Direct oral factor IIa inhibitors represent a new class of anticoagulants for the prevention and treatment of venous and selected arterial thomboembolisms. Dabigatran etexilate is the most studied and promising of the oral direct thrombin inhibitors. Preclinical and early-phase clinical studies show it to have a predictable and reliable pharmacokinetic and pharmacodynamic profile, whereas advanced phase 3 trials prove it to be noninferior to traditional anticoagulants in selective conditions for the prevention and treatment of venous and arterial thromboembolism. Other advantages of this drug, including a lack of interaction with cytochrome P450 enzymes or with food and drugs, rapid onset of action, good safety profile, lack of need for routine monitoring, broad therapeutic window, and fixed-dose administration, make this a competitive oral anticoagulant.
From the early 1940s, the vitamin K antagonists (VKAs; warfarin, phenprocoumon, acenocoumarol) have been the only oral anticoagulants available for the long-term treatment of venous and selected arterial thromboembolic disease. In the last decade, new classes of oral anticoagulants have emerged and undergone extensive study. At present, the oral direct thrombin inhibitors and oral factor Xa inhibitors are the most promising agents and the most advanced in study. Because of their size, structure, and pharmacokinetics, these agents have specific advantages over the VKAs and they may also be suited to replace the heparin anticoagulants for selected conditions. Specifically, they directly inhibit their target enzyme, they are small molecules and can interact with their target in the fibrin clot or prothrombinase complex, respectively; their pharmacokinetics are predictable; and they have few if any drug or food interactions such that monitoring is not required. This article reviews the oral direct thrombin inhibitors and the class of new anticoagulants of which ximelagatran is the first example of an alternative to the VKAs.
Ximelagatran
Ximelagatran (Exanta) was the first oral direct thrombin inhibitor to be developed. Although it was found to be an effective therapy for the prevention and treatment of venous thromboembolism (VTE) and stroke prevention in atrial fibrillation (AF), it was associated with hepatotoxicity and removed from the market in Europe, and was never approved in North America. However, because it served as a template for the study of other direct thrombin inhibitors, it is briefly reviewed here. Ximelagatran, the inactive prodrug for melagatran, has approximately 80 times greater membrane permeability when taken orally, which makes it a rapidly absorbable drug. Once absorbed, ximelagatran is converted to melagatran via hydrolysis of the ester group and reduction of the hydroxyl group. It reaches peak plasma levels in 2 to 3 hours, has a half-life of 3 to 5 hours, and is administered as a twice-daily dose drug. Ximelagatran has minimal drug interactions and a predictable response, therefore not requiring monitoring. Its primary route of excretion is via the kidneys. Ximelagatran has been studied for the prevention of venous thromboembolism following major orthopedic surgery, treatment of acute and chronic venous thromboembolism, stroke prevention in AF, and secondary prevention of cardiovascular events after an acute myocardial infarction.
Prevention of Venous Thromboembolism in Orthopedic Surgery
Several phase 3 trials in total hip and knee replacement surgery showed ximelagatran to have excellent efficacy and safety in VTE prevention following orthopedic surgery. The MElagatran for THROmbin inhibition in orthopedic surgery (METHRO) III study of 2800 patients undergoing hip or knee replacement showed that postoperative administration of 3 mg subcutaneous melagatran followed by ximelagatran 24 mg twice a day for 8 to 11 days had an efficacy comparable to enoxaparin initiated preoperatively (total VTE 31% vs 27.3%, respectively; P = .053). Rates of severe bleeding between ximelagatran (1.4%; 95% confidence interval [CI], 0.9%–2.2%) and enoxaparin (1.7%; 95% CI, 1.1%–2.5%) did not differ. In the expanded prophylaxis evaluation surgery (EXPRESS) study, administration of melagatran 2 mg preoperatively followed by a 3 mg dose postoperatively, and then ximelagatran 24 mg twice a day starting the next day versus preoperative initiation of enoxaparin demonstrated significantly lower rates of proximal deep vein thrombosis (DVT) and pulmonary embolism (PE) (2.3% vs 6.3%, respectively; P <.001) and total VTE (20.3% vs 26.6%, respectively; P <.0014). However, severe bleeding was greater with melagatran/ximelagatran than with enoxaparin (3.1% vs 1.2%, respectively; P <.001).
In a North American trial of 1800 patients undergoing total hip replacement, ximelagatran 24 mg twice a day and enoxaparin 30 mg twice a day, both started postoperatively, failed to meet noninferiority criteria (total VTE 7.9% vs 4.6%, absolute difference, 3.3%; 95% CI, 0.9%–5.7%). Major bleeding events were similar in both groups. In the Exanta Used To Lessen Thrombosis (EXULT) A trial, 1800 patients undergoing total knee replacement surgery received ximelagatran 24 mg or 36 mg twice a day started postoperatively and were compared with postoperative administration of warfarin (adjusted for international normalized ratio [INR] 1.8–3.0). The 36 mg dose of ximelagatran achieved lower rates of VTE and all-cause mortality compared with warfarin (20.3% vs 27.6%, respectively; P = .003). Major bleeding occurred in 0.8% of both ximelagatran groups versus 0.7% in the warfarin group. A follow-up study, EXULT B, confirmed the benefit of the higher ximelagatran dose of 36 mg twice daily compared with warfarin (adjusted for INR = 2.5) after total knee replacement in 2300 patients. Total venous thromboembolism and all-cause mortality combined occurred in 22.5% vs 31.9%, respectively ( P <.001).
Stroke Prevention in AF
Stroke prevention with the oral direct thrombin inhibitor ximelagatran compared with warfarin in patients with nonvalvular atrial fibrillation (SPORTIF) III AND SPORTIF V studies compared ximelagatran 36 mg twice a day with warfarin (target INR = 2.5) for prevention of stroke and systemic thromboembolism in more than 7300 patients with nonvalvular AF and additional stroke risk factors. SPORTIF III was a randomized, open-label trial whereas SPORTIF V was blinded. In SPORTIF III, an intention to treat analysis demonstrated an absolute risk reduction of 0.7% (95% CI, −0.1–1.4; P = .10) favoring ximelagatran. There was no significant difference in major bleeding. Elevations in alanine aminotransferase (ALT) levels greater than 3 times the upper limit of normal were experienced by 6% of patients receiving ximelagatran compared with 1% of the patients in the warfarin group ( P <.001), and 3.4% of ximelagatran patients had ALT elevations greater than 5 times the upper limit of normal. Elevations in ALT levels in the ximelagatran group occurred in the first 2 to 6 months of the start of therapy.
In SPORTIF V (3922 patients), the primary end points occurred in 51 patients in the ximelagatran group and in 37 patients in the warfarin group (1.6% vs 1.2%, respectively, absolute difference 0.45%; 95% CI, −0.13%–1.03%; P <.001). This study demonstrated ximelagatran to be noninferior to warfarin for the prevention of stroke and systemic embolism in relatively high-risk patients with AF. Significantly more minor and major hemorrhages occurred in SPORTIF III in patients receiving warfarin than in those receiving ximelagatran (relative risk reduction [RRR] of ximelagatran, 14%; 95% CI, 4–22, P <.007). Similarly, SPORTIF V demonstrated significantly fewer major and minor hemorrhagic events in the group receiving ximelagatran versus the group receiving warfarin (RRR, 21%; 95% CI, −14%–6.0% per year; P <.001). Combined data from both studies showed a reduced relative risk of major hemorrhage, including intracranial hemorrhage, for ximelagatran versus warfarin (RRR 0.74; 95% CI, 0.57–0.97) and a reduced risk of minor and major hemorrhage (32.0% and 39.1% for the ximelagatran group and the warfarin group, respectively). Elevations of ALT greater than 3 times the upper limit of normal were experienced by 6% of patients in the ximelagatran group compared with 0.8% of patients in the warfarin group ( P <.001). One patient died as a result of liver failure 20 days after cessation of ximelagatran use and was found to have hepatic necrosis on biopsy, while another patient’s ALT was greater than 11 times the upper limit of normal and consequently suffered from a fatal gastrointestinal hemorrhage.
Secondary Prevention of VTE
The treatment of high-density lipoproteins to reduce the incidence of vascular events (THRIVE) trial was a 6-month, randomized, double-blind, noninferiority study in 2490 patients with acute venous thromboembolism receiving oral ximelagatran 36 mg twice daily versus enoxaparin/warfarin. Recurrent VTE was found in 26 ximelagatran patients (2.1%) versus 24 enoxaparin/warfarin patients (2.0%) meeting prespecified noninferiority criteria (95% CI, −1.0%–1.3%). Rates of major bleeding were similar. ALT was greater than 3 times the upper limit of normal in 9.6% of ximelagatran patients versus 2.0% of warfarin patients.
An extension of the THRIVE III study assessed prolonged prophylaxis with ximelagatran 24 mg twice daily versus placebo after 6 months of standard therapy or ximelagatran therapy. Symptomatic, recurrent venous thromboembolism occurred in 12 patients in the ximelagatran group versus 71 patients in the placebo group (hazard ratio [HR], 0.16; 95% CI, 0.09–0.30; P <.001). Elevations of ALT greater than 3 times the upper limit of normal were seen in a larger portion of individuals assigned to the ximelagatran group versus the placebo group (6.4% vs 1.2%, respectively; P <.001).
Treatment in Acute Coronary Syndrome
Efficacy and safety of oral direct thrombin inhibitor ximelagatran in patients with recent myocardial damage (ESTEEM) was a placebo-controlled, double-blind, phase 2 dose-guiding investigation of ximelagatran and aspirin (ASA) in individuals who had recent ST-elevation or non-ST elevation myocardial infarctions. The primary end point was the occurrence of death, nonfatal myocardial infarction, and severe recurrent ischemic events after a recent myocardial infarction. All patients received ASA and either oral ximelagatran at varying doses (24 mg, 36 mg, 48 mg, or 60 mg twice daily) or placebo for a total of 6 months. At 6 months, ximelagatran was superior to placebo in reducing the cumulative risk of reaching an adverse outcome (16.3% for placebo vs 12.7% in the combined ximelagatran groups; HR, 0.76; 95% CI, 0.59–0.98, P = .036). There was no difference in efficacy between the various doses of ximelagatran. An ALT level greater than 3 times the upper limit of normal was more frequent in the ximelagatran groups versus the placebo group ( P <.0001), and 4 patients developed an elevated serum bilirubin and ALT.
Cessation of Ximelagatran Usage
Although ximelagatran was demonstrated to be as effective as traditional anticoagulants in the prevention and treatment of venous thrombosis and stroke prevention in AF, a signal of hepatotoxicity led the US Food and Drug Administration (FDA) agency to re-evaluate the benefits of long-term use of the drug. In the majority of patients who experienced transient elevations in liver enzymes, these levels returned to normal either spontaneously or with cessation of ximelagatran therapy. Further investigations demonstrated that even regular monitoring of hepatic enzymes while on ximelagatran could not predict the severity of possible hepatic injury, and a 35-day study demonstrated that severe hepatic injury could result even after cessation of ximelagatran use. Thus, after careful investigation and analysis of completed trials with ximelagatran, it was determined that the risks of the drug outweighed its benefits, and it was ultimately denied by the FDA in 2004.
Dabigatran etexilate
Dabigatran etexilate (PRADAXA, BIBR 1048) is the oral prodrug of dabigatran (BIBR 953), a low molecular weight molecule that acts as a specific, potent, and reversible direct thrombin inhibitor (DTI). Approved in Canada and Europe in 2008 for the prevention of VTE after elective total hip replacement and/or total knee replacement, dabigatran etexilate is not yet FDA-approved in the United States. The efficacy and safety of dabigatran etexilate compared with standard anticoagulant therapy is currently in phase 3 of development. The extensive RE-VOLUTION trial program supported by Boehringer Ingelheim includes multiple phase 3 trials that have focused on the use of dabigatran etexilate for primary VTE prevention after hip and knee surgeries, treatment of acute DVT and/or PE and their secondary prevention, prevention of stroke and systemic embolism in patients with nonvalvular AF, and secondary prevention of cardiac events in patients with acute coronary syndromes (ACS) ( Table 1 ).
| Study | Indication | Number of Patients | Dabigatran Etexilate Regimen | Comparator Regimen | 1° Outcome Measure | 1° Outcome Measure Results (%) | Serious Bleeding (%) |
|---|---|---|---|---|---|---|---|
| RE-MOBILIZE | VTE prevention after TKR | 2615 | 150 or 220 mg once-daily (starting with a half-dose 6–12 h postop) for 12–15 d | Enoxaparin 30 mg twice-daily starting postop for 12–15 d | Total VTE and all-cause mortality | Dabigatran (150 mg), 33.7% Dabigatran (220 mg), 31.1% Enoxaparin, 25.3% Both dabigatran etexilate doses failed to show noninferiority to enoxaparin | Dabigatran (150 mg), 0.6% Dabigatran (220 mg), 0.6% Enoxaparin, 1.4% |
| RE-MODEL | VTE prevention after TKR | 2076 | 150 mg or 220 mg once-daily (starting with a half-dose 1–4 h postop) for 6–10 d | Enoxaparin 40 mg every day (starting the evening before surgery) for 6–10 d | Total VTE and all-cause mortality | Dabigatran (150 mg), 40.5% Dabigatran (220 mg), 36.4% Enoxaparin, 37.7% Both doses of dabigatran etexilate were noninferior to enoxaparin for VTE prevention | Dabigatran (150 mg), 1.3% Dabigatran (220 mg), 1.5% Enoxaparin, 1.3% |
| RE-NOVATE | VTE prevention after THR | 3494 | 150 or 220 mg once-daily (starting with a half-dose 1–4 h postop) for 28–35 d | Enoxaparin 40 mg once-daily preop for 28–35 d | Total VTE and all-cause mortality | Dabigatran (150 mg), 8.6% Dabigatran (220 mg), 6% Enoxaparin, 6.7% Both doses of dabigatran etexilate were noninferior to enoxaparin for VTE prevention | Dabigatran (150 mg), 1.3% Dabigatran (220 mg), 2% Enoxaparin, 1.6% |
| RE-COVER | Treatment in acute VTE | 2564 | 150 mg twice-daily (starting after 8–11 d of parenteral therapy) for 6 mo | Warfarin (dose adjusted for INR 2.0–3.0) after 8–11 d of initial parenteral anticoagulation | Recurrent VTE and related deaths | Dabigatran, 2.4% Warfarin, 2.1% Dabigatran etexilate is noninferior to warfarin in the prevention of recurrent or fatal VTE | Dabigatran, 16.1% Warfarin, 21.9% |
| RE-LY | Prevention of stroke in nonvalvular atrial fibrillation | 18,113 | 110 mg or 150 mg twice-daily for 2 y | Warfarin (dose adjusted for INR 2.0–3.0) for 2 y | Stroke and systemic embolism | Dabigatran (110 mg), 1.53%/y Dabigatran (150 mg), 1.11%/y Warfarin, 1.69%/y Both doses of dabigatran etexilate were noninferior to warfarin | Dabigatran (110 mg), 0.12%/y Dabigatran (150 mg), 0.10%/y Warfarin, 0.38%/y |
Studies of dabigatran etexilate in healthy volunteers have identified key clinical advantages of this drug, including a rapid conversion to its active form dabigatran, which has early peak plasma concentrations and a half-life between 12 and 17 hours, a predictable and consistent anticoagulant effect with low potential for drug-drug or drug-food interactions, and a lack of (or induction) metabolism by the cytochrome P450 enzymes. All of these characteristics allow dabigatran etexilate to be given as a fixed, once- or twice-daily dose without the need for coagulation monitoring.
Absorption and Metabolism of Dabigatran Etexilate and Dabigatran
Dabigatran is a 472 Da peptidomimetic that directly inhibits thrombin by binding to its active site via ionic interactions. It is a highly basic and hydrophilic compound and therefore has poor intestinal absorption after oral administration. To increase its bioavailability, the addition of a less basic, lipophilic side chain, N -hexyl carbamate ester, led to the development of a 628 Da, gastrointestinally absorbed double prodrug dabigatran etexilate. After oral administration, the prodrug is rapidly absorbed in the stomach and small intestine and converted to its active form, dabigatran, via 2 intermediate metabolites (BIBR 951 and BIBR 1087) through cleavage of the lipophilic ester moiety by rapidly acting serine esterase-catalyzed hydrolysis. The advantage of this ethyl ester-structured prodrug compared with other DTIs with hydroxylamine-structured prodrugs, such as ximelagatran, is that the P450 (CYP) isoenzymes or other oxidoreductases are not significantly involved in the proteolytic reactions that convert dabigatran etexilate to dabigatran. Also, in vitro studies reveal that dabigatran does not inhibit the cytochrome P450 enzymes. Therefore, the potential for drug-drug interactions is low.
The bioavailability of dabigatran after oral absorption is low (6%–7%) and is independent of the dose of the prodrug. The linear relationship between the prodrug dose and plasma concentrations of dabigatran excludes saturable first-pass elimination as the cause for low bioavailability. Dose-escalation studies have shown that dabigatran plasma concentrations increase in a dose-proportional manner and therefore, high doses of dabigatran etexilate are necessary to achieve adequate plasma concentrations. The absorption and metabolism of dabigatran etexilate is also dependent on an acidic microenvironment. The original tablet form used in the Boehringer Ingelheim Study in thrombosis (BISTRO) 1a trial resulted in low plasma concentrations of dabigatran after the first dose in 20% of the patients undergoing total hip replacement surgery, causing a high interindividual variability in the pharmacokinetic parameters. Therefore, a new capsule formulation was developed, which contains multiple small pellets containing a tartaric acid core and coated with dabigatran etexilate. Slight variations in intrinsic gastric pH have not significantly affected the extent of absorption of dabigatran etexilate. As a result of the improved dissolving characteristics, earlier postoperative administration of dabigatran etexilate has been possible, and an immediate onset of absorption and peak plasma concentrations at 6 hours after administration has been observed.
After absorption of dabigatran etexilate, bioconversion to dabigatran occurs in enterocytes, hepatocytes, and portal vein. Glucuronic acid is conjugated with 20% of dabigatran to yield pharmacologically activated glucuronide conjugates that have almost exact properties as free, unconjugated dabigatran, and therefore have no impact on the efficacy of dabigatran. Once metabolized to dabigatran, the prodrug and its intermediate metabolites, BIBR 951 and BIBR 1087, are trace detectable in the plasma of healthy subjects. The absorption and metabolism of dabigatran etexilate has also been tested in patients with hepatic impairment. In patients with moderate hepatic impairment (Child-Pugh Category B), the bioconversion rate from dabigatran etexilate to its active form via the intermediate BIBR 1087 was slightly slower than that in matched healthy control subjects. The area under the plasma concentration-time curves (AUCs) for dabigatran etexilate and its intermediates BIBR 1087 and BIBR 951 relative to the AUC of dabigatran in plasma were 1.8%, 5.9%, and 0.3%, respectively, compared with AUCs of less than 0.4% in the healthy control group.
Studies using radiolabeled ( 14 C) oral dabigatran etexilate and 14 C intravenous dabigatran show that dabigatran is the predominant compound in plasma, urine, and feces, and that more than 80% of systemically available dabigatran is renally excreted. The remainder undergoes conjugation with glucuronic acid to form acylglucuronides, which are excreted via the bile. Unchanged dabigatran accounts for approximately 77% of the intravenous dose of dabigatran and the remainder are glucuronide conjugates. Dabigatran also has moderate tissue distribution, because the volume of distribution of dabigatran given by intravenous infusion is 60 to 70 L.
Pharmacokinetic Characteristics of Dabigatran Etexilate and Dabigatran
The pharmacokinetic profile (PK) of dabigatran etexilate and dabigatran has been studied in healthy subjects, both young and old, and in patients undergoing hip arthroplasty or in those with AF at risk of cardioembolic events. Studies of healthy subjects not undergoing surgery reveal that dabigatran is well tolerated and that its time to peak plasma concentrations (C max ) is achieved within 1 to 3 hours following either single or multiple doses of oral administration of dabigatran etexilate, regardless of age or gender. The onset of absorption is also immediate in healthy patients undergoing total hip replacement surgery; however, the rate and degree of absorption is slightly decreased in the first 24 hours postoperatively (C max [75.8 ng/mL] is achieved at 6 hours). Despite these results found among surgical patients, the extent of absorption (mean AUC 0–24 h ) is acceptable and comparable to values obtained in healthy volunteers (962 and 904 ng/mL, respectively). The slower time progression to C max with an unchanged AUC may help decrease the risk of bleeding in patients in the early postoperative period. Studies in the orthopedic and elderly populations reveal that coadministration of pantoprazole reduces overall drug exposure by 20% to 25%, although this may not be clinically relevant. Food prolongs the time to peak plasma dabigatran levels by 2 hours without effect on its extent of absorption (AUC). In addition, the PK is not affected by the use of comedications, such as opioids, diclofenac, atorvastatin, or digoxin. However, a reduced dose of dabigatran etexilate is recommended when coadministered with amiodarone, because studies in patients taking amiodarone who underwent orthopedic procedures showed an increased C max and AUC for dabigatran.
Early clinical studies also reveal time- and dose-independent pharmacokinetics of dabigatran. There is a proportional and steady increase in the C max and AUC with increasing single or multiple doses of dabigatran etexilate. After absorption, a rapid distribution phase occurs followed by a prolonged elimination time. The mean plasma terminal half-life of dabigatran in healthy young and elderly adults is 12 to 14 hours, independent of dose, and accordingly, steady-state concentrations are attained in 3 days with multiple dosing and without evidence of significant accumulation. Although 35% of dabigatran is bound to plasma proteins, it is unlikely that displacement interactions affect its pharmacokinetics or pharmacodynamics.
Renal insufficiency is natural and common in the elderly, and age-related reduction in creatinine clearance (CL CR ) is associated with decreased elimination of dabigatran. Increased trough concentrations have been reported in patients with CL CR less than 50 mL/min. The bioavailability of dabigatran is 1.7 to 2.0 times greater in elderly subjects than in young healthy subjects of the same sex. Also, the extent of exposure (AUC) to dabigatran is 2.7- and 6-fold greater with moderate (CL CR 30–50 mL/min) or severe renal insufficiency (CL CR 10–30 mL/min, respectively). Therefore, dose reduction of dabigatran etexilate is appropriate in the elderly or patients with renal insufficiency (CL CR <50 mL/min). Gender-dependent differences in CR CL with downstream differences in dabigatran exposure are also apparent (mean AUC ∼3%–19% higher in elderly women) yet do not require modifications in dosing. Although dabigatran is dialyzable, it is contraindicated in patients with severe renal impairment (CL CR <30 mL/min).
Pharmacodynamic Characteristics of Dabigatran Etexilate and Dabigatran
In vitro studies show that dabigatran rapidly and reversibly inhibits clot-bound and free thrombin in a concentration-dependent manner with an inhibition constant (K i ) of 4.5 nmol/L. Its specificity is highly selective for thrombin over other serine proteases. In addition, dabigatran inhibits thrombin-activated human platelet aggregation with a half maximal inhibitory concentration (IC 50 ) value of 10 nmol/L. Tissue factor–induced thrombin generation in human platelet-poor plasma is also inhibited by dabigatran in a concentration-dependent manner.
Different clot-based assays have been used to assess the pharmacodynamic effects of dabigatran. The activated partial thromboplastin time (aPTT) assay measures the time to thrombin generation and clot formation via the intrinsic pathway, which involves fibrinogen and factors II, V, X, VIII, IX, XI, and XII, whereas the PT assay represents the clotting time in the extrinsic pathway involving fibrinogen and factors II, V, X, and VII. The thrombin time (TT) and ecarin clotting time (ECT) tests also measure the time to clot formation by directly measuring the activity of thrombin and the inhibitory activity of the DTI. In the latter test ecarin is used, which converts prothrombin into its intermediate meixothrombin, another substrate that is inhibited by dabigatran. It has been shown that prolongation of clotting times (aPTT, PT, TT, ECT) occurs in parallel with increasing concentrations of dabigatran, and that peak clotting times coincide with the C max of dabigatran (2 hours after oral administration). This observation suggests that the effects of dabigatran on thrombin inhibition are rapid and without a time delay. Similarly, at the time of the first half-life (12 hours after oral administration) of dabigatran, the prolongation of blood coagulation returned to 50% of the maximum effect.
TT and ECT both exhibit a linear dose-response relationship with therapeutic concentrations of dabigatran; however, because 50-fold prolongations of the TT have been reported, the TT coagulation test may prove to be too sensitive in the clinically relevant plasma concentration range. The ECT assay may be a more useful test for measuring thrombin inhibition and has been reported as having adequate sensitivity and precision with predictable and reproducible results. Prolongation of the aPTT also occurs with increasing plasma concentrations of dabigatran but in a nonlinear manner. A plateau in aPTT prolongation is noted at high dabigatran concentrations (>400 ng/mL). These observations suggest that the aPTT coagulation assay may not be acceptable as a precise measurement of anticoagulation, particularly at high doses of dabigatran. Prothrombin time is not affected by dabigatran at clinically relevant plasma concentrations. The intersubject variability of aPTT, PT, TT, and ECT is low, with a coefficient of variation ranging from 6% to 11%. Given the predictable pharmacokinetics and pharmacodynamics of dabigatran and its lack of drug-drug interactions, careful coagulation monitoring is not needed.
Prevention of Venous Thromboembolism After Orthopedic Surgery
The preclinical and early-phase clinical trials established predictable pharmacokinetic and pharmacodynamic profiles for dabigatran. Two phase 2 clinical trials, involving more than 2000 patients, were conducted to examine the safe therapeutic range of dabigatran etexilate and its optimal dosing regimens for prevention of venous thromboembolism following orthopedic surgery. In both studies, the primary efficacy outcome was the rate of thromboembolic events during the treatment period, while the primary safety outcome was the incidence of major bleeding.
BISTRO trials consisted of 2 large studies that investigated the dose-response relationships and the safety and efficacy of dabigatran etexilate in VTE prevention for patients undergoing total elective hip and knee replacement surgery. BISTRO 1a was an open-label, multicenter dose-escalation study of dabigatran etexilate in 314 patients undergoing hip replacement surgery. Patients received 1 of 9 doses (12.5, 25, 50, 100, 150, 200, and 300 mg twice daily or 150 vs 300 mg once daily) of the tablet formulation of dabigatran etexilate beginning 4 to 8 hours after surgery for total treatment duration of 6 to 10 days.
The BISTRO 1a study showed a relatively low incidence of DVT rates across all doses of dabigatran etexilate, with an overall rate of 12.4%. Pharmacokinetic and pharmacodynamic parameters increased proportionally with dose. The highest rates of DVT (20.8%) were found in the group receiving 12.5 mg twice daily as opposed to 6.1% in the 300 mg once-daily group. Regarding safety, an increased incidence of bleeding events was noted with higher doses of dabigatran etexilate. Although no major bleeding episodes were observed at any of the doses studied, the dose-escalation was terminated at 300 mg twice daily because 2 patients who received this dose experienced bleeding from multiple sites. This study concluded that a therapeutic window between 12 mg once daily and 300 mg twice daily was considered acceptable for its efficacy and safety.
Because the BISTRO 1a study revealed low plasma concentrations of dabigatran after the first dose of the tablet formulation in 20% of patients, BISTRO 1b was conducted to evaluate the efficacy of the capsule formulation of dabigatran etexilate. The data showed that absorption of the first postoperative dose (1–3 hours after surgery) occurred slowly compared with healthy controls. Peak plasma concentrations were achieved in 6 hours following administration; however, on subsequent days the absorption of dabigatran was rapid (time to C max = 2 hours). This delay in absorption on the first postoperative day may represent a “safety net” for these patients given their high risk of bleeding. Thus, this PK justified the use of the capsule form and early dosing regimen to evaluate dabigatran etexilate on clinical outcomes in the BISTRO II study.
BISTRO II trial was a large, multicenter, parallel-group, randomized double-blind study whose primary objectives were to further evaluate the efficacy and safety of the dose-response relationship of dabigatran etexilate for VTE prevention in patients undergoing hip and knee replacement surgery, as well as to compare these outcomes against the current standard therapy, low molecular weight heparin (enoxaparin). Patients were randomized to receive doses of dabigatran etexilate of 50, 150, or 225 mg twice daily, or 300 mg once daily, versus enoxaparin 40 mg once daily. Results of the study again demonstrated a dose-dependent decrease in total VTE with increasing doses of dabigatran etexilate. Also, the incidence of major VTE (proximal DVT and/or PE) was lower in groups with any dose of dabigatran etexilate (1.7%–5.0%) than with enoxaparin (5.6%). Investigators concluded that a total daily dose of 100 to 300 mg of dabigatran etexilate appeared to be as effective as 40 mg of enoxaparin once daily; however, larger studies were recommended to assess the risk of bleeding. An equally important finding was the association between early administration of dabigatran etexilate (1–4 hours postoperative) and a reduced risk of DVT. Further pharmacokinetic and pharmacodynamic studies on patients enrolled in BISTRO II supported a dosing regimen for phase 3 trials, consisting of once-daily doses of 150 mg or 220 mg, with half doses of 75 mg and 110 mg given on the day of surgery.
Pooled analysis of the results from the completed RE-MOBLIZE, RE-MODEL, and RE-NOVATE phase 3 trials, which involved a combined total of 8210 patients, determined that dabigatran etexilate is as efficacious and safe as enoxaparin in the prevention of VTE after hip or knee arthroplasty. Two different doses of dabigatran etexilate, 150 mg or 220 mg once daily with half doses, 75 mg or 110 mg, on the first day were examined in all 3 trials. The lower dose of dabigatran etexilate was reserved for patients with a higher risk of bleeding, such as in those with reduced creatinine clearance or the elderly patients. The North American approved regimen of enoxaparin, 30 mg twice daily beginning 12 to 24 hours after surgery, was the comparator in the RE-MOBILIZE trial, whereas the European enoxaparin regimen of 40 mg daily starting 12 hours before surgery was used as the comparator in RE-MODEL and RE-NOVATE. It is also noteworthy that the first dose of dabigatran etexilate was administered 6 to 12 hours after surgery in RE-MOBLIZE, whereas in RE-MODEL and RE-NOVATE prophylaxis began 1 to 4 hours postoperatively.
Results from RE-MOBILIZE failed to show noninferiority of dabigatran to enoxaparin. The primary end point, total VTE and all-cause mortality rates, were significantly higher in the dabigatran 220 mg (31.1%; P = .02) and 150 mg (33.7%; P <.001) groups compared with enoxaparin (25.3%). Rates of major bleeding were not significantly different between dabigatran groups and the enoxaparin group. The failure to achieve noninferiority is likely secondary to the delay in the administration of dabigatran etexilate after surgery as well as the higher dosing of enoxaparin.
RE-MODEL and RE-NOVATE, both performed in the European Union and other non–North American countries, found both doses of dabigatran noninferior to enoxaparin. Rates of total VTE and all-cause mortality were 36.4% ( P <.0003) and 40.5% ( P <.017) for doses of 220 mg and 150 mg, respectively, versus 37.7% for enoxaparin in RE-MODEL. Similar findings were observed in RE-NOVATE (6.0% [ P <.0001], 8.6% [ P <.0001], and 6.7% in dabigatran 220 mg, 150 mg, and enoxaparin 40 mg daily, respectively). In both studies, rates of major bleeding events were similar across all treatment groups. Administration of dabigatran etexilate beginning 1 to 4 hours postoperatively in both trials with resultant improved outcomes support early administration of thromboprophylaxis. Data from these 2 trials led to the approval of dabigatran etexilate for the primary prevention of VTE after total knee or hip replacement in the European Union and Canada. A fourth phase 3 trial, RE-NOVATE II, is currently ongoing in North America with a goal to evaluate the efficacy of dabigatran 220 mg once daily versus enoxaparin 40 mg once daily over a course of 28 to 35 days for VTE prevention post hip replacement surgery.
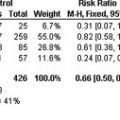
Pooled results from the 3 trials revealed no statistically significant difference between both doses of dabigatran etexilate versus the comparator enoxaparin for the primary end point of total VTE and all-cause mortality (RR 1.06, 95% CI 0.94–1.18), as well as the secondary end point of major VTE or VTE-related death (RR 0.92, 95% CI 0.66–1.29). Similarly, a lack of a statistically significant difference in major bleeding events between dabigatran etexilate 220 mg and 150 mg treatment groups (1.4% and 1.1%, respectively) versus enoxaparin (1.4%) was observed. Further post hoc pooled analysis in elderly patients (>75 years) recommended a 150 mg daily dose of dabigatran etexilate for prevention of VTE. This dose may extend to other patients at higher risk of bleeding, such as those taking amiodarone or verapamil, or with a creatinine clearance less than 50 mL/min.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree