed partial responses in 6 of 11 (55%) patients with primary refractory disease or who relapsed on treatment. The combination of fludarabine plus cyclosphosphamide was also evaluated in a recent study by Tamburini and colleagues involving 49 patients, 35 of whom were previously treated. Seventy-eight percent of the patients in this study achieved a response and median time to treatment failure was 27 months. Hematological toxicity was commonly observed and 3 patients died of treatment-related toxicities. Findings in this study included the development of acute leukemia in 2 patients, histologic transformation to diffuse large cell lymphoma in 1 patient, 2 cases of solid malignancies (prostate and melanoma), and failure to mobilize stem cells in 4 of 6 patients. Tedeschi and colleagues recently completed a multicenter study with fludarabine, cyclophosphamide, and rituximab in symptomatic WM patients with untreated or relapsed/refractory disease to 1 line of chemotherapy. Treatment consisted of rituximab at 375 mg/m 2 on day 1, fludarabine at 25 mg/m 2 , and cyclophosphamide at 250 mg/m 2 by IV administration on days 2 to 4 every 4 weeks. Forty-three patients were accrued to this study. The ORR was 89%, with 83% of patients attaining a major remission and 14% a CR. Prolonged neutropenia was observed in up to one-third of patients. With a median follow-up of 15 months, the median PFS for this study has not been reached.
The safety of nucleoside analogs has been the subject of investigation in several recent studies. Thomas and colleagues recently reported their experiences in harvesting stem cells in 21 patients with symptomatic WM in whom autologous peripheral blood stem cell collection was attempted. Autologous stem cell collection succeeded on the first attempt in 14 of 15 patients who received non-nucleoside analog-based therapy versus 2 of 6 patients who received a nucleoside analog. The long-term safety of nucleoside analogs in WM was recently examined by Leleu and colleagues in a large series of WM patients. A 7-fold increase in transformation to an aggressive lymphoma and a 3-fold increase in the development of acute myelogenous leukemia/myelodysplasia were observed among patients who received a nucleoside analog versus other therapies for their WM. A recent metanalysis by Leleu and colleagues of several trials using nucleoside analogs in WM patients, which included patients who had previously received an alkylator agent, showed a crude incidence of 6.6% to 10% for development of disease transformation and 1.4% to 8.9% for development of myelodysplasia or acute myelogenous leukemia. None of the studied risk factors (ie, gender, age, family history of WM or B-cell malignancies, typical markers of tumor burden and prognosis, type of nucleoside analog therapy [cladribine vs fludarabine], time from diagnosis to nucleoside analog use, nucleoside analog treatment as primary or salvage therapy, and treatment with an oral alkylator [ie, chlorambucil]) predicted for the occurrence of transformation or development of myelodysplasia/acute myelogenous leukemia for WM patients treated with a nucleoside analog. Nucleoside analogs should, therefore, be avoided in younger patients given the constellation of short- as well as long-term toxicity risks.
Proteasome inhibitors
Bortezomib has been extensively investigated in WM. In a multicenter study of the WMCTG, 27 patients received up to 8 cycles of bortezomib at 1.3 mg/m 2 on days 1, 4, 8, and 11. All but 1 patient had relapsed/or refractory disease. After therapy, median serum IgM levels declined from 4660 mg/dL to 2092 mg/dL ( P <.0001). The ORR was 85%, with 10 and 13 patients achieving a MR and major response, respectively. Responses were prompt and occurred at median of 1.4 months. The median time to progression for all responding patients in this study was 7.9 months, and the most common grade III/IV toxicities occurring in greater than or equal to 5% of patients were sensory neuropathies (22.2%), leukopenia (18.5%), neutropenia (14.8%), dizziness (11.1%), and thrombocytopenia (7.4%). Importantly, sensory neuropathies resolved or improved in nearly all patients after cessation of therapy. As part of a National Cancer Institute of Canada study, Chen and colleagues treated 27 patients with both untreated (44%) and previously treated (56%) disease. Patients in this study received bortezomib using the standard schedule until they demonstrated progressive disease or 2 cycles beyond a CR or stable disease. The ORR in this study was 78%, with major responses observed in 44% of patients. Sensory neuropathy occurred in 20 patients, 5 with grade greater than 3, and occurred after 2 to 4 cycles of therapy. Among the 20 patients developing a neuropathy, 14 patients resolved and 1 patient demonstrated a 1-grade improvement at 