Chapter 16
Ketogenic Diets
Georgiana Fitzsimmons and Marian Sewell
Introduction
The ketogenic diet is very high in fat, very low in carbohydrate with sufficient protein to allow for growth. The diet was first used to treat epilepsy in the 1920s [1]. Following observations that fasting decreased seizure frequency, the diet was designed to induce a similar metabolic response. Use of the diet decreased when new antiepileptic drugs were introduced in the 1950s. It has more recently regained popularity internationally due to concerns about side effects of medications and is considered when medications fail to control seizures. Current guidelines from the UK National Institute for Health and Care Excellence (NICE) for childhood epilepsy recommend considering the ketogenic diet to treat children with medication resistant epilepsy [2].
Approximately 60 000 children in the UK have epilepsy [3] and first line treatment is antiepileptic medication; however, in approximately 25% seizures cannot be controlled this way. After a first medication fails, there is only a 15% chance the second will be effective and only a 20% chance it will stop the seizure activity [4]. The ketogenic diet provides an alternative treatment option.
Ketogenic diets in other disorders
The diet is the treatment of choice for children with glucose transporter 1 (Glut 1) deficiency syndrome [5]. In this disorder an inherited protein defect causes disruption of glucose transport from the blood vessels into the brain. The diet is considered first line treatment because neurological symptoms, including seizures, are regarded as consequences of the energy deficit of the brain. The high fat ketogenic diet produces ketone bodies which the brain uses as an alternative energy source to glucose. Treatment with diet is currently recommended in Glut 1 patients at least until puberty.
Ketogenic diets may also be used as a treatment for pyruvate dehydrogenase (PDH) deficiency [6] and there is also increasing interest in the efficacy of the diet for children presenting with fever induced refractory epilepsy of childhood syndrome (FIRES) [7].
How the ketogenic diet works
The precise mechanism(s) of how the diet works to reduce seizures in epilepsy remains unknown, but there are numerous studies investigating this. It may be due to the direct action of the ketone bodies producing an anticonvulsant effect, or to metabolic changes associated with ketosis. Hypotheses of anticonvulsant effects include:
- Changes to energy metabolism in the brain. Failure to meet brain energy needs may contribute to the initiation and spread of epileptic activity; the ketogenic diet increases brain energy reserves by bypassing less efficient glycolysis pathways and maximising tricarboxylic acid cycle function. This may influence seizure activity by increased production of inhibitory neurotransmitters, improved ability to buffer the extracellular milieu, or altering the resting membrane potential [8].
- Alteration of brain amino acid handling. Utilisation of ketone bodies as a brain substrate alters the metabolism of glutamate, an important excitatory neurotransmitter. Reduced transamination of glutamate to aspartate increases glutamate availability for synthesis of the main inhibitory brain neurotransmitter, gamma amino butyric acid (GABA), both directly and via glutamine production [9].
- Role of neuropeptides and norepinephrine. Anticonvulsant neuropeptides, galanin and neuropeptide Y, are regulated by energy states and may mediate action of the ketogenic diet [10], with increased levels being released in the brain [11]. Norepinephrine, an inhibitory neurotransmitter, may also contribute to the anticonvulsant mechanism of the diet [12].
- Calorie restriction. Calorie (energy) restriction may underlie the anticonvulsant mechanism of the diet; this alone has been shown to reduce seizure susceptibility in mice [13]. Calorie intake is not restricted to less than normal requirements when using the ketogenic diet as it is important to optimise the child’s growth.
Figure 16.1 shows how the brain metabolises energy.
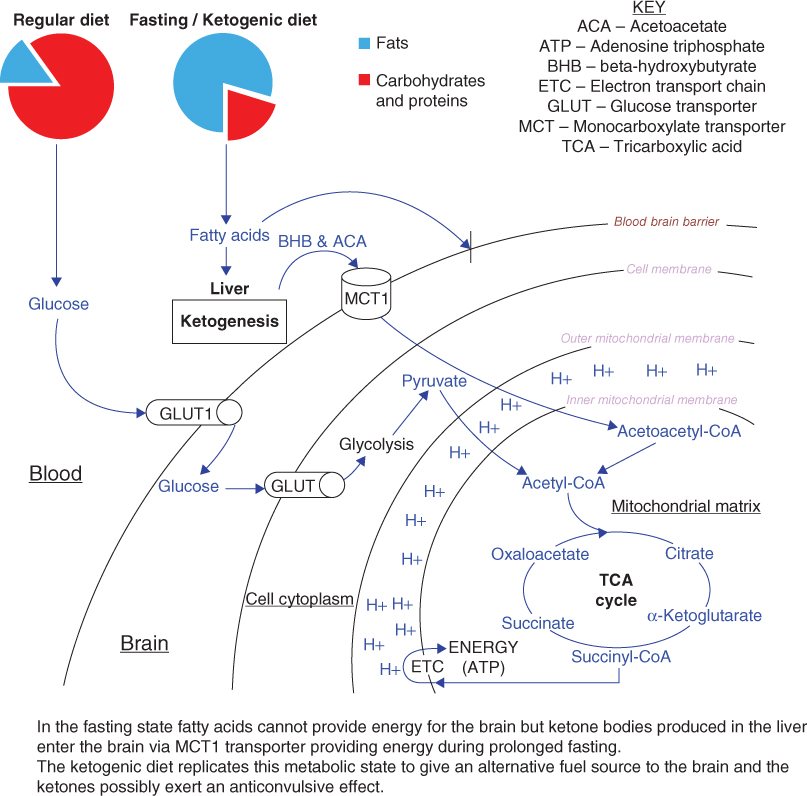
Figure 16.1 Energy metabolism in the brain.
Types of ketogenic diet
Until recently there have been two types of ketogenic diet: the classical and the medium chain triglyceride (MCT) diet. Two further diets have been developed: the modified ketogenic diet (MKD) and the low glycaemic index treatment (LGIT). All types of diet are high in fat and restricted in carbohydrate.
Classical diet
The classical diet is based on a ratio of grams of fat (long chain triglycerides) to grams of carbohydrate and protein [fat:(carbohydrate + protein)]. A 4:1 ratio equates to 90% calories from fat with the remaining 10% from protein and carbohydrate (Fig. 16.2).
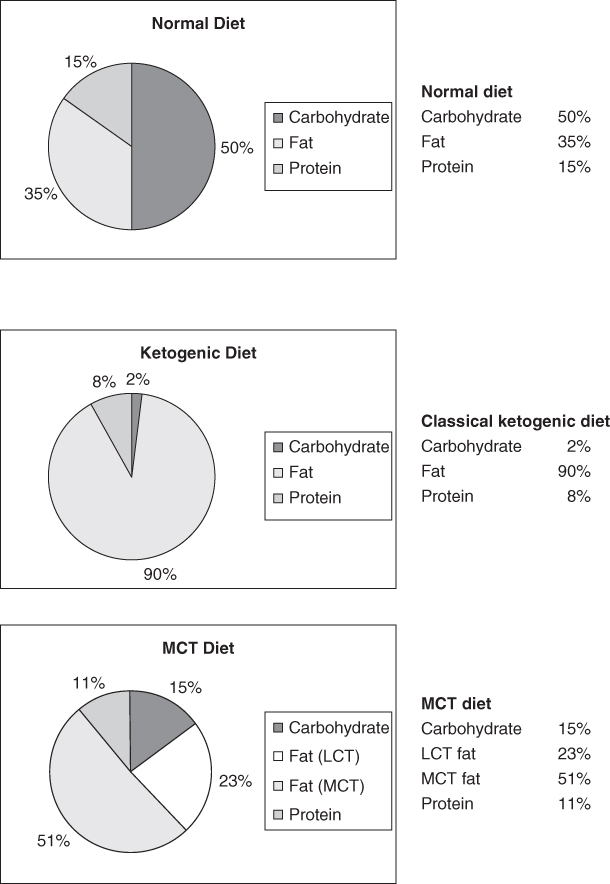
Figure 16.2 Percentages of macronutrients in normal diet, classical ketogenic diet and MCT diet.
MCT diet
A modification of this diet was introduced in the 1970s using MCT as an alternative fat source [14]. MCT yields more ketones per calorie of energy than long chain triglycerides (LCT); MCT is absorbed more efficiently and carried directly to the liver in the portal blood. This increased ketogenic potential means less total fat is needed in this diet and thus allows inclusion of more carbohydrate and protein. The traditional MCT diet has approximately 75% of calories from fat, 45%–55% calories from MCT and 21%–25% from LCT (Fig. 16.2). MCT oil or Liquigen, a 50% MCT emulsion in water, can be used as the source of MCT.
Modified ketogenic diet (MKD) or modified Atkins diet
This is a modification of the Atkins diet and recent studies show similar results in efficacy to the classical and MCT diets in both children [15, 16] and adults [17]. At the authors’ centre this diet is referred to as the modified ketogenic diet; in the USA it is known as the modified Atkins diet. This diet allows a free intake of protein; it is very low in carbohydrate and requires a liberal fat intake (approximately 75% of daily calories). It is frequently used in older children and adolescents who may not cope with the dietary restrictions of the classical or MCT diets.
Low glycaemic index treatment (LGIT)
The LGIT is designed to limit increases in post prandial blood glucose levels by restricting the quantity of carbohydrate to 10% total calories and using only carbohydrate with low glycaemic index (GI) of <50; 60% of calories come from fat and 30% from protein [18, 19].
Efficacy of the ketogenic diet
Seizure reduction
Many studies have reported efficacy of the ketogenic diet. Freeman et al. [20] found approximately 30% of 150 children achieved 90% seizure reduction and 50% achieved 50% reduction. In a systematic review of 11 studies in 2000, Lefevre and Aronson [21] concluded there was sufficient evidence to determine that the diet is efficacious in children with refractory epilepsy although they were concerned about the lack of randomised controlled studies. A Cochrane review of the diet also noted this [22]. A recent randomised controlled trial showed success in treating children with epilepsy with no difference in efficacy between classical and MCT diets [23, 24].
Seizure types
The diet has traditionally been reported to be most successful in treating patients with myoclonic-astatic seizures, or the mixed seizures seen in Lennox–Gastaut syndrome, although Freeman et al. [20] found no significant difference in seizure control between different types of seizure or epilepsy syndrome. The diet is increasingly used as a treatment for infants with infantile spasms [25–27].
Other benefits
Whilst few studies have systematically reviewed the improvements in development and behaviour reported by parents, one study noted significant improvements in development quotient, attention and social functioning whilst on the diet [28]. Although only some children become free of seizures, a larger proportion show a reduction in seizure frequency. Fewer seizures and side effects caused by antiepileptic medications may lead to children being more alert and able to make better developmental progress.
Successful ketogenic diet therapy may result in cost benefits as it may be possible to reduce antiepileptic medications for some children on the diet and some may be able to discontinue medications altogether [29]. Improved seizure control on the diet could potentially also reduce hospital admissions, emergency department attendances and GP visits with both cost benefits and improved quality of life for families [30].
Patients with epileptic seizures often have sleep abnormalities. Hallbook et al. found the diet increased rapid eye movement sleep which contributed to improvement in quality of life [31].
Some children have sustained reduction in seizures following discontinuation of the dietary treatment. A study by Martinez et al. in children who were free of seizures on the ketogenic diet showed the risk of seizure recurrence after discontinuation of the diet was slightly lower than after stopping anticonvulsants, and similar to that following surgery [32].
Possible side effects of the ketogenic diet
Many of the side effects of the diet can be resolved with dietary modifications as shown in Table 16.1.
Table 16.1 Possible side effects of the ketogenic diet
| Side effects | Contributing factors | Possible dietary manipulations |
| Constipation | Limited fibre intake | Increase intake of low carbohydrate, high fibre vegetables |
| Add fibre supplement, e.g. Resource Optifibre | ||
| Insufficient fluid intake | Increase fluid intake | |
| Add sugar free laxative, e.g. Movicol Paediatric | ||
| Excess ketones | Incorrect administration of diet by family/carers | Check how diet is administered |
| Treat with fruit juice (see Managing illness, p. 377) | ||
| Fat intake too high | Reduce LCT/MCT (see Troubleshooting, p. 376) | |
| Nausea, vomiting, diarrhoea | Incorrect administration of fat intake | Check how diet is administered |
| Can occur on initiation of diet or if unwell | Introduce fat more slowly | |
| Food refusal | Excess ketones | See Managing illness |
| Calorie prescription overestimated | Adjust calories (see Troubleshooting) | |
| Dislike of meal choices | Offer alternative menu ideas | |
| Consider change of diet type | ||
| Excess ketones | See Troubleshooting | |
| Underlying food behavioural problems | Offer alternative meal ideas, involve parent support groups to assist with menu planning | |
| Consider referral to specialist feeding team | ||
| Elevated blood lipids | High fat diet | |
| Check blood lipid profile regularly | ||
| Repeat fasting lipid profile if initial test non fasting | ||
| Exchange saturated fats for mono-unsaturated fats and polyunsaturated fats | ||
| Exacerbation of reflux | High fat diet resulting in delayed gastric emptying | Smaller more frequent feeding |
| Add/optimise anti-reflux medication | ||
| Referral to specialist gastrointestinal team for further investigation | ||
| Renal stones | Occur in 3%–6% of patients [33, 34] | |
| Causes may include: | ||
| (1) Dehydration | Optimise fluids | |
| Renal ultrasound is required prior to ketogenic therapy | ||
| (2) Some antiepileptic medications, Topirimate, Zonisamide, Acetozalomide, can increase risk of renal stones [35] | Monthly urine dip stick test for protein recommended for all patients – if three consecutive positive tests will need renal ultrasound | |
| Metabolic acidosis | Excess ketones | See Troubleshooting |
| Increased risk due to decreased blood pH if on Topiramate, Zonisamide or Acetozalomide | Medication may need adjusting – discuss with neurologist | |
| Bone demineralisation and increased risk of fractures | Inadequate calcium, vitamin D supplementation [36] | Supplement with sufficient calcium and vitamin D (see Monitoring the diet, p. 374 ) |
| Chronic acidosis | ||
| Previous use of steroids | ||
| Non compliance with vitamin/mineral supplements Immobility | Consider alternative vitamin/mineral preparations Bone density scan | |
| Possible effect of antiepileptic medications, e.g. sodium valproate | Discuss with neurologist |
Raised lipid levels
Raised serum cholesterol and triglyceride levels are common [37, 38]; this may be less of a problem on the MCT diet [39]. There is no evidence from case reports that the diet causes any adverse effects on cardiovascular function later in adulthood [40].
Delayed growth
Many children on the diet show a reduction in growth velocity [41–44]. Weight gain may be slower than before commencement of diet, particularly during the first 6 months, and the weight centile may drop gradually [45]. Weight and height should therefore be monitored and dietary prescriptions reviewed regularly.
Other reported side effects
These include cardiac complications due to selenium deficiency [46], pancreatitis [47] and hypoproteinaemia [48]. Haematological disturbances have been reported, both impaired neutrophil function [49] and alterations in platelet function with increased tendency to bruising and bleeding [50].
Considerations before commencing the diet
Before dietary treatment is considered it is important that a diagnosis of epilepsy has been made and there has been a trial of appropriate treatments. Children should therefore be referred for the diet by a paediatric neurologist or a paediatrician with a special interest in epilepsy. Contraindications for dietary treatment are given in Table 16.2. Other factors to consider are the following.
- Feeding difficulties. Ketogenic diets are restricted diets requiring exact measurements of food and completion of meals. Before being considered for the diet any behavioural feeding difficulties should be identified and the child referred for appropriate treatment, preferably to a behavioural feeding clinic.
- Dysphagia and significant gastro-oesophageal reflux. This must be appropriately managed prior to starting the diet and ideally should be addressed before the child is referred. If the child’s dysphagia has been treated by a speech and language therapist, an up to date review will be required before referral. If enteral feeding is indicated a gastrostomy needs to be inserted prior to starting the diet.
- Dietary restrictions and food allergy. Although not a contraindication, multiple food allergies may make a ketogenic diet impractical; each child’s case should be assessed individually by the dietitian.
- Medical history. Children with renal stones or hyperlipidaemia and those taking diuretics or medications which increase the risk of acidosis may be unsuitable for dietary treatment and should be assessed by a paediatric neurologist.
Table 16.2 Contraindications for dietary treatment
| Definite contraindications [51] | Possible contraindications |
| Fatty acid oxidation defects | Concomitant steroid use will limit ketosis |
| Organic acidurias | |
| Pyruvate carboxylase deficiency | |
| Familial hyperlipidaemia | |
| Hypoglycaemia under investigation | |
| Ketoneogenesis/ketolysis defects | |
| Diabetes mellitus | |
| Severe gastro-oesophageal reflux | |
| Severe liver disease | |
| Non compliance of patient/carers |
Practical management of the ketogenic diet
Energy intake is expressed as calorie intake throughout this section on dietary management.
Implementation of the ketogenic diet can be difficult and success depends on core staff: a dietitian trained in the use of the ketogenic diet; a paediatric neurologist or a paediatrician with a special interest in epilepsy and experience of the diet; a specialist nurse. This multidisciplinary team (MDT) must be able to advise and assist families when they encounter problems either caused or exacerbated by the diet. Community support and easy hospital access must be available at all times. The support of a dietetic assistant, pharmacist and clinical biochemist may also be helpful for the core team.
Implementing and supporting a child on the ketogenic diet requires sufficient dietetic time. The authors’ centre advises one whole time equivalent (WTE) dietitian for a caseload of 20 children (based on an audit of the time taken to establish children on the MCT or classical diet). However, with additional input from 0.3WTE dietetic assistant the dietitian to patient ratio can be increased to 1:24.
Hospital or home initiation
The diet can be initiated in an outpatient setting, but only if the dietitian can be in telephone contact with the family regularly to provide support; this needs to be daily for the first few weeks. Once established on the diet families require ongoing support to monitor and make dietary adjustments (p. 374). Some centres prefer a hospital admission to start the diet. Infants should start the diet in hospital as they require particularly close monitoring.
