Introduction
Sarcomas are a rare and heterogenous group of malignant tumors that arise from soft tissues or bone. They constitute less than 1% of all adult malignancies and approximately 12% of pediatric cancers. Approximately 80% to 84% of sarcomas originate from soft tissues, and the rest originate from bone. , Given the histopathologic spectrum and ability of sarcomas to arise anywhere in the body, treatment paradigms are often dictated by the histology, grade, site of origin, stage, and age of the patient.
Surgical resection remains the mainstay of treatment when possible. For soft tissue sarcomas, radiation therapy (RT) has an integral role in the treatment approach. The combination of RT and a wide local excision (limb-sparing surgery) leads to better local control (LC) than does either modality alone for most soft tissue sarcomas. This was demonstrated in two randomized trials, both of which showed that combined-modality therapy resulted in a reduced risk of local recurrence by approximately 20% to 25%. RT can be administered preoperatively or postoperatively, and both have their advantages. Our practice consistently uses preoperative RT, which is endorsed by our multidisciplinary teams because of the lower rates of radiation-related late toxicities.
The use of RT for bone sarcomas depends strongly on the histology and resectability of the tumor. For bone sarcomas that are unresectable, primary RT is commonly used to achieve LC, with varying degrees of success; one study showed a 5-year LC rate of 56% among patients with nonmetastatic extremity osteosarcoma who refused resection. Postoperative radiation may also be used for sarcomas originating in bone, depending on the margin status and response of the tumor to chemotherapy.
Although the list of sarcoma histologies is too exhaustive to review in this chapter, readers should be aware of several of the more common types. Some of the most common types of soft tissue sarcomas include undifferentiated pleomorphic sarcoma (historically called malignant fibrous histiocytoma), liposarcoma, leiomyosarcoma, and synovial sarcoma. Unusual but important subtypes also include desmoid tumors and dermatofibrosarcoma protuberans, both of which can be locally aggressive. Several of the more common bone sarcomas include osteosarcoma, Ewing sarcoma, and chondrosarcoma.
Much of the treatment paradigm, consisting of surgery and RT, with or without chemotherapy, is based on histology. However, the anatomic site of origin is also a critical factor to consider, particularly for the choice of local therapy. For instance, the strategy to treat a sarcoma in the distal leg is often different than that for a sarcoma that arises in the base of skull or retroperitoneum. Not only does the site of origin matter from a surgical standpoint, but also it factors into the approach taken by the radiation oncologist. Different anatomic sites require thoughtful radiation planning because of treatment volumes, neighboring critical structures, and toxicity considerations.
One radiation modalities with increasing applicability for the treatment of sarcomas is proton beam radiation. The goal of this chapter is to provide some perspective on the use of proton beam RT and summarize the available data supporting its use in the treatment of sarcomas.
Sarcomas and proton beam radiation
Both access to and justification for the use of proton beam therapy (PBT) for treatment of various malignancies is increasing. However, debate still exists among the wider health care community regarding its value. Contextually, it is important to understand that protons were initially developed during the era of two-dimensional (2D) photon therapy. Therefore, PBT provided, at one time, a way to deliver more conformal and often higher doses of radiation than could be achieved with photons. Yet with the development of intensity-modulated RT (IMRT), photons gained the capability of highly conformal dose-escalated distributions as well. The disadvantage of IMRT is its higher integral dose; PBT delivers 50% to 60% less integral dose to the rest of the body at the same or higher doses. ,
In a joint phase II study by the MD Anderson Cancer Center and the Massachusetts General Hospital, pediatric patients with rhabdomyosarcoma were treated with PBT from 2005 through 2012. A secondary objective of the study involved generating comparison IMRT plans for each patient. The authors observed a statistically higher integral dose for IMRT plans for treating tumors of the head and neck (1.8× higher), genitourinary system (1.8× higher), trunk and extremities (2.0× higher), and orbit (3.5× higher). The main concern related to higher integral doses is that they may influence the risk of secondary malignancies, which was observed in a study by Chung and colleagues. They reported that patients treated at Harvard with PBT had an observed 6.9 cancers per 1000 person-years compared with 10.3 cancers per 1000 person-years in matched patients from the Surveillance, Epidemiology, and End Results (SEER) registry receiving photon-based radiation. The higher integral dose of IMRT and the potential difference in late toxicities make PBT particularly enticing for patients with sarcoma. Sarcomas disproportionately affect younger patients than do other common types of cancers, which makes these concerns particularly relevant ; sarcomas affect 1% to 2% of adults with cancer worldwide, compared with 11% of adolescents and young adults (15–29 years old) and 6% to 15% of pediatric children (<15 years old). Also, sarcomas are also commonly quite large, meaning that higher integral doses are needed to achieve comparable coverage. Finally, as previously mentioned, sarcomas occur throughout the body. For certain cancers arising at various anatomic sites (e.g., head and neck cancers; gastrointestinal cancers), growing bodies of literature support the benefit of PBT in sparing late toxicities; these findings may be extrapolated to sarcomas arising at the same anatomic sites.
Chordomas and chondrosarcomas
Sarcomas of the skull base, vertebral column, and sacrum are uncommon and pose particular management challenges. Given the anatomic locations, negative margin resections are difficult to achieve without significant morbidity, and radiation doses are limited by nearby critical structures. Chordomas and chondrosarcomas are the most common types of sarcomas to arise in these locations. Because of their similar behavior and central axis involvement, they are often described together.
Chordomas are rare tumors of bone that arise from notochordal remnants. Approximately 50% of chordomas are found in the sacrococcygeal region, whereas 35% occur in the base of skull and 15% within the vertebral column. Chondrosarcomas are a heterogenous group of bone sarcomas that arise from cartilaginous elements; with several subtypes of various biology and behavior, chondrosarcomas are the second-most-common primary bone sarcoma after osteosarcoma, and they are generally thought to have a better prognosis than chordomas. However, survival rates for both chordomas and chondrosarcomas have improved over the past two decades, with expected 5-year overall survival (OS) of about 80%.
The clinical presentation for both tumor types is similar and often is compressive in nature, with patients commonly experiencing pain or symptoms related to nerve dysfunction. Chordomas and chondrosarcomas are both locally aggressive tumors that pose challenges for management. Resection, if possible, is the treatment of choice. However, negative margins are difficult to achieve, and the rate of local recurrence is high. Therefore, radiation was integrated into the treatment approach as a way to improve outcomes. Notably, however, both histologies are considered quite radioresistant, requiring high doses of radiation to achieve disease control.
The radiation doses required for maximum tumor control often in excess of 70 Gy, exceed the tolerances of nearby structures , ; because of this small therapeutic ratio, chordomas and chondrosarcomas were some of the first tumors to be successfully treated by PBT, as it provided a way to deliver more precise radiation than was possible with its 2D/three-dimensional (3D) photon counterparts, thereby allowing the therapeutic ratio to be widened. One early study reported in 1982 by Suit and colleagues reported that doses of up to 76 cGE were delivered using PBT without significant morbidity. Several subsequent studies helped to establish the dose-response relationship of chordomas and chondrosarcomas. Rich and colleagues reported on 48 patients with chordoma treated at the Massachusetts General Hospital, of whom 32 received postoperative RT. In that study, none of the patients ( n = 18) receiving less than 60 Gy had no evidence of disease (NED), whereas 64% of the patients ( n = 14) who received greater than 60 Gy did achieve NED status. In a similar report Pearlman and Friedman, LC rates of 80% were achieved by using doses of 80 Gy compared with a 20% after doses of up to 60 Gy were delivered. , Finally, Keisch and colleagues reported on 12 patients treated with doses from 35 Gy to 70 Gy; all patients experienced relapse.
Since these early experiences with using PBT to treat chordomas and chondrosarcomas, doses have been increased, with noticeable corresponding gains in LC. A large retrospective review by Munzenrider and Liebsch of more than 600 patients with chordoma and chondrosarcoma of the base of skull and spine treated with PBT with doses ranging between 66 to 83 cGE revealed 5-year LC rates of 73% for skull base chordomas and 98% for chondrosarcomas. In a separate report on untreated spine chordomas receiving definitive mixed proton/photon RT to a median dose of 77.4 Gy, the 5-year LC rate was 80%.
Significant amounts of retrospective evidence of favorable LC are available for chordomas and chondrosarcomas after dose-escalated PBT; however, prospective findings remain rare. One of the few prospective studies done to date was reported by Delaney and colleagues, who undertook a phase II trial for patients with spine and paraspinal sarcomas. Fifty patients were treated with pre- or postoperative proton/photon RT, with or without resection. Twenty-five patients received less than 73 Gy, and the other half received 76.6 to 77.4 cGE. The 5-year LC rates were 94% for patients with de novo presentations compared with 81% for the entire cohort. ,
These findings, considered in their entirety, tell an interesting story. Chordomas and chondrosarcomas are relatively radioresistant tumors that are locally aggressive after surgery or moderate doses of radiation. However, higher doses of PBT can overcome this radioresistance and provide durable LC for tumors in some of the most anatomically difficult places to treat; for these tumor types, PBT is considered the standard of care.
Rhabdomyosarcoma
Another type of malignant soft tissue tumor is rhabdomyosarcoma, for which outcomes data after PBT are increasing. These tumors originate from skeletal muscle progenitor cells and are more common in children and adolescents or young adults than in older patients. Outcomes over the past few decades have improved with the increasing use of a multimodality approach to care, including radiation, which as noted earlier has an important role in LC. Because of its tendency to occur in younger patients, there is significant interest in using PBT to minimize late toxicity.
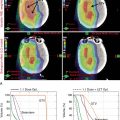
One of the first studies to report proton-associated outcomes in the treatment of rhabdomyosarcomas was published in 2005. The authors reported favorable tumor control outcomes for seven children with orbital rhabdomyosarcomas treated with PBT and chemotherapy, with excellent orbit function. When comparing conformal 3D photon plans to the delivered PBT plans, Yock and colleagues showed that PBT reduced the average radiation dose to all the orbital and central nervous system structures, with the largest reductions observed for the optic nerve (95%), pituitary (94%), orbital bone (93%), retina (91%), and chiasm (90%). A similar study by Cotter and colleagues evaluated outcomes in seven children treated with PBT for bladder/prostate rhabdomyosarcomas; five patients remained NED at last follow-up with acceptable toxicities. Dosimetric comparisons of the PBT plans with those generated for IMRT revealed that PBT provided significantly lower mean organ doses to the bladder, testes, femoral heads, growth plates, and pelvic bones.
These preliminary reports provided enough evidence to prompt a phase II study to prospectively evaluate PBT for patients with rhabdomyosarcoma. That trial enrolled 57 patients aged up to 21 years with rhabdomyosarcomas primarily classified as parameningeal ( n = 27) or orbital ( n = 13) to a median dose of 50.4 Gy (radiobiological effectiveness [RBE]) (range 36–50.4). After a median follow-up interval of 47 months (range 14–102), the 5-year event-free survival, OS, and LC rates were 69%, 78%, and 81%, with 5-year LC rates of 93% for low-risk disease and 77% for intermediate-risk disease. Eleven patients (13%) had a grade 3 acute toxicity attributable to RT, mostly odynophagia and dermatitis. Only three patients (7%) had a grade 3 late toxicity (cataract, chronic otitis, and retinopathy). The authors concluded that these findings indicated that PBT had a favorable toxicity profile compared with photons, while acknowledging the lack of a modern prospective IMRT cohort for comparison.
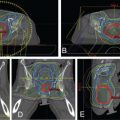
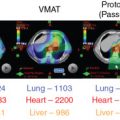
A subsequent study reported the comparison IMRT plans that were generated as part of the phase II prospective proton trial for patients with rhabdomyosarcoma. Although the target coverage (mean clinical target volume [CTV]: V 95 ) was similar between the PBT and the IMRT plans ( P = .82), the IMRT plans delivered a significantly higher integral dose regardless of the tumor site, with as much as a 3.5-fold increase for tumors in the orbit. The PBT plans also delivered significantly lower doses to 26 of the 30 critical structures evaluated. It will be interesting to determine if these dosimetric advantages translate into reductions in late toxicities when the long-term follow-up data have matured.
The role of radiation for pediatric patients with rhabdomyosarcomas is well defined, and for those who are to receive radiation, we consider PBT the standard modality. PBT should be given careful consideration when RT is recommended for adolescents and young adults. Importantly, though, practices vary in the use of radiation for rhabdomyosarcoma for older adult patients. We commonly treat rhabdomyosarcomas in such patients like other soft tissue sarcomas, delivering preoperative radiation in combination with surgery for improved LC. PBT should be considered for young patients in light of its dosimetric advantages.
Retroperitoneal sarcomas
Retroperitoneal sarcomas are often discussed together despite their heterogeneity because of the shared intraabdominal anatomic considerations. Retroperitoneal sarcomas constitute about 10% to 15% of all soft tissue sarcomas, with the most common histologies in adults being liposarcomas and leiomyosarcomas. Their management is somewhat controversial because of varying management practices, limited high-quality data, and high rates of tumor recurrence. Most series report 5-year LC rates of around 50%, which is why many practitioners advocate a combined-modality approach with the goal of improving outcomes. However, the role of radiation is controversial. No prospective, randomized data are available to compare the role of radiation and surgery versus surgery alone. Because of this lack of prospectively defined benefit, some question the utility of radiation, given its risk of toxicity.
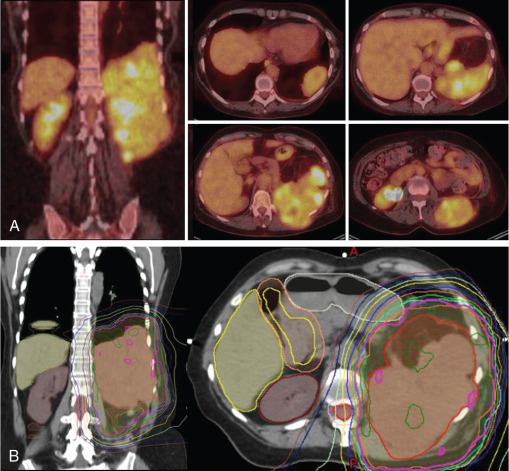
When radiation is incorporated into the treatment approach for retroperitoneal sarcomas, most, if not all, multidisciplinary high-volume sarcoma groups advocate preoperative treatment. , Several advantages exist when radiation is delivered preoperatively, including: (1) the target delineation is clear, (2) normal tissues and bowel are displaced, (3) treatment may increase the probability of a negative resection margin, and (4) cytoreduction of the tumor may occur. Therefore, when radiation is used in combination with surgery, it should ideally occur preoperatively, and in the absence of prospective data showing improved outcomes with combination therapy, patient selection is important to minimize toxicity. The properties that make PBT an enticing tool may improve the therapeutic ratio, particularly for sarcomas in the retroperitoneum ( Fig. 16.1 ).