, Ahmad Ameri1 and Mona Malekzadeh2
(1)
Department of Clinical Oncology, Imam Hossein Educational Hospital, Shahid Beheshti University of Medical Sciences (SBMU), Shahid Madani Street, Tehran, Iran
(2)
Department of Radiotherapy and Oncology, Shohadaye Tajrish Educational Hospital Shahid Beheshti University of Medical Sciences (SBMU), Tehran, Iran
Anticancer properties of radiation were discovered by skin changes after exposure to radiation; initially, radiation was used for the treatment of skin diseases such as lupus erythematous. Skin cancer was the first cancer treated by radiation, and dermatitis accompanies radiation therapy since its discovery.
Radiation dermatitis is one of the most common adverse effects of radiation therapy. Approximately 90–95% [1, 2] of all patients treated with radiation therapy will experience a skin reaction at the treated area. Radiation dermatitis is especially seen in radiation therapy for breast cancer, head and neck cancer, lung cancer, and sarcoma due to the superficial position of these cancers and higher radiation doses to the skin. Various degrees of radiation dermatitis are experienced by patients undergoing radiation therapy. In most patients, the radiation dermatitis is mild to moderate (Grades 1 and 2); about 15–25% of patients experience severe reactions [3–5].
1.1 Mechanism
Radiation to the skin results in direct cellular injury and inflammatory cell influx. After the initial dose of radiation, cellular damage to epidermal basal cells, endothelial cells, Langerhans cells, and vascular components occur, and an inflammatory response in the epidermis and dermis develops following every subsequent fraction of radiation [1].
Initial cellular injury results from short-lived free-radical production and irreversible double-stranded breaks in nuclear and mitochondrial DNA [6]. Clumping of nuclear chromatin, swelling of the nucleus, nuclear disfiguration or loss of the nuclear membrane, mitochondrial distortion, and degeneration of the endoplasmic reticulum, as well as direct cellular necrosis and apoptosis, occur due to cellular damage. Repetitive cellular damage caused from each fraction of treatment does not allow time for cells to repair DNA damage [7–11].
Because of free-radical production, an inflammatory cytokine cascade is induced, and several cytokines and chemokines are produced including interleukins 1 and 6, tumor necrosis factor-α, and transforming growth factor-β [12]. These cytokines act on the endothelial cells of local vessels, which express adhesion molecules constitutively and induce migration of leukocytes and other immune cells from circulation to irradiated skin [13].
Reduction and impairment of functional stem cells induced by radiation lead to an alteration in the normal turnover of skin cells [6].
A prompt skin reaction may appear after exposure that is related to the inflammatory response, release of histamine-like substances, permeability and dilation of capillaries, and subsequent dermal edema. It presents with early transient skin erythema that can be seen within a few hours after radiation and subsides after 24–48 h [14].
After subsidence of initial erythema, the later phase includes additional edema, an increased dilation in capillaries, and erythrocyte extravasation, which resulted in an erythematous reaction. This phase is followed by thinning of the epidermis, damage to epithelial cells, degeneration of glands, reduced secretion of sebum, and sweat and clumps of exfoliated corneocytes (scales), which manifests as dry desquamation.
If damage to the basal cells and glands is more severe, epidermal necrosis, fibrinous exudates, and moist desquamation occur. The manifestation of moist desquamation results from the formation of small blisters in and around the basal layer of the epidermis that may also extend into the more superficial layers. The epidermis sloughs when these blisters rupture and coalesce, denuding the dermis and causing permanent epilation [15–17]. Finally, skin necrosis or ulceration of the full-thickness dermis may develop as the most severe damage of higher radiation doses [15, 16].
1.2 Timing
As noted above, a transient early erythema can be seen within a few hours of radiation, even after a single fraction of radiation (2 Gy) and subsides after 24–48 h [14]. The main erythematous reaction appears at about the second to third week (within 1–4 weeks) of radiation. Earlier reaction can be seen in patients with particularly sensitive skin. Effects typically continue to progress during treatment [18–20], and subsequently dry desquamation can develop after the third week or after a cumulative dose of 30 Gy, which is clinically characterized by dryness, scaling, and pruritus. Following 4–5 weeks (45–60 Gy) of therapy, moist desquamation may occur, which is characterized by serous oozing and exposure of the dermis [14]. Symptoms persist for the duration of radiation therapy and peak 1–2 weeks after treatment completion. At about 3–5 weeks after radiation, the skin begins to recover with epidermal regeneration and within 1–3 months (depending on the severity of reaction), complete healing occurs [1, 6, 18, 19, 21, 22]. Post-inflammatory hyperpigmentation may persist for 5–7 weeks or even longer after radiation [20, 23]. Patient skin type may also influence timing and severity of radiation reactions, reflected by interindividual variation in skin presentation of radiation damage.
1.3 Sign and Symptoms
Mild dermatitis (Grade I) presents with erythematous skin accompanied by mild edema, pruritus, or pain. Dry, peeling skin (desquamation) and temporary epilation due to involvement of adnexal structures may occur (Fig. 1.1).
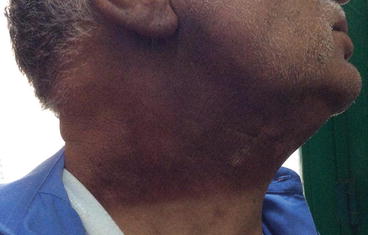

Fig. 1.1
Grade 1 dermatitis of neck after radiation therapy for laryngeal cancer
Moderate dermatitis (Grades 2 and 3) consists of tender or edematous reactions and moist desquamation (Fig. 1.2). The integrity of the dermis is impaired, and secondary infection with Staphylococcus aureus may occur (Fig. 1.3).
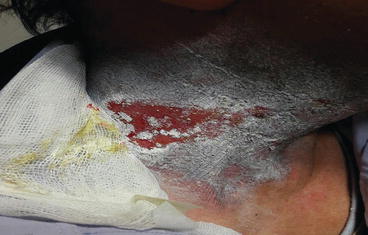
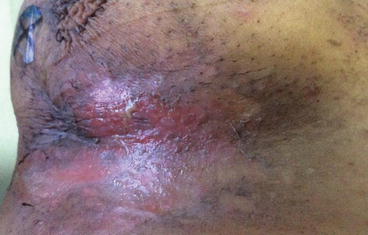

Fig. 1.2
Grade 2 dermatitis of neck after radiotherapy for nasopharyngeal cancer with multiple node involvement

Fig. 1.3
Grade 3 dermatitis in inframammary fold after breast irradiation
Severe dermatitis (acute radionecrosis) is rarely seen and may occur in the setting of very extensive superficial tumors treated with massive radiation doses over a very short period. It manifests as a very painful inflammatory or hemorrhagic plaque with deep necrosis, which can expose the muscles, tendons, and bones [19, 21, 22].
1.4 Scoring
The severity of radiation dermatitis is graded using several grading systems such as Common Terminology Criteria for Adverse Event (NCI-CTCAE grading), Radiation Therapy Oncology Group (RTOG) toxicity scoring system, and the Radiation-Induced Skin Reaction Assessment Scale (RISRAS).
Common Terminology Criteria for Adverse Event (NCI-CTCAE grading) is one of the most commonly used, and NCI-CTCAE v4.3 has been shown in Table 1.1 [24]. RTOG scoring is quite similar to NCI-CTCAE grading.
Table 1.1
CTCAE v4.3 for dermatitis
Definition | |
|---|---|
Grade 1 | Faint erythema or dry desquamation |
Grade 2 | Moderate to brisk erythema; patchy moist desquamation, mostly confined to skin folds and creases; moderate edema |
Grade 3 | Moist desquamation in areas other than skin folds and creases; bleeding induced by minor trauma or abrasion |
Grade 4 | Skin necrosis or ulceration of full-thickness dermis; spontaneous bleeding from involved site |
1.5 Risk Factors
Treatment and patient-related factors may affect incidence and severity of dermatitis.
1.5.1 Treatment-Related Factors Including
The higher total dose, higher dose per fraction, reduced overall treatment time, beam energy (depend on type and quality of the beam), higher volume and surface area exposed, the junction between the electron and photon field, use of tissue expander and bolus material [25–27], concurrent systemic therapy with radiation (radiosensitizing drug such as paclitaxel, docetaxel, anthracyclines, dactinomycin, methotrexate, 5-fluorouracil, hydroxyurea, bleomycin, and cetuximab) [6, 28] are related to the development of radiation dermatitis.
Epidermal growth factor receptor (EGFR) is highly expressed in the epidermis, particularly in the proliferative basal cell layers, and has a major role in the regulation of multiple phases of epithelial biology, including cell cycle progression, differentiation, cell movement, and cellular survival; it also has an essential impact on the inflammatory reactions of the skin [29–31]. There is a reciprocal relationship between radiation and epidermal growth factor receptor inhibitors’ (like cetuximab) effects on skin.
More severe radiation dermatitis is seen in patients receiving cetuximab plus radiation therapy than radiation therapy alone or even concurrent chemoradiotherapy [5, 15]. There is a slightly longer duration of radiation dermatitis (11.1 versus 9.4 weeks) [32] and an earlier onset of radiation dermatitis (week 1 or 2 compared with 3–5 weeks, respectively) [15], when cetuximab is added to radiation therapy. Dermatitis usually reveals rapid recovery with no scarring following the end of treatment of cetuximab plus radiation therapy [15]. A deferred cetuximab-induced acne-like rash appears within irradiated fields in concurrent treatment of cetuximab with radiation therapy (about 3–5 weeks after initiation of treatment versus 7–10 days, respectively). It seems that there is no significant relationship between the severity of cetuximab-associated acne-like rash outside irradiated fields and the severity of radiation dermatitis [32].