2 to 13 months. In addition to these experiences with bortezomib monotherapy in WM, Dimopoulos and colleagues observed major responses in 6 of 10 (60%) previously treated WM patients. The combination of bortezomib, dexamethasone, and rituximab (BDR) has been investigated as primary therapy in WM patients by the WMCTG. An ORR of 96%, major response rate of 83%, and complete attainment in 22% was observed with BDR. The updated median PFS in this study was greater than 56.1 months. The incidence of grade 3 neuropathy was 30% in this study, which used a twice-a-week schedule for bortezomib administration at 1.3 mg/m 2 . Peripheral neuropathy from bortezomib was reversible in most patients in this study after discontinuation of therapy, and patients benefitted from pregabalin. An increased incidence of herpes zoster was also observed with BDR, prompting the use of prophylactic antiviral therapy. An alternative schedule for bortezomib administration (ie, weekly at 1.6 mg/m 2 ) in combination with rituximab and/or dexamethasone has been investigated in several studies with ORRs of 80% to 90%. A lower incidence of peripheral neuropathy was observed in studies with weekly dosed bortzomib. In a hybrid study by the European Myeloma Network, patients received twice-weekly bortezomib for the first cycle, then weekly bortezomib for cycles 2 to 5 with dexamethasone and rituximab. An ORR of 85%, with VGPR or better in 10% of patients, was observed. The median PFS was 43 months, and patients with VGPR/CR had longer PFS. Grade greater than or equal to 2 treatment–related peripheral neuropathy occurred in 24% of patients and led to bortezomib discontinuation in 8% of patients.
Because bortezomib frequently produces severe treatment-related peripheral neuropathy in WM, the WMCTG investigated the use of carfilzomib, a neuropathy-sparing proteasome-inhibitor, in combination with rituximab and dexamethasone (CaRD) in symptomatic WM patients naïve to bortezomib and rituximab. Protocol therapy consisted of IV carfilzomib 20 mg/m 2 (cycle 1); 36 mg/m 2 (cycles 2–6) with IV dexamethasone 20 mg on days 1, 2, 8, 9; and rituximab 375 mg/m 2 on days 2 and 9 every 21 days. Maintenance therapy followed 8 weeks later with IV carfilzomib 36 mg/m 2 and IV dexamethasone 20 mg on days 1 and 2 and rituximab 375 mg/m 2 on day 2 every 8 weeks for 8 cycles. Post-therapy, median serum IgM levels declined from 3375 to 749 mg/dL ( P <.0001); bone marrow disease declined from 60% to 5% ( P <.0001); hematocrit rose from 32.3% to 41.3% ( P <.0001). ORR was 87.1% (1 CR, 10 VGPRs, 10 PRs, and 6 MRs) and was not impacted by MYD88 L265P or CXCR4 WHIM mutation status. With a median follow-up of 15.4 months, 20 patients remain progression-free, including 8 on maintenance-therapy. Grade greater than or equal to 2 toxicities included asymptomatic hyperlipasemia (41.9%), reversible neutropenia (12.9%), and cardiomyopathy in 1 patient (3.2%) with multiple risk factors. Declines in serum IgA and IgG were common and were associated with recurring infections necessitating IVIG therapy in several patients.
Immunomodulatory agents
Thalidomide as monotherapy and in combination with dexamethasone and/or clarithromycin has been examined in WM. Dimopoulos and colleagues demonstrated a major response in 5 of 20 (25%) previously untreated and treated patients who received single-agent thalidomide. Dose escalation from the thalidomide start dose of 200 mg daily was hindered by development of side effects, including the development of peripheral neuropathy in 5 patients, obligating discontinuation or dose reduction. Low doses of thalidomide (50 mg orally daily) in combination with dexamethasone (40 mg orally once a week) and clarithromycin (250 mg orally twice a day) have also been examined, with 10 of 12 (83%) previously treated patients demonstrating at least a major response. In a follow-up study by Dimopoulos and colleagues, however, using a higher thalidomide dose (200 mg orally daily) along with dexamethasone (40 g orally once a week) and clarithromycin (500 mg orally twice a day), only 2 of 10 (20%) previously treated patients responded. The combination of immunomodulator agents (thalidomide, lenalidomide, and pomalidomide) with rituximab was investigated by the WMCTG. Thalidomide was administered at 200 mg daily for 2 weeks, followed by 400 mg daily and thereafter for 1 year. Patients received 4 weekly infusions of rituximab at 375 mg/m 2 beginning 1 week after initiation of thalidomide, followed by 4 additional weekly infusions of rituximab at 375 mg/m 2 beginning at week 13. The ORR and major response rate were 72% and 64%, respectively, and the median time to progression was 38 months in this series. Dose reduction and/or discontinuation of thalidomide was common and mainly attributed to treatment-related neuropathy. The investigators concluded in this study that lower doses of thalidomide (ie, 50–100 mg/d) should be considered in this patient population. The combination of lenalidomide with rituximab was investigated by the WMCTG using lenalidomide at 25 mg daily on a syncopated schedule wherein therapy was administered for 3 weeks, followed by a 1-week pause for an intended duration of 48 weeks. Patients received 1 week of therapy with lenalidomide, after which rituximab (375 mg/m 2 ) was administered weekly on weeks 2 through 5, then 13 through 16. The ORR and a major response rates in this study were 50% and 25%, respectively, and a median time to progression for responders was 18.9 months. In 2 patients with bulky disease, significant reduction in extramedullary disease was observed. An acute decrease in hematocrit was observed, however, during first 2 weeks of lenalidomide therapy in 13 of 16 (81%) patients, with a median absolute decrease in hematocrit of 4.8%, resulting in anemia-related complications and hospitalizations in 4 patients. Despite dose reduction, most patients in this study continued to demonstrate aggravated anemia with lenalidomide. There was no evidence of hemolysis or more general myelosuppression with lenalidomide in this study. Pomalidomide was recently investigated in a dose-escalating phase 1 study with rituximab. Patients showed intolerance at doses above 1 mg daily, and rituximab flaring led to symptomatic hyperiscosity and emergent plasmapheresis in 3 of 7 patients. The ORR in this study was 43% with median response duration of 15 months.
Everolimus
Everolimus is an oral inhibitor of the Akt-mTOR pathway. Inhibition of this pathway leads to apoptosis of primary WM cells and WM cell lines. Sixty patients with a median of 3 prior therapies were treated with everolimus in a joint Dana-Farber/Mayo Clinic study. The ORR was 73%, with 50% of patients attaining a major response. The median PFS in this study was 21 months. Grade 3 or higher related toxicities were observed in 67% of patients, with cytopenias constituting the most common toxicity. Pulmonary toxicity occurred in 5% of patients, and dose reductions due to toxicity occurred in 52% of patients. A clinical trial examining the activity of everolimus in 33 previously untreated patients with WM was recently reported by the WMCTG that included serial bone marrow biopsies in response assessment. The ORR in this study was 72%, including partial or better responses in 60% of patients. Discordance between serum IgM levels, on which consensus criteria for response are based, and BM disease response were common and complicated response assessment. At 6-month assessments, in 10 of 22 (45.5%) patients for whom both serum IgM and BM assessments were performed, discordance between serum IgM and BM disease involvement were observed. Among these patients, 2 had no change and 8 had increased bone marrow disease involvement despite decreases in serum IgM levels. Grade greater than or equal to 2 hematologic and nonhematologic toxicities related to everolimus were predominately hematological, including anemia (39.4%), thrombocytopenia (12%), and neutropenia (18.2%). Nonhematological toxicities included oral ulcerations (27.3%), which improved with oral dexamethasone swish and spit solution, and pneumonitis (15%), the latter leading to treatment discontinuation in 5 patients.
Ibrutinib
BTK is a target of MYD88 L265P mutation, which leads to its activation. Furthermore, ibrutinib inhibits BTK and in vitro induces apoptosis of WM cells bearing MYD88 L265P. Given these preclinical findings, symptomatic WM patients who received at least 1 prior treatment were enrolled on a prospective clinical trial examining the safety and efficacy of daily dosed ibrutinib. Intended therapy consisted of 420 mg of oral ibrutinib daily for 2 years or until progression or unacceptable toxicity. Sixty-three patients, including 17 with refractory disease, were enrolled. With a short median follow-up of 6 cycles, the best ORR was 81% (4 VGPR, 32 PR, and 15 MR), with a major response rate (PR or better) of 57.1% and a median time to response of 4 weeks. Grade greater than 2 treatment-related toxicities include thrombocytopenia (14.3%) and neutropenia (19.1%), which occurred mainly in heavily pretreated patients. Responses were impacted by mutations in CXCR4 in those patients who underwent tumor genotypic. The major response rate was 77% for patients with wild-type CXCR4 versus 30% in those with WHIM-like CXCR4 mutations. Decreases in serum IgM as well as improvements in hemoglobin were also greater in patients with wild-type CXCR4. Patients with wild-type CXCR4 also had increased peripheral lymphocytosis after ibrutinib treatment versus those with WHIM-like CXCR4 mutations.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree