Unknown Primary
INTRODUCTION
Most gynecologic cancers present with bleeding, pain, or other symptoms caused by an enlarging primary tumor. However, in some cases, patients are initially diagnosed when lymphadenopathy or extragenital masses are found to contain squamous carcinoma or mullerian adenocarcinoma suggestive of a gynecologic primary cancer. In rare cases, even an exhaustive search fails to reveal any primary source.
Although this is an uncommon problem, it is worthy of special consideration for several reasons:
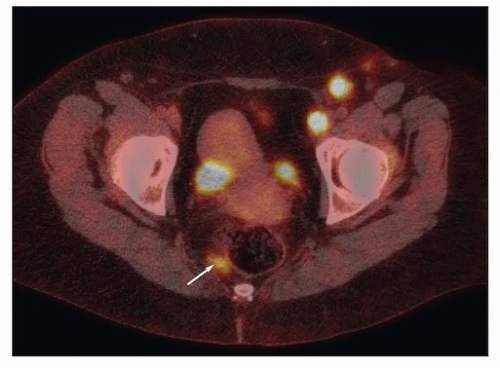
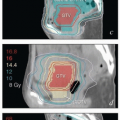
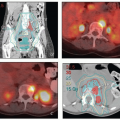
Many cases initially labeled as having an unknown primary have a source discovered after additional search (Fig. 15.1, CS 12.8 and 12.9). Failure to identify the source before radiotherapy is initiated can severely compromise the chance for cure.
Even patients who have large pelvic masses or extensive lymphadenopathy are frequently curable with definitive chemoradiation (CS 15.2-15.4). For this reason, such patients should not be equated with the generally incurable patients who present with hematogenously disseminated disease from an unknown primary source.
Because they do not develop symptoms of a primary lesion, these patients often have very bulky lymphadenopathy. For this reason, they provide good case studies for the management of patients with extensive inguinal or pelvic lymphadenopathy.
These are always challenging cases that require careful workup, multidisciplinary collaboration, and skillful technique.
PRETREATMENT EVALUATION
The primary goals of the pretreatment evaluation of patients who present with inguinal or pelvic cancers without an evident primary are to:
Rule out a previously undetected primary source
Evaluate the extent of locoregional involvement
Rule out distant metastases that would preclude a definitive treatment approach
Assess and treat secondary symptoms of tumor
Search for a Primary Source
Several features including the distribution of nodal disease (Fig. 15.1, Chapter 5, Table 5.1) and the type of carcinoma can suggest a direction for the initial evaluation. Squamous carcinomas are more likely to arise in the lower genital tract and müllerian adenocarcinomas in the cervix, endometrium, tubes, or ovaries. Testing for high-risk HPV types can be helpful in determining the potential source of poorly differentiated carcinomas or adenocarcinomas but cannot differentiate between squamous carcinomas of the anus, urethra, or lower genital tract, all of which can be sources of HPV-positive squamous lesions.1 Extragenital pelvic cancer can also arise from endometriosis (CS 15.5).2,3,4
If the patient has a history of pelvic or vulvar surgery, an effort should be made to obtain histologic material from that surgery to determine whether a cancer was present. However, if a source is not found in the most obvious sites, patients should always have a thorough evaluation of all potential primary sites.
Evaluation should begin with a careful pelvic examination including colposcopy of the cervix and vagina if no gross lesions are found. Endometrial and endocervical curettage should be obtained, and any suspicious lesions should be biopsied. In our experience, the vulva and vagina are the most common sites of initially undetected primary cancers (CS 11.6, 12.8, and 12.9). Small cancers of the anal canal can also be missed on initial examination (Fig. 15.1). In many cases, anesthesia is required to perform an optimal examination. Examination under anesthesia can be combined with cystoscopy to rule out primaries of the bladder or urethra and with proctoscopy to rule out primary cancers of the anus or rectum. If there is evidence of or a history of cervical intraepithelial neoplasia, a cervical cone biopsy may be indicated (CS 15.1).
Evaluation of Regional and Distant Disease Extent
Techniques for evaluation of regional disease extent are discussed in detail in Chapter 4. PET-CT is recommended in all cases to evaluate regional sites and to help to rule out distant metastases. CT with contrast can be helpful and can be fused with radiation planning CT scans to assist in target volume designation. If patients undergo treatment with lymphadenectomy, postoperative CT should always be obtained to rule out residual gross nodal disease. MRI is useful in evaluating soft tissue masses and the extent of soft tissue infiltration from large nodal masses.
Evaluation and Management of Secondary Symptoms
At diagnosis, most patients have pain that can be severe. Good pain control is important for patient comfort and also to achieve stable positioning for radiation treatment. Bulky lymphadenopathy or pelvic masses can also cause hydronephrosis, and bulky lymphadenopathy places patients at increased risk for venous thrombosis. Management of these symptoms is discussed in Chapter 7.
RISK ASSESSMENT
Very few data have been published concerning the outcome of patients presenting with regionally confined pelvic or inguinal carcinoma of unknown primary. In a study of 37 patients with soft tissue or nodal lesions presenting in the abdomen or pelvis, Kelly et al. reported better survival rates for patients with squamous carcinomas versus adenocarcinoma or other histologic types.5
MULTIDISCIPLINARY TREATMENT STRATEGY
The Role of Surgery
Adenocarcinoma Arising in Pelvic Soft Tissues
Adenocarcinoma of unknown primary can arise in the soft tissues of the pelvis, usually in the region of the pouch of Douglas or adjacent to the rectosigmoid. Endometrioid histology with associated endometriosis or a history of endometriosis suggests an origin in pelvic endometriosis.
For patients who have adenocarcinoma thought to be arising from endometriosis, some form of surgical evaluation, often by laparoscopic approach, may be recommended to rule out microscopic intraperitoneal disease. The decision whether to attempt surgical resection of gross disease may be influenced by the size of the tumor, proximity to critical structures, and likelihood of complete resectability. Large tumors that are intimately associated with the sigmoid can be difficult to control with definitive radiotherapy without causing serious bowel damage; if it can be accomplished, surgical resection of tumors in this region is probably beneficial although close or positive operative margins are frequent; for this reason postoperative radiotherapy is usually recommended if there is no evidence of peritoneal or distant metastases (CS 15.5).
Carcinoma Involving Lymph Nodes
As demonstrated in CS 15.1-15.4, even patients who have extensive lymphadenopathy can often be cured if locoregional control can be achieved. Although it is tempting to remove gross lymphadenopathy, the side effects of lymphadenectomy can easily outweigh the benefits. Because these cases usually present with very large nodal masses, resected nodes are nearly always found to have extracapsular disease extension,
requiring postoperative radiation therapy doses nearly as high as those given for definitive treatment. Postoperative complications can delay radiation, potentially decreasing its effectiveness (CS 15.4). However, in many cases, optimal treatment of regionally extensive cancers requires skill in advanced techniques including IMRT, image guidance, and adaptive planning. If these are unavailable, the arguments for initial surgical treatment may be stronger.
requiring postoperative radiation therapy doses nearly as high as those given for definitive treatment. Postoperative complications can delay radiation, potentially decreasing its effectiveness (CS 15.4). However, in many cases, optimal treatment of regionally extensive cancers requires skill in advanced techniques including IMRT, image guidance, and adaptive planning. If these are unavailable, the arguments for initial surgical treatment may be stronger.
In general, well-circumscribed, moderate-sized nodes adjacent to critical structures that might limit the ability to deliver a definitive radiation dose may be good candidates for surgical resection. However, partial debulking of unresectable tumors is usually not recommended because it does not increase the effectiveness of radiation therapy and increases the likelihood of lymphocyst, infection, lymphedema, and harmful treatment delays. See Chapter 3 for further discussion of this subject.
The Role of Chemotherapy
Because these cases nearly always present with large tumor masses or extensive regional disease, chemotherapy is usually recommended either as a concurrent treatment with radiation or as an adjuvant. The sequencing is guided by the established role of chemotherapy for primary cancers of similar histologic type. For example, squamous carcinomas that present with an unknown primary are treated with concurrent chemoradiation, using regimens (typically weekly cisplatin) that have been proven effective for squamous carcinomas of the cervix or vulva. Concurrent chemotherapy is also given with radiation to maximize the chance of local control of adenocarcinomas arising in endometriosis or lymph nodes; however, in these cases we have usually also recommended the addition of adjuvant chemotherapy (typically carboplatin and paclitaxel) as would be used for node-positive müllerian adenocarcinomas arising in the endometrium or ovary.
RADIATION THERAPY—TECHNICAL CONSIDERATIONS