1.5.2 Patient-Related Factors Include
Demographic and behavioral factors, comorbid diseases, inherited disorders, and site of radiation therapy can affect incidence and severity of radiation-induced dermatitis [2, 33–41] (Table 1.2).
Table 1.2
Patient-related predisposing factors for radiation-induced dermatitis
Demographic and behavioral factors | Comorbid disease | Inherited disorders | Site of radiation therapy |
|---|---|---|---|
Black race Obesity Female gender Advanced age Breast implant Smoking Poor nutrition | Actinic skin lesions Seroma aspiration after breast surgery Systemic lupus erythematosus Systemic sclerosis Juvenile rheumatoid arthritis HIV infection Diabetes mellitus Hypertension | Basal cell nevus disease Fanconi anemia Bloom syndrome Xeroderma Pigmentosum Ataxia-telangiectasia | Anterior neck Extremities Submammary Skin folds |
Smoking seems to impair wound healing by cutaneous vasoconstriction [17].
The impact of increasing age on radiation dermatitis is related to decreased epidermal turnover resulting in extended healing times. Coexisting diseases like hypertension, diabetes, obesity, or malnutrition in older patients affect the severity and resolution of radiation dermatitis [17].
It has been reported that aspiration of a seroma following breast surgery may lead to damage to the lymphatic system, compromising wound healing and leading to a severe skin reaction during radiation therapy [41].
Patients with systemic lupus erythematosus (SLE), juvenile rheumatoid arthritis (JRA), and systemic sclerosis have significantly greater radiosensitivity and are at greater risk for developing radiation side effects. The presence of this connective tissue disease is considered as a relative contraindication to radiation therapy [42, 43].
Wound healing is complicated in patients with uncontrolled diabetes mellitus due to poor macrophage function including phagocytic activity, prolonged inflammatory phase, and greater risk of infection. Diabetes’ effect on normal tissue reaction during radiation therapy needs further study [17].
There are a few rare genetic mutations that may predispose patients to severe radiation dermatitis, including mutations in the ataxia-telangiectasia gene. It has been suggested that unanticipated severe radiation dermatitis may indicate the presence of undetected genetic abnormalities in the ATM gene (ataxia-telangiectasia mutated) that can predispose patients to cutaneous complications [44, 45].
No studies have specifically investigated the effect of hemoglobin level on normal skin exposed to radiation therapy.
1.6 Diagnosis
Acute radiation dermatitis is a clinical diagnosis based on the finding of skin changes that shows a sharp demarcation of the skin changes and their limitation to the irradiated areas and the history of radiation therapy and its duration. A skin biopsy is usually not necessary for the diagnosis of radiation dermatitis [13].
Two important differential diagnoses of acute radiation dermatitis are radiation recall and cellulitis. Radiation recall is an acute inflammatory skin reaction limited to the area that was previously irradiated and occurs following cytotoxic agents or other drug administration. It occurs at least 7 days after radiation therapy and may occur weeks to years after radiation therapy [46–48]. Cytotoxic drugs such as docetaxel, doxorubicin, and paclitaxel have been associated with radiation recall. Radiation recall manifestations include a rash, dry desquamation, erythema, ulceration, hemorrhage, or even necrosis. Radiation recall generally resolves within 1–2 weeks after drug discontinuation. Patient’s history of skin reaction following radiation treatment and medications will help to reach an accurate diagnosis [49].
Cellulitis is a bacterial infection of skin and subcutaneous tissues. It is characterized by localized erythema, warmth, swelling, pain, tenderness, fevers, and even purulent drainage. These clues can differentiate cellulitis from radiation dermatitis. However, radiation dermatitis is a predisposing factor of secondary cellulitis, and it may be possible for both conditions to exist together. In this condition a culture from the wound may confirm the diagnosis of cellulitis. Empiric antibiotic treatment may also be helpful in these patients [49].
Eczema is a chronic, relapsing, inflammatory skin condition that is diagnosed by clinical presentation of symmetrical, pruritic, dry skin that persists for <6 months. Chronic eczema may exacerbate radiation dermatitis as both conditions are inflammatory in nature. Eczema may be distinguished from radiation dermatitis by its symmetrical distribution. In contrast, radiation dermatitis will only appear within radiation fields [49].
Recurrent or secondary tumors should be considered if atypical plaques and nodules develop within the radiation field. Although irradiated skin may present some difficulty in healing, a biopsy is useful in this clinical situation.
1.7 Prevention
1.7.1 General Measures
General recommendations for skin care in patients that are treated with radiation therapy include [13, 54]:
Washing the irradiated area daily with water and mild soap (pH-neutral agents) or normal saline then dry it and use a water-based moisturizer.
Wearing loose-fitting clothes.
Avoiding skin irritants such as perfumes and alcohol-based lotions and wax or other depilatory creams.
Avoiding extremes of heat and cold.
Avoiding sun exposure and use of sunscreen with a minimum SPF 30.
Currently, application of topical steroids is the only prophylactic intervention for radiation dermatitis protection suggested with a high level of evidence [55, 56]. Prophylactic application of topical steroids can reduce the severity of radiation skin reaction because of their anti-inflammatory effects (i.e., reducing the production of IL-1, IL-2, IL6 IFN-γ, TNF, and histamine and inhibiting leukocyte migration) [14, 57].
Low- to medium-potency topical corticosteroids (groups 4–6) such as mometasone furoate 0.1% or hydrocortisone 1% cream or methylprednisolone aceponate cream 0.1% or beclomethasone dipropionate spray are applied to the treatment field once or twice daily after each radiotherapy treatment [13, 58–61].
Treatment should be stopped if there is any exudate from the affected area [54] or if a skin infection is suspected.
Other protective options with primarily positive results that warrant further research on their efficacy in preventing and managing acute radiation dermatitis include:
1.7.2 Intensity-Modulated Radiation Therapy (IMRT)
1.7.3 Low-Level Laser Therapy
Low-level laser therapy is a form of phototherapy that induces the wound-healing process [65]. It has generated great interest in preventing radiation side effects such as dermatitis, xerostomia, or oral mucositis. The primary promising results of these trials warrant further research on its efficacy in preventing and managing acute radiation dermatitis [66].
1.7.4 Amifostine
Amifostine is a thiol that selectively protects normal cells from radiation damage by scavenging oxygen-derived free radicals. It has been shown that the severity of radiation dermatitis is significantly lower among patients receiving amifostine [67–69]. Further studies are needed to determine the impact and safety of amifostine as a cytoprotective agent against acute radiation dermatitis.
1.7.5 Ascorbic Acid
Ascorbic acid has antioxidant capacity and anti-free-radical action. No benefit of its topical application has been found for radiation dermatitis protection. However, oral ascorbic acid in breast cancer radiation therapy with a volume lower than 500 mL and in those that received a radiation dose between 107% and 110% of the prescribed dose showed positive results [70, 71].
1.7.6 Oral Zinc
1.7.7 Silver Leaf Dressing
1.7.8 MAS065D
MAS065D (Xclair) is a nonsteroidal water-in-oil cream that contains hyaluronic acid, shea butter (emollient), glycyrrhetinic acid (licorice extract with anti-inflammatory properties), Vitis vinifera (antioxidant activity), and telmesteine (anti-elastase and anti-collagen activity in vitro) [77]. MAS065D seems to be effective in the management of radiation dermatitis, but further studies are necessary [78, 79].
1.7.9 Soft Dressing or Barrier Film
It seems that prophylactic use of friction protections with soft dressing or barrier film in areas at risk such as the axilla, head and neck region, perineum, and skin folds could be an effective intervention for reducing skin reaction severity [81].
Other options without well-designed trial support include:
1.7.10 Hyaluronic Acid
Hyaluronic acid cream appears to reduce inflammatory response and oxidative free-radical damage and have been evaluated in radiation therapy studies as a preventive method for radiation dermatitis. Additional studies are required to clarify hyaluronic acid role as a preventive option in radiation dermatitis [82, 83].
1.7.11 Trolamine
A topically applied salicylates analgesic, this agent has been shown to promote the wound-healing process by recruitment of macrophages and enhancement of granulation tissue formation, which has therapeutic applications in dermatology. Current evidence does not support the use of trolamine for radiation dermatitis and requires more research to clarify its efficacy in prevention of radiation dermatitis [84–87].
1.7.12 Topical Sucralfate
Sucralfate is a topical drug that has been applied for the treatment of several types of epithelial wounds such as ulcers, inflammatory dermatitis, mucositis, and burn wounds. Some studies have evaluated the efficacy of sucralfate cream for prevention of radiation dermatitis with differing results. Current data do not support it to prevent radiation dermatitis [87–90].
1.7.13 Theta Cream
1.7.14 Dexpanthenol (Provitamin B5)
1.7.15 Calendula
1.7.16 Pentoxifylline
A useful drug in a variety of vaso-occlusive disorders, pentoxifylline has ant-inflammatory properties. The pentoxifylline effects on acute skin reactions need to be determined in well-designed studies [98, 99].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree