Similar trial and Inclusion criteria
Agents
Number of patients
Months (95 % IC)
Number of patients
Months (95 % IC)
Patients receiving first-line TT
mOS from patients in IMDC
PFS from patients in IMDC
ADAPT [14]
Autologous dendritic cell immunotherapy (AGS-003) plus standard treatment of advanced RCC
1189
14.7 (13.3–16.5)
1174
5.6 (5.3–6.1)
Patients with an intermediate or poor risk disease and whose diagnosis to treatment interval was less than 1 year
TIVO-1[10]
Tivozanib vs. sorafenib
2117
24.8 (23.1–27.3)
2080
8.2 (7.8–8.6)
Patients who previously underwent nephrectomy
Patients who received second-line TT after at least one other VEGF-TT
INTORSECT [21]
Temsirolimus vs. sorafenib
1157
13.0 (12.2–14.7)
1151
3.9 (3.6–4.3)
Patients who have failed first-line Sunitinib
Patients who received third-line TT and were previously exposed to one VEGF inhibitor and one mTOR inhibitor
GOLD [22]
Dovitinib vs. sorafenib
147
18.0 (11.8–24.0)
140
4.4 (3.3–5.2)
Patients with mRCC in third line after failure to one VEGF inhibitor and one mTOR inhibitor
Once a patient surpasses the predicted survival milestone, survival projections may lose their accuracy. At this juncture, physicians have little guidance on how to counsel patients who wish to revisit the prognosis discussion. We tend to think of prognosis only at the beginning of treatment, but in fact prognosis is a very dynamic process. The concept of conditional survival is defined as the probability of surviving an additional amount of time after the patient has already survived a specific period of time.
This concept can provide practical information, because it accounts for the length of survivorship and changes in hazard rates over time. It is a clinically powerful measure that can dynamically adjust prognosis as months and years pass and can aid substantially in the counseling of subsequent treatment decisions.
For this purpose, 1673 patients were analyzed from the IMDC database with a median follow-up for alive patients of 20.1 months (IQR 9.0–34.4). A patient’s chance of living an additional two years (2-year conditional survival probability, conditioned on time survived regardless of whether still on the first-line targeted therapy) increases from 44 % (95 % CI 41–47) at 0 months of targeted therapy to 51 % (46–55) at 18 months since beginning targeted therapy. When stratified by the IMDC prognostic risk criteria at therapy initiation, 2-year conditional survival changed little in the favorable and intermediate groups, but in the poor-risk group, 2-year conditional survival improved from 11 % (8–15) at 0 months to 33 % (18–48) after 18 months of being on targeted therapy. It was demonstrated that conditional survival is a clinically useful prediction measure that adjusts prognosis of patients with mRCC on the basis of survival since treatment initiation or therapy duration and it might be especially relevant to adjust prognosis for poor-risk patients [23].
9.3 Prognostic Factors in Non-clear Cell RCC
Patients with rarer histologies of mRCC are often difficult to prognosticate. The largest cohort of patients to date, with non-clear cell RCC (nccRCC) histology treated with targeted therapy (20 % of patients), was investigated in the temsirolimus phase 3 study [25]. To characterize the applicability of the IMDC prognostic model and the survival outcome of patients with nccRCC who were treated with first-line VEGF and mTOR inhibitors, the IMDC criteria have been validated in both clear cell (ccRCC) and nccRCC for mRCC. Data on 2215 patients (1963 with ccRCC and 252 with nccRCC) treated with first-line VEGF and mTOR-targeted therapies were collected from the IMDC. mOS (12.8 vs 22.3 months; P < .0001) and time to treatment failure (TTF) (4.2 vs 7.8 months; P < .0001) were worse in nccRCC patients compared with ccRCC patients. The hazard ratio for death and TTF when adjusted for the prognostic factors was 1.41 (95 % CI, 1.19–1.67; P < .0001) and 1.54 (95 % CI, 1.33–1.79; P < .0001), respectively. The IMDC prognostic model reliably discriminated the three risk groups to predict mOS and TTF in nccRCC; the median mOS of the favorable, intermediate, and poor prognosis groups was 31.4, 16.1, and 5.1 months, respectively (P < .0001), and the median TTF was 9.6, 4.9, and 2.1 months, respectively (P < .0001). Even in the targeted-therapy era, the majority of nccRCC patients still have inferior clinical outcomes compared with patients with ccRCC [26]. There is actually no other modern prognostic model that has been assessed exclusively in advanced nccRCC. Moreover, the accuracy in prognosticating mOS was slightly higher than with the MSKCC risk model.
9.4 Prognostic Factors in Second-Line Therapy
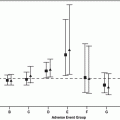
The IMDC criteria recently have been validated in second-line therapy. A total of 1021 patients who received second-line targeted therapy (TT) after progressing on 1st-line TT for mRCC at 19 centers were analyzed. For the patients who had immunotherapy (22 %) prior to their 1st TT, their second TT was examined (i.e., 3rd-line therapy). The median time on second-line TT was 3.9 months (range 0–76+). 871 (85 %) of patients had stopped secnd-line TT by the time of analysis. Median OS since second-line TT was 12.5 months (95 % CI: 11.3–14.3 months), with 369 (36.1 %) of patients remaining alive. Five out of six predefined factors in IMDC model (anemia, thrombocytosis, neutrophilia, KPS <80 %, and <1 year from diagnosis to treatment) measured at the time of second-line TT were independent predictors of poorer OS (HR between 1.39 and 1.58, p<0.05). Hypercalcemia was not statistically significant in multivariable analysis (p = 0.3008) likely due to the low incidence of hypercalcemia (9 %). The concordance index using all six prognostic factors was 0.70, and it was 0.66 compared with the three-factor-MSKCC model. When patients were divided into three risk categories using IMDC criteria, mOS was 35.8 months (95 % CI 28.3–47.8) in the favorable risk group (n = 76), 16.6 months (95 % CI 14.9–17.9) in the intermediate risk group (n = 529), and 5.4 months (95 % CI 4.7–6.8) in the poor risk group (n = 261) [24].
Radiologic evaluation of mRCC is also changing in TT era, and the assessment with contrast-enhanced computed tomography (CECT) is an example. About 84 standard CECT examinations with mRCC on first-line sunitinib or sorafenib therapy were retrospectively evaluated comparing morphology, attenuation, size, and structure (MASS) criteria; response evaluation criteria in solid tumors (RECIST); size and attenuation CT (SACT) criteria; and modified Choi criteria. The objective response to therapy was compared with clinical outcomes including time to progression (TTP) and disease-specific survival. A favorable response according to MASS criteria had a sensitivity of 86 % and specificity of 100 % in identifying patients with a good clinical outcome (i.e., progression-free survival of > 250 days) versus 17 % and 100 %, respectively, for RECIST partial response. The objective categories of response used by MASS criteria—favorable response, indeterminate response, and unfavorable response—differed significantly from one another with respect to TTP (p < 0.0001, log-rank test) and disease-specific survival (p < 0.0001, log-rank test). Furthermore, the use of MASS criteria for imaging response assessment showed high interobserver agreement and predicted disease outcome in patients with metastatic RCC on TT [25].
Other factors that have been identified to confer a poorer prognosis to mRCC patients are the presence of bone and/or liver metastases, compared with other metastatic sites. A retrospectively review from 2027 patients from the IMDC was conducted for this purpose. The presence of bone and liver metastases were 34 % and 19 % overall, respectively. For bone metastases (BMs), when stratified by IMDC risk groups was 27 %, 33 %, and 43 % in the favorable-, intermediate-, and poor-risk groups, respectively (p < 0.001). For liver metastases (LMs), the presence was higher in the poor-risk patients (23 %) compared with the favorable- or intermediate-risk groups (17 %) (p = 0.003). When patients were classified into four groups based on the presence of BMs and/or LMs, the hazard ratio, adjusted for IMDC risk factors, was 1.4 (95 % CI, 1.22–1.62) for BMs, 1.42 (95 % CI, 1.17–1.73) for LMs, and 1.82 (95 % CI, 1.47–2.26) for both BMs and LMs compared with other metastatic sites (p < 0.0001). The prediction model performance for mOS was significantly improved when BMs and LMs were added to the IMDC prognostic model (likelihood ratio test p < 0.0001). These two metastatic sites can be added for the risk stratification of patients with mRCC and improve the predictive accuracy of the IMDC criteria [26].
Data exists for other laboratory values, like hyponatremia and neutrophil to lymphocyte ratio (NLR) > 2.5 which are independently associated with a worse outcome in mRCC patients treated with VEGF and mTOR-targeted agents. Hyponatremia has been associated with poor survival in many solid tumors and a recent study sought to investigate this association on treatment outcomes in mRCC patients treated with contemporary TT. Hyponatremia was found in 14.6 % of 1661 mRCC patients from the IMDC. On univariate analysis, hyponatremia was associated with shorter mOS (7.0 vs 20.9 months), shorter TTF (2.9 vs 7.4 months), and lower disease control rate (DCR) rate (54.9 % vs 78.8 %) (p< 0.0001 for all comparisons). In multivariate analysis, these effects remain significant (hazard ratios: 1.51 [95 % CI, 1.26–1.80] for mOS and 1.57 [95 % CI, 1.34–1.83] for TTF [30]. To evaluate the prognostic value of hematologic parameters in patients with mRCC, a retrospective review of 157 patients aimed to explore the association between NLR and platelet to lymphocyte ratio (PLR) with response to tyrosine kinase inhibitor (TKI) treatment in mRCC. On multivariable analysis NLR > 2.5 and Karnofsky Performance Status (KPS) < 90 % were associated with a lower likelihood of response. PLR did not retain association with response in multivariable analysis [31]. Unfortunately adding these factors to our current prognostic models has not led to substantially better c-indices.
9.5 Predictive Factors
Compared to prognostic factors, there are few predictive factors described in mRCC patients, aside from TT-induced hypertension and other side effects of anti-VEGF therapy (hypothyroidism and hand-foot syndrome reaction (HFSR)). Prediction is used to predetermine the efficacy of a treatment and ideally aid in treatment selection. Unfortunately, compared to other malignancies (breast, lung, colon, and melanoma) mRCC lacks effective predictive factors at this moment. We lack validated, reproducible markers to accurately predict response.
Sunitinib-associated hypertension (HTN) is associated with improved clinical outcomes. Sunitinib-induced HTN is associated with improvement in clinical outcomes (objective response rate, PFS, and mOS). It was reported that patients with mRCC and sunitinib-induced HTN defined by a maximum SBP of ≥140 mmHg had better outcomes than those without treatment-induced HTN (objective response rate: 54.8 % vs 8.7 %; median PFS = 12.5 months, 95 % confidence interval [CI] = 10.9–13.7 vs 2.5 months, 95 % CI = 2.3–3.8 months; and mOS =: 30.9 months, 95 % CI = 27.9–33.7 vs 7.2 months, 95 % CI = 5.6–10.7 months; P < .001 for all). Similar results were obtained when comparing patients with vs without sunitinib-induced HTN defined by a maximum DBP of ≥ 90 mmHg [32]. Comparable information has been observed with other anti-VEGF agents [27, 28].
It is suggested that sunitinib-induced hypothyroidism is also associated with improved response rates to TT. There was a statistically significant association between the occurrence of hypothyroidism during treatment and the rate of objective response: hypothyroid mRCC patients treated with targeted therapy displayed higher ORR (28.3 vs 3.3 %, respectively; P < 0.001) and prolonged median mOS (not reached vs. 13.9 months, respectively; P = 0.016) than euthyroid patients. In multivariate analysis, the development of subclinical hypothyroidism was identified as an independent predictor of survival (HR 0.31; P = 0.014) [29, 30].
Attention should be paid to the potential emergence of sorafenib-induced HFSR because it has been reported it could become a predictive marker of clinical outcome in mRCC patients. In a recent retrospective review, 36 Japanese mRCC patients treated with sorafenib were analyzed. A sorafenib-induced HFSR was observed at a significantly higher rate in patients in the favorable-risk group in the MSKCC model criteria and with Eastern Cooperative Oncology Group Performance Status of one or less, prior nephrectomy, higher hemoglobin, lower lactate dehydrogenase, and lower C-reactive protein. The mean best tumor response was significantly better in the group with HFSR (16.7 %) than that in the group without it (17.9 %; P < 0.001). The median progression-free survival was significantly longer in the group with HFSR (4.6 months) compared to the group without it (1.5 months; P = 0.002). In multivariate analysis, only HFSR was shown to be a predictive factor of progression-free survival (hazard ratio 0.312, P = 0.010) [31].
All of these predictive factors (HTN, hypothyroidism, HFSR) for VEGF TT are somewhat helpful however they are not useful for treatment selection, because patients need to start the drug first and then develop toxicities for prediction. Many other described predictive factors in mRCC awaiting validation include MET [32], PDL-1 [33] and PBRM1/BAP1 [34].
Recently, it was reported that certain biomarkers could be considered as molecular entry criteria for prospective clinical studies in selecting mTOR or VEGFR inhibitors. Potential correlations between somatic mutations and treatment efficacy in RECORD-3 trial (a randomized phase 2 trial comparing first-line everolimus then sunitinib with first-line sunitinib then everolimus at progression in 471 treatment-naïve mRCC patients) [35]. Polybromo-1 (PBRM1) mutations were associated with longer PFS within everolimus (median PFS 11.1 vs 5.3 months; unadjusted p = 0.0031). Patients with PBRM1 mutations (41 % of the cohort) derived comparable PFS benefit from everolimus vs. sunitinib. Lysine demethylase 5C (KDM5C) mutations were associated with longer PFS within sunitinib (median PFS 20.6 vs 8.4 months; unadjusted p = 0.0511). However, it needs a prospective validation [36].
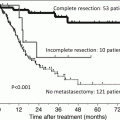
The benefit of cytoreductive nephrectomy (CN) for overall survival is unclear in patients with synchronous mRCC in the era of TT, and it is not well understood if CN should remain a part of the standard treatment protocol. For this purpose, retrospective data from 1658 patients with synchronous mRCC from the IMDC were used to compare 982 mRCC patients who had a CN with 676 mRCC patients who did not. All patients received targeted therapy, with most receiving first-line sunitinib (72 %). The results demonstrated that patients who had CN had better IMDC prognostic profiles versus those without (favorable, intermediate, or poor in 9 %, 63 %, and 28 % vs 1 %, 45 %, and 54 %, respectively). Fewer CN patients had non-clear cell pathology, bone metastases, and liver metastases, but CN patients had more sarcomatoid features. The median OS of patients with CN versus without CN was 20.6 versus 9.5 mo (p < 0.0001). When adjusted for IMDC criteria to correct for imbalances, the HR of death was 0.60 (95 % confidence interval, 0.52–0.69; p < 0.0001). The authors concluded that CN is beneficial in synchronous mRCC patients treated with targeted therapy, even after adjusting for prognostic factors. However, patient selection is very important. Patients with estimated survival times < 12 months or four or more IMDC prognostic factors may not benefit from CN. Perhaps it is because their prognosis is so limited that they should have started on targeted therapy as soon as possible instead of delaying it with cytoreduction for which the patient will take time to recover from. This information may aid in patient selection as we await results from randomized controlled trials [43]. Although we await the results of randomized trials to truly determine the benefit of cytoreductive nephrectomy, the use of the IMDC prognostic factors may help clinicians decide which patient stands to benefit from surgery the most.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree