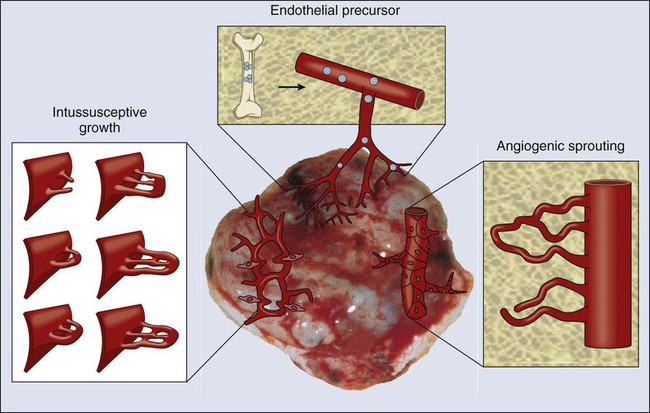
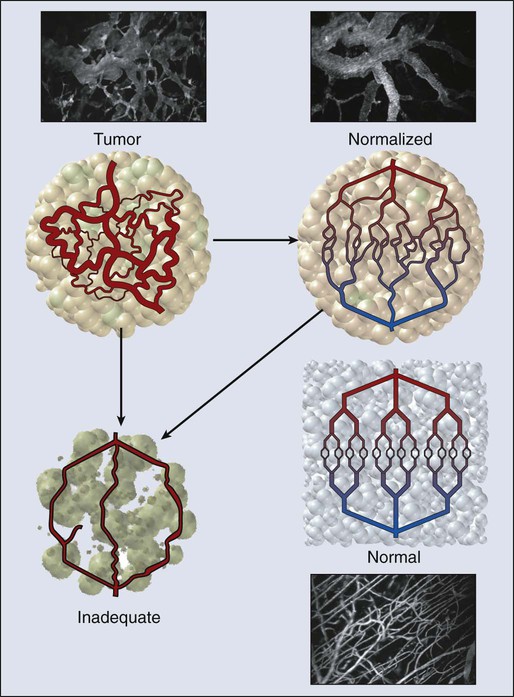
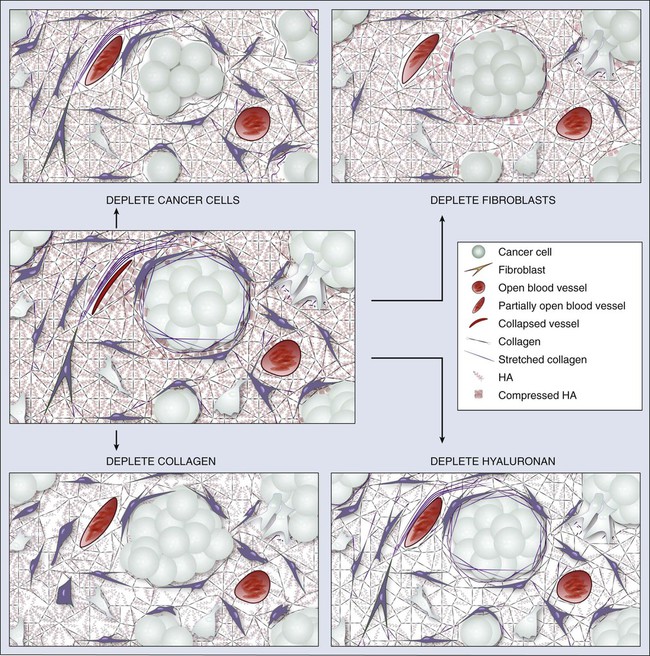
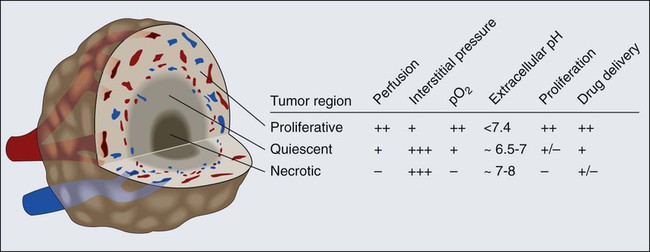
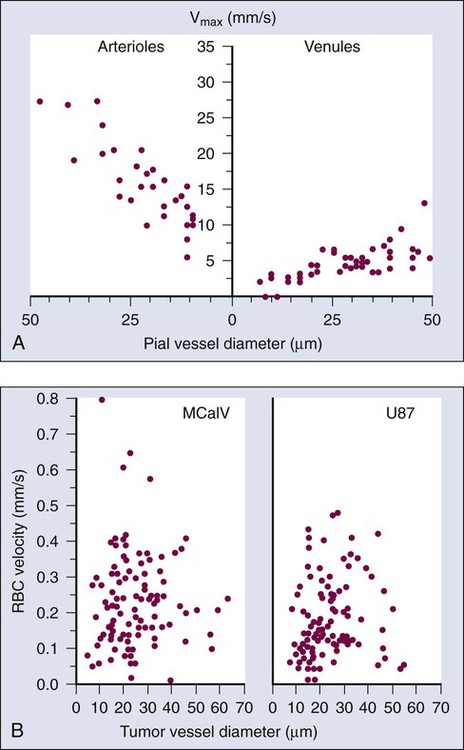
Rakesh K. Jain, Vikash P. Chauhan and Dan G. Duda • A solid tumor is an organ composed of neoplastic cells and stromal cells nourished by a vasculature made of endothelial cells—all embedded in an extracellular matrix. The interactions among these cells and between these cells, their surrounding matrix, and their local microenvironment control the expression of various genes. The products encoded by these genes, in turn, control the pathophysiological characteristics of the tumor. Tumor pathophysiology governs not only tumor growth, invasion, and metastasis but also the response to various therapies. • Tumor vasculature is made of host vessels co-opted by cancer cells and by new vessels formed by the processes of vasculogenesis and angiogenesis. A constellation of positive and negative regulators of angiogenesis governs the process of neovascularization. • Tumor vessels are abnormal in terms of their organization, structure, and function. These abnormalities contribute to heterogeneity in vascular permeability, blood flow, and the microenvironment. • Tumor interstitial matrix is formed of proteins secreted by stromal and cancer cells and by those leaked from the nascent blood vessels. • Tumor interstitium is heterogeneous, with some regions fairly permeable and others difficult to penetrate. Modification of collagen and hyaluronan in the matrix can improve penetration of large-molecular-weight therapeutics. • Solid components of tumors—cancer cells, stromal cells, and matrix molecules—mechanically compress blood and lymphatic vessels, resulting in reduced perfusion that limits oxygen and drug supply. Depleting these constituents decompresses blood vessels to enhance perfusion and drug delivery. • Interstitial hypertension is a hallmark of solid tumors and results from vessel leakiness, lack of functional lymphatics, and compression of vessels. This elevated fluid pressure contributes to blood flow heterogeneity and directly hinders the penetration of large-molecular-weight therapeutics. Alleviating interstitial hypertension improves oxygen and drug delivery to tumors. • Judicious application of angiogenic therapy can normalize the tumor vessels and make them more efficient for delivery of oxygen (a known radiosensitizer) and drugs. Antiangiogenic agents can prune tumor vessels, induce cancer cell apoptosis, and lower interstitial hypertension in tumors. • Thus far, eight antiangiogenic agents have been approved for patients with certain types of cancer. Based on these successes, antiangiogenic therapy is expected to make a difference in many other tumor types. Two main hurdles to further development of antiangiogenic agents are the better understanding of the mechanisms of action of these agents and the development of biomarkers to select patients for these drugs and to predict and monitor their effects. A solid tumor is an organ composed of neoplastic cells and host stromal cells nourished by a vasculature made of endothelial cells—all embedded in an extracellular matrix (Fig. 8-1). The interactions among these cells and between these cells, their surrounding matrix, and their local microenvironment control the expression of various genes. The products encoded by these genes, in turn, control the pathophysiological characteristics of the tumor. Tumor pathophysiology governs not only tumor growth, invasion, and metastasis but also the response to various therapies. In this chapter we will discuss various pathophysiological parameters that characterize the vascular and extravascular compartments of a tumor and the mechanisms governing the formation and function of these compartments. It has been known for nearly a century that the vascular system is associated with tumor growth in animals and humans.1 Powerful insights into the neovascularization of transplanted tumors using transparent window techniques were developed in the 1940s.2–5 The possibility that tumors produce an “angiogenic” substance was suggested in 1968.6 The hypothesis that blocking angiogenesis should block tumor growth and metastasis was proposed shortly thereafter in 1971.7 The concept that a tissue acquires angiogenic capacity during neoplastic transformation—and, by extension, that antiangiogenesis could be used to prevent cancer—was put forward in 1978.8 The first antiangiogenic agent approved for patients with cancer was bevacizumab, an antibody specific to vascular endothelial growth factor (VEGF), on the basis of the increased survival seen in patients with metastatic colorectal cancer with the combination of bevacizumab and standard chemotherapy in a pivotal randomized placebo-controlled phase III trial.9 At present, various antiangiogenesis and proangiogenesis strategies are being evaluated clinically to prevent or treat a large number of diseases, including cancer.12–12 Both normal and pathological angiogenic processes are governed by the net balance between proangiogenic and antiangiogenic factors.13,14 This balance is spatially and temporally regulated under physiological conditions, so that the “angiogenic switch” is “on” when needed (e.g., during embryonic development, wound healing, and formation of the corpus luteum) and “off” at other times. During neoplastic transformation and tumor progression, this regulation is deranged, and blood vessels form ectopically to support a growing tumor mass. At least four cellular mechanisms are involved in the vascularization of tumors: co-option, intussusception, sprouting (angiogenesis), and vasculogenesis (Fig. 8-2).11 Tumor cells can co-opt and grow around existing vessels to form “perivascular” cuffs. However, as stated earlier, these cuffs cannot grow beyond the diffusion limit of critical nutrients and may actually cause the collapse of the vessels because of growth pressure (referred to as “solid stress”). Alternatively, an existing vessel may enlarge in response to the growth factors released by tumors, and an interstitial tissue column may grow in the enlarged lumen and partition the lumen to form an expanded vascular network. This mode of intussusceptive microvascular growth has been observed during tumor growth, wound healing, and gene therapy.15–18 “Sprouting” angiogenesis is perhaps the most widely studied mechanism of vessel formation. During sprouting angiogenesis, the existing vessels become leaky in response to growth factors released by normal cells or cancer cells; the basement membrane and the interstitial matrix dissolve; pericytes dissociate from the vessel; endothelial cells (ECs) migrate and proliferate to form an array/sprout; a lumen is formed in the sprout (a process referred to as canalization); branches and loops are formed by confluence and anastomosis of sprouts to permit blood flow; and finally, these immature vessels are invested in basement membrane and pericytes. During physiological angiogenesis, these vessels differentiate into mature arterioles, capillaries, and venules, whereas in tumors they remain largely immature.5,11,12,19 During embryonic development, a primitive vascular plexus is formed from endothelial precursor cells (EPCs, also known as angioblasts) by a process referred to as vasculogenesis. In adults, EPCs—mobilized from bone marrow niches into the peripheral blood circulation—also can contribute to neovascularization (a process referred to as “postnatal” vasculogenesis) in tumors and other tissues.22–22 In addition, recent reports demonstrated that a fraction of endothelial cells in tumor vessels may be derived via transdifferentiation from cancer cells,23 or from cancer stemlike cells (e.g., in glioblastomas).11,24–26 In this larger context, the term “vasculogenesis” might be invoked to describe neovascularization in tumors from tumor stem cells.27 Tumor vasculogenesis also may occur from EPCs of non–bone marrow origin that are recruited from adjacent tissues and/or circulation.28,29 The current challenge is to discern the relative contribution of each of the these mechanisms of neovascularization during tumor growth and during and after treatment of tumors.27,30 Various proangiogenic and antiangiogenic molecules that orchestrate different steps in vessel formation, along with their functions, are listed in Table 8-1. VEGF currently is considered the most critical proangiogenic molecule. Originally discovered in 1983 as the vascular permeability factor and cloned in 1989, VEGF increases vascular permeability, promotes migration and proliferation of ECs, serves as an EC survival factor, can mobilize EPC populations from the bone marrow, and is known to upregulate leukocyte adhesion molecules on ECs.15,22,31–33 During tumor progression, or with treatment, the number of distinct angiogenic molecules produced by a tumor can increase.36–36 Thus after VEGF signaling is blocked, a tumor might rely on other, alternative angiogenic molecules (e.g., basic fibroblast growth factor [bFGF], stromal-derived factor 1α [SDF1α], placental growth factor [PlGF], or interleukin-8 [IL-8]).37 Other positive regulators of angiogenesis include the angiopoietins that are involved in stabilizing vessels and controlling vascular permeability; various proteases involved in dissolving/remodeling matrix and releasing growth factors; and recently discovered organ-specific angiogenic stimulators (e.g., endocrine gland–derived VEGF).19,38,39 Angiogenesis inhibitors include endogenous soluble receptors of various proangiogenic ligands (e.g., sVEGFR1/sFLT1) and molecules that downregulate the expression of stimulators (e.g., interferons) or that interfere with the release of the stimulators or binding with their receptors (e.g., platelet factor 4). Thrombospondins are among the first and most well-characterized endogenous inhibitors that interfere with the growth, adhesion, migration, and survival of ECs.13 Other endogenous inhibitors include fragments of various plasma or matrix proteins (e.g., angiostatin, a fragment of plasminogen; endostatin, a fragment of collagen XVIII; and tumstatin, a fragment of collagen IV).42–42 Neither the mechanisms of action of the matrix-derived inhibitors nor their physiological role are well understood.43 The generation of proangiogenic and antiangiogenic molecules can be triggered by metabolic stress (e.g., low po2, low pH, or hypoglycemia), mechanical stress (e.g., shear stress or solid stress), immune/inflammatory cells that have infiltrated the tissue, and genetic mutations (e.g., activation of oncogenes or deletion of suppressor genes that control the production of angiogenesis regulators).13,14,44–47 These molecules can emanate from cancer cells, endothelial cells, stromal cells, blood, and extracellular matrix (Fig. 8-3).48–51 Because the normal host cells differ among organs, the detailed mechanisms of angiogenesis might depend on the specific host-tumor interactions operating within a given tissue.52–59 Furthermore, because the tumor microenvironment is likely to change during tumor growth, regression, and relapse, profiles of proangiogenic and antiangiogenic molecules are likely to change with time and space.60,61 The challenge currently is to develop a unified conceptual framework to describe the temporal and spatial profiles of this increasingly diverse array of angiogenesis regulators with the aim of developing effective therapeutic strategies.37,62–64 Table 8-1 Angiogenesis Activators and Inhibitors* *Selected list updated from reference 11; for complete function and references, see supplementary information (http://steele.mgh.harvard.edu). †Also present in or affecting nonendothelial cells. In normal tissue, blood flows from an artery to arterioles to capillaries to venules to a vein. Although the tumor vasculature originates from these host vessels and the mechanisms of angiogenesis are similar, its organization may differ dramatically, depending on the tumor type, its location, and whether it is growing, regressing, or relapsing.15,65–68 In general, tumor vessels are dilated, saccular, tortuous, and chaotic in their patterns of interconnection.69 For example, whereas normal vasculature is characterized by dichotomous branching, tumor vasculature has many trifurcations and branches with uneven diameters.70,71 The fractal dimensions and minimum path lengths of tumor vasculature are different from those of normal host vasculature.67–67 The molecular mechanisms of this abnormal vascular architecture are not understood, but it seems reasonable to hypothesize that the imbalance of VEGF and angiopoietins is a key contributor.19,72 In mice, “normalization” of the tumor vasculature observed during therapies that reduce VEGF (e.g., hormone withdrawal from a hormone-dependent tumor), interfere with VEGF signaling (e.g., treatment with anti-VEGF or anti-VEGFR2 antibody; Fig. 8-4), or mimic an antiangiogenic cocktail (e.g., trastuzumab [Herceptin] treatment of a HER2-overexpressing tumor) is in concert with this molecular hypothesis.61,63,73–76 Mechanical solid stress (i.e., physical forces exerted by solid tissue components) generated by proliferating cancer and stromal cells and their excessive interstitial matrix production also can lead to the partially compressed or totally collapsed vessels often found in tumors (Fig. 8-5).64,77,78 The decompression of blood vessels observed after induction of apoptosis in perivascular cells or stromal cells, or enzymatic degradation of matrix hyaluronan, supports this mechanical hypothesis.78–81 Perhaps the combination of both molecular and mechanical factors renders the tumor vasculature abnormal, and thus both types of factors must be taken into account when designing novel strategies for cancer treatment. Blood flow in a vascular network, whether normal or abnormal, is governed by the arteriovenous pressure difference and flow resistance. Flow resistance is a function of the vascular architecture (referred to as geometric resistance) and of the blood viscosity (rheology, referred to as viscous resistance).69 Abnormalities in both vasculature and viscosity increase the resistance to blood flow in tumors.71,82–84 As a result, overall perfusion rates (i.e., blood flow rate per unit volume) in tumors are lower than in many normal tissues.85,86 Both macroscopically and microscopically, tumor blood flow is temporally and spatially chaotic. Macroscopically, four spatial regions can be recognized in a tumor (Fig. 8-6): At the microscopic level, in normal tissues, erythrocyte velocity is dependent on vessel diameter, but there is no such dependence in most tumors.54,59,89 Furthermore, the average erythrocyte velocity can be an order of magnitude lower in some tumors compared with that of normal host tissue (Fig. 8-7).59 In a given vessel within a tumor, blood flow fluctuates with time and can reverse its direction.87,89,90 In addition to the elevated geometric and viscous (rheologic) resistance, other molecular and mechanical factors contribute to this spatial and temporal heterogeneity. These factors include imbalance between proangiogenic and antiangiogenic molecules, “solid stress” generated by proliferating tumor cells and matrix production, vascular remodeling by intussusception, and coupling between luminal and interstitial fluid pressure via hyperpermeability of tumor vessels.6,16,64,65,77–79,91–93 This heterogeneity contributes to both acute and chronic hypoxia in tumors—a major cause of resistance to radiation and other therapies. Considerable effort has gone into increasing tumor blood flow for improving radiation therapy, or decreasing tumor perfusion in the case of hyperthermia. Increased perfusion also correlates with a positive response to radio/chemotherapy and patient survival.94,95 Increased perfusion has been difficult to achieve reproducibly, because tumor vasculature consists of both vessels co-opted from the preexisting host vasculature and vessels resulting from the angiogenic response of host vessels to cancer cells. The former are invested in normal contractile perivascular cells, whereas the latter lack these perivascular cells or these cells are abnormal.48,69,96 Presumably as a result, efforts to increase tumor blood flow by pharmacologic or physical agents have not always been reproducible or successful.48,69,85,96 On the other hand, the strategy of decreasing or shutting down tumor blood flow by “stealing” blood away from the “passive component” of the tumor vasculature by vasodilators, by vascular targeting, or by intravascular coagulation has shown promise in experimental systems.85,86,97–99 It also appears that judiciously applied antiangiogenic therapy could “normalize” the abnormal tumor microcirculation by pruning the immature vessels (see Fig. 8-4), thus rendering the remaining vasculature more responsive to vasoactive agents.100 Once a blood-borne molecule has reached an exchange vessel, its extravasation occurs by diffusion, convection, and, to some extent, presumably by transcytosis.100,101 The effective permeability, P, of a molecule depends on the size, shape, charge, and flexibility of the molecule and on the size, shape, charge, and dynamics of the transvascular transport pathway. In normal vessels, these pathways include diffusion along the EC membrane (for lipophilic solutes), trans-EC diffusion, interendothelial junctions (<7 nm), open or closed fenestrations (<10 nm), and transendothelial channels (including vesicles or vesicovacuolar channels).15,102 Some of these anatomic pathways may be lined with glycocalyx on ECs, thus effectively reducing the size of the pathway. A basement membrane may further retard the movement of molecules. Ultrastructure studies show widened interendothelial junctions, increased numbers of fenestrations, vesicles, and vesicovacuolar channels in tumor vessels, and a lack of normal basement membrane and pericytes.15,51,96,102,103 In concert with these ultrastructural findings, both vascular permeability and hydraulic conductivity (a measure of water movement by pressure gradient) of tumors, in general, are significantly higher than those for various normal tissues.59,104–108 Furthermore, unlike normal vessels in the surrounding tissue, tumor vessels partially lose molecular size–based selectivity in permeability to different molecules.109 Despite increased overall permeability, not all blood vessels of a tumor are leaky (Fig. 8-8, A). Even the leaky vessels have a finite pore size that is tumor dependent (Fig. 8-8, B), and ultrastructural studies show that the larger pore size in tumors represents wide interendothelial junctions.56,103 Thus permeability is inversely related to molecular size due to steric and hydrodynamic hindrance from these pores, and very large molecules, such as nanoparticles, do not penetrate tumors efficiently.110,111 Molecular shape also influences penetration of these pores, such that rod-shaped molecules have a higher permeability than do spherical molecules.112 Similarly, positively charged molecules have a higher affinity for the negatively charged glycocalyx in the pores of these angiogenic tumor vessels, reducing this hindrance and giving them a higher permeability.113–116 Not only do the vascular permeability and pore size vary from one tumor to the next (see Fig. 8-8, A-C), but within the same tumor they vary spatially and temporally, as they do during tumor growth, regression, and relapse.56,60,61 The local microenvironment plays an important role in controlling vascular permeability (Fig. 8-8, D). For example, a human glioma (HGL21) has fairly leaky vessels when grown subcutaneously in immunodeficient mice, but it exhibits blood-brain barrier properties in the brain.59,72 Such site-dependent differences for other tumors have been observed in other orthotopic sites.54,57,58 One possible explanation is that the host-tumor interactions control the production and secretion of cytokines associated with permeability increase (e.g., VEGF) and decrease (e.g., angiopoietin 1).19,39,72,117,118 A better understanding of the molecular mechanisms of permeability regulation in tumors is likely to yield strategies for improved delivery of molecular medicine to tumors. Both cancer cells and immune cells frequently move across the walls of blood vessels—the former in the process of metastasis, and the latter during immune response or cell-based immunotherapy. Both transendothelial pathways (through ECs) and periendothelial pathways (between ECs) have been proposed as a route for intravasation and extravasation of cells. Very little is known about intravasation except that tumors might shed more than a million cells per gram per day and most of these are not clonogenic.119–122 Recently, it was demonstrated that metastatic cells could bring their own stroma from the primary tumor in the form of hereotypic cell clumps.123 More is known about the molecular and cellular mechanisms of extravasation.124 When a cell enters a blood vessel, it can continue to move with the flowing blood, collide with the vessel wall, adhere transiently or stably, and finally extravasate. These interactions are governed both by local hydrodynamic forces and adhesive forces. The former are determined by the vessel diameter and fluid velocity and the latter by the expression, strength, and kinetics of bond formation between adhesion molecules and by the surface area of contact.125–129 Deformability of cells affects both types of forces.130 In addition, cancer cells may grow intravascularly at a distant site (e.g., in the lungs).131 Rolling of endogenous leukocytes is generally low in tumor vessels, whereas stable adhesion (≥30 sec) is comparable between normal vessels and tumor vessels.132 On the other hand, both rolling and stable adhesion are nearly zero in angiogenic vessels induced in collagen gels by bFGF or VEGF, two of the most potent angiogenic factors.133 Whether this observation is due to a low flux of leukocytes into angiogenic vessels and/or to downregulation of adhesion molecules in these immature vessels currently is not known. The age of the animal also plays an important role in leukocyte-endothelial interactions.134 Further insight into the types of cells that adhere to tumor vessels comes from studies on the localization of IL-2-activated natural killer (A-NK) cells in normal and tumor tissues in mice using positron emission tomography.135,136 After systemic injection, these cells localized primarily in the lungs immediately after injection and could not be detected in the tumor.135 Increased rigidity caused by IL-2 activation might contribute to the mechanical entrapment of these cells in the lung microcirculation.137,138 Constitutive expression of certain adhesion molecules in the lung vasculature also might facilitate their retention in the lungs.124 One approach to reducing lung entrapment is to reduce the rigidity of these cells.112,117 Alternatively, the lung can be circumvented by injecting A-NK cells directly into the blood supply of tumors. In this case, A-NK cells, both xenogenic and syngeneic, adhered to some blood vessels in three different tumor models via CD18 and very large antigen-4 (VLA-4) on the A-NK cells and intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and E-selectin on the activated endothelium of angiogenic vessels.33,136,139–142 These molecules can be upregulated by tumor necrosis factor–α (TNF-α) and a protein of 90 kd molecular weight (p90) that is secreted by some neoplastic cells and downregulated by transforming growth factor–β (TGF-β)—also, presumably, secreted by cancer cells.55,125,143–147 Surprisingly, the proangiogenic VEGF also can upregulate these molecules, whereas another proangiogenic molecule, bFGF, can downregulate these molecules.33,51,61,76,148 Inflammatory cells such as monocyte/macrophages or neutrophils also are recruited by VEGF and may play important roles in promoting matrix remodeling and angiogenesis.149 The challenge currently is to decrease nonspecific entrapment of immune cells in normal vessels and to increase their delivery to tumor vessels to improve various cell-based therapies, including gene therapy. The extravascular compartment of a solid tumor consists of neoplastic cells (parenchyma) and host cells (e.g., inflammatory cells and fibroblasts) residing in an interstitial matrix bathed by the interstitial fluid (see Fig. 8-1). Depending on the tumor type and its stage of differentiation, neoplastic cells might be dispersed in the matrix as individual cells (e.g., lymphomas and melanomas) or as clumps, sheets, or nests (e.g., carcinomas). More than 80% of tumors are carcinomas arising from epithelial cells. The remaining cancers include sarcomas arising from mesenchymal cells (e.g., bone or muscle cells), lymphomas arising from lymphoid tissue, leukemias arising from hematopoietic cells, and hemangiomas arising from ECs. In a poorly differentiated carcinoma, the cancer cells might be packed loosely in clumps, whereas in a well-differentiated carcinoma, the cells might be connected with intercellular junctions and tightly packed in a nest enveloped by a basement membrane. With tumor progression, cancer cells can invade the basement membrane and spread to other regions.15 These various types of normal host cells must migrate into the tumor from normal tissue. Inflammatory cells might enter the tumor via blood vessels or might infiltrate from the adjacent tissue or lymphatics.124 Other host cells, such as fibroblasts, might proliferate and migrate from the adjacent connective tissue.19,48,150 The interstitial subcompartment of a tumor is bounded by the walls of the blood vessels on one side and by the membranes of cancer and stromal cells on the other. In normal tissues, the blood vessels are surrounded by a basement membrane, which, as previously discussed, is defective in tumors.19 In addition, functional lymphatics might be confined to the tumor margin.151,152 The interstitial space of tumors, like that of normal tissues, is composed of a collagen and elastin fiber network that provides structural support to the tissue. Interdispersed in this cross-linked structure are the interstitial fluid and macromolecular constituents (polysaccharides, hyaluronan, and proteoglycans), which form a hydrophilic gel. Compared with current understanding of blood vessel formation, understanding of stroma generation is minimal. Dvorak and coworkers have proposed that the extravasated plasma protein fibrinogen, a key component of the tumor interstitial fluid, clots to form fibrin, which serves as a major component of the provisional stroma. This provisional stroma eventually is replaced by more mature connective tissue stroma. The tumor interstitial fluid also contains several other Arg-Gly-Asp (RGD)-containing proteins, including fibronectin, vitronectin, osteopontin, thrombospondin, decorin, and tenacin.15 These proteins are present in both free and bound forms. Their Arg-Gly-Asp sequence provides a binding site for adhesion that assists in the migration of various cells, including stromal cells. In addition to extravasating from the leaky tumor vessels, these proteins, along with collagen and various proteoglycans, also are synthesized by the stromal cells, albeit in a form that differs from that in the plasma or normal tissues.15 Tumor interstitial fluid also can contain various growth factors that facilitate stroma formation. For example, in vitro studies suggest that platelet-derived growth factor-BB (PDGF-BB) is involved in the recruitment of fibroblasts to tumors, and TGF-β induces the production of collagen and other matrix molecules in tumors.19,153 With the increasing interest in using the fragments of matrix constituents for controlling angiogenesis, increased understanding of the molecular and cellular mechanisms of stroma generation in tumors will increase.43 Once a molecule has extravasated, its movement through the interstitial space occurs by diffusion and convection.101,154 Diffusion is proportional to the concentration gradient in the interstitium, and convection is proportional to the interstitial fluid velocity, which, in turn, is proportional to the fluid pressure gradient in the interstitium. Just as the interstitial diffusion coefficient D (cm2/s) relates the diffusive flux to the concentration gradient, the interstitial hydraulic conductivity K (cm2/mmHg/sec) relates the interstitial velocity to the pressure gradient.134 Values of these transport coefficients are governed by the structure and composition of the interstitial compartment and by the physicochemical properties of the solute molecule.155–165 The value of K for a human colon carcinoma xenograft (LS174T), measured using two different methods, was found to be higher than that of a hepatoma, which, in turn, was higher than that of the normal liver.167–167 Using fluorescence recovery after photobleaching, D of various molecules in tumors was found to be about one third that in water and higher than the values in the host tissue (Fig. 8-9, A).157,168 Collagen content and structure have a significant effect on D in tumors.159,163,166,169–173 This finding is surprising because hyaluronan and proteoglycans, not collagen, account for most of the resistance to transport in normal tissues. Because collagen is produced by host cells (e.g., fibroblasts), the penetrability into a tumor depends on the host-tumor interaction (see Fig. 8-9, A). Thus agents that interfere with collagen synthesis and/or organization (e.g., relaxin, losartan, and bacterial collagenase) might increase interstitial transport in tumors156,169,170 (Fig. 8-9, B). The time constant for a molecule with diffusion coefficient D to diffuse across a distance L is approximately L2/4D. For diffusion of immunoglobulin G (IgG) in tumors, this time constant is on the order of 1 hour for a 100-µm distance, days for a 1-mm distance, and months for a 1-cm distance. Thus for a 1-mm distance in tumor, diffusional transport would take days, and for a 1-cm distance in tumor, it would take months. Furthermore, the path through the interstitium is tortuous over large length scales, greatly increasing the distance a molecule must travel.158 If the central vessels have collapsed completely as a result of cellular proliferation and interstitial matrix rearrangement, the reduced delivery of macromolecules by blood flow would make diffusion the primary mechanism of delivery to this necrotic center.6,79 Binding of a low- or high-molecular-weight drug to plasma proteins and various tissue components could further retard their transport in tumors.168,174–180 The role of binding is clearly illustrated in Figure 8-9, C, which compares the rate of fluorescence recovery of a photobleached spot in tumor tissue injected with a nonspecific versus a specific IgG. In addition to the heterogeneity of D in tumors, the most unexpected result of these photobleaching studies was the large extent (30% to 40%) of nonspecific binding.168
Vascular and Interstitial Biology of Tumors
Introduction
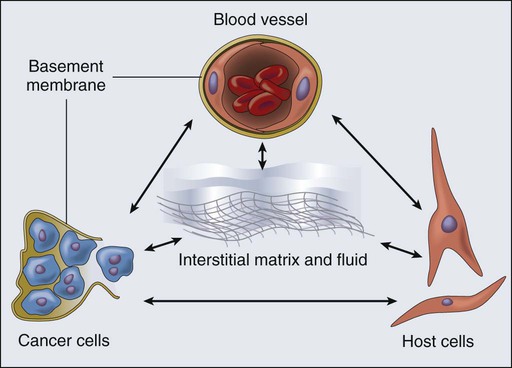
Vascular Compartment
New Vessel Formation
Cellular Mechanisms
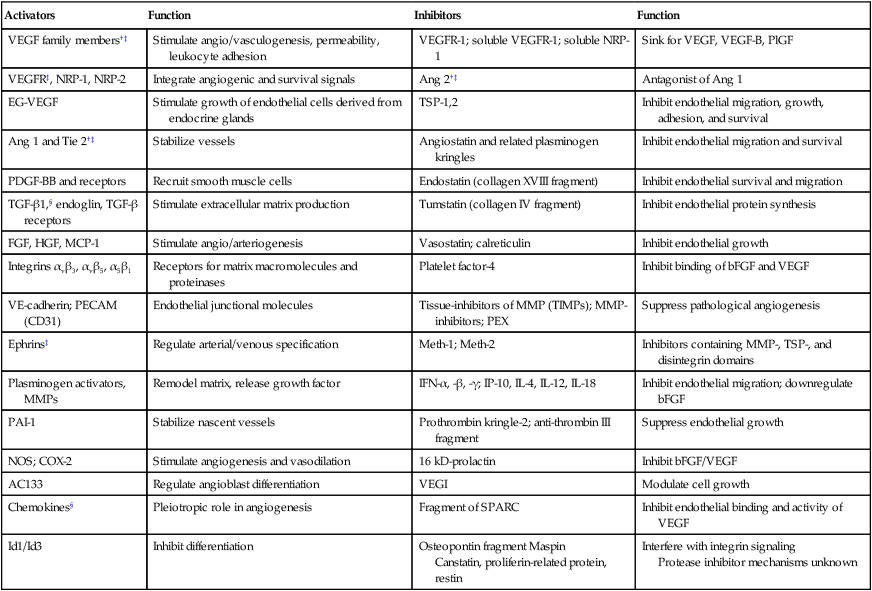
Molecular Mechanisms
Activators
Function
Inhibitors
Function
VEGF family members†‡
Stimulate angio/vasculogenesis, permeability, leukocyte adhesion
VEGFR-1; soluble VEGFR-1; soluble NRP-1
Sink for VEGF, VEGF-B, PlGF
VEGFR‡, NRP-1, NRP-2
Integrate angiogenic and survival signals
Ang 2†‡
Antagonist of Ang 1
EG-VEGF
Stimulate growth of endothelial cells derived from endocrine glands
TSP-1,2
Inhibit endothelial migration, growth, adhesion, and survival
Ang 1 and Tie 2†‡
Stabilize vessels
Angiostatin and related plasminogen kringles
Inhibit endothelial migration and survival
PDGF-BB and receptors
Recruit smooth muscle cells
Endostatin (collagen XVIII fragment)
Inhibit endothelial survival and migration
TGF-β1,§ endoglin, TGF-β receptors
Stimulate extracellular matrix production
Tumstatin (collagen IV fragment)
Inhibit endothelial protein synthesis
FGF, HGF, MCP-1
Stimulate angio/arteriogenesis
Vasostatin; calreticulin
Inhibit endothelial growth
Integrins αvβ3, αvβ5, α5β1
Receptors for matrix macromolecules and proteinases
Platelet factor-4
Inhibit binding of bFGF and VEGF
VE-cadherin; PECAM (CD31)
Endothelial junctional molecules
Tissue-inhibitors of MMP (TIMPs); MMP-inhibitors; PEX
Suppress pathological angiogenesis
Ephrins‡
Regulate arterial/venous specification
Meth-1; Meth-2
Inhibitors containing MMP-, TSP-, and disintegrin domains
Plasminogen activators, MMPs
Remodel matrix, release growth factor
IFN-α, -β, -γ; IP-10, IL-4, IL-12, IL-18
Inhibit endothelial migration; downregulate bFGF
PAI-1
Stabilize nascent vessels
Prothrombin kringle-2; anti-thrombin III fragment
Suppress endothelial growth
NOS; COX-2
Stimulate angiogenesis and vasodilation
16 kD-prolactin
Inhibit bFGF/VEGF
AC133
Regulate angioblast differentiation
VEGI
Modulate cell growth
Chemokines§
Pleiotropic role in angiogenesis
Fragment of SPARC
Inhibit endothelial binding and activity of VEGF
Id1/Id3
Inhibit differentiation
Osteopontin fragment Maspin
Canstatin, proliferin-related protein, restin
Interfere with integrin signaling
Protease inhibitor mechanisms unknown
Vascular Architecture
Blood Flow and Microcirculation
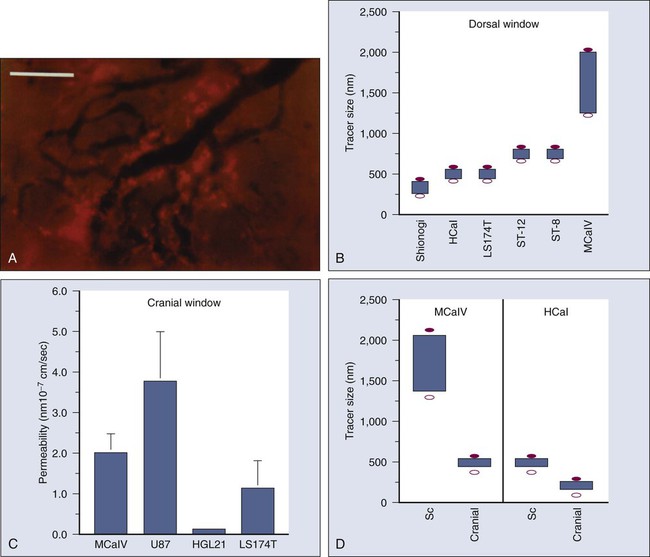
Vascular Permeability
Movement of Cells Across Vessel Walls
Extravascular Compartment
Composition and Origin
Interstitial Transport
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Vascular and Interstitial Biology of Tumors