Commitment from the family
The success of the diet requires the family and carers to be committed and willing to comply with the dietary regimen and monitoring. Families are required to monitor and record the child’s weight, test and record twice daily urine or blood ketones, keep a seizure diary, communicate frequently with the dietitian and attend regular clinic appointments. The extent of the dietary restrictions and the need to weigh all food accurately must be stressed from the outset.
Preparing for the diet
Medical investigations and screening tests may be required before starting the diet, at the discretion of the paediatric neurologist. Any potential side effects should be discussed with the family at the initial screening visit (Table 16.1).
Laboratory tests and urinalysis
Urine organic acids and a blood spot carnitine test should be completed before the diet is initiated to screen for metabolic disorders that would contraindicate its use (see Table 16.2). These may have already been done to help determine a diagnosis. As these tests take time to be analysed they should be completed early in the assessment period.
Establishing the goals of dietary treatment
Realistic expectations should be discussed with the patient and family. Treatment goals should be agreed and documented before the diet is started and be reviewed at follow-up appointments to help decide if continuing treatment is indicated.
Dietetic assessment
A 3–4 day food diary is essential to assess suitability for ketogenic dietary treatment and give an indication of the child’s food preferences and eating patterns (Table 16.3). This is used to help decide which diet is most suitable for the child and forms a basis for meal plans.
Table 16.3 Factors to consider in the pre ketogenic diet dietetic assessment
| 3–4 day food record diary to assess current dietary intake, eating patterns and nutritional status |
| Food preferences and restrictions |
| Behavioural issues with food |
| Food allergies or intolerances |
| Swallowing difficulties |
| Developmental feeding skills, food textures, thickened fluids |
| Fluid intake and types |
Anthropometric measurements and recent growth trends
|
| Level of mobility and activity; immobile children may need fewer calories than active children |
| Vitamin and mineral supplements |
Bowel habits
|
Others involved in the child’s care
|
Seizures may influence energy requirements
|
Medications
|
| Baseline bloods and urinalysis completed (p. 374) |
Pre diet meal manipulation
Some children may benefit from a reduction in sources of concentrated carbohydrate (CHO) and the introduction of high fat foods such as cream and mayonnaise, if not already part of the usual diet. Caution is required as introducing too much fat before starting the ketogenic diet could cause excessive weight gain. Samples of Ketocal (p. 363) Betaquik or Liquigen can be given to try depending on the type of diet proposed.
Ketogenic meals appear much smaller than normal meals due to so many of the calories being provided by high fat foods. Sample menus illustrate what a typical ketogenic meal looks like and helps families understand all that is involved in meal preparation, e.g. accurate weighing and incorporating fat.
Pre diet manipulations should be supervised by a dietitian as excessive CHO restrictions could lead to hypoglycaemia, hyperketosis, weight loss or other detrimental effects. It may not be advisable to restrict CHO intake pre diet for children with Glut 1 deficiency.
Deciding which ketogenic diet to use
The 3–4 day food diary will form the basis for the type of diet chosen, but the child’s and family’s preference should be taken into account as well as palatability and ease of use. It is possible to change from one diet to another if the initial diet is unsuitable. The advantages and disadvantages of each type of diet are shown in Table 16.4.
Table 16.4 Advantages and disadvantages of different diet types
| Diet type | Advantages | Disadvantages |
| Classical | Uses ‘normal’ foods More experience in use of classical diet and more widely used EKM can be used to calculate meal plans Suitable for tube feeds (Ketocal p. 370) | Meals appear small because of the high fat content Very restricted CHO and protein |
| MCT | Less total fat allows more protein and CHO so meals may appear more ‘normal’ May be suited to those with limited food choices, high intakes of carbohydrate or the more selective eater | Possible gastrointestinal side effects or laxative effect MCT required with every meal/snack |
| MKD | Free protein, calories No weighing of foods, household measures used May help families or teenagers engage with a ketogenic diet who might otherwise find it too difficult | Generous fat intake still required (75% of calories) Very restricted CHO (10–20 g/day) May make dietetic management difficult as no control on calories May require more parent/carer input in designing meals |
| LGIT | Food not weighed, uses simple exchanges Currently limited experience in the UK | Limited CHO (10% total calories) Generous fat intake still required (60% of calories) |
EKM, electronic ketogenic manager; CHO, carbohydrate; MCT, medium chain triglyceride; MKD, modified ketogenic diet; LGIT, low glycaemic index treatment.
Duration of dietary treatment
In most cases there is a 3 month trial period. Progress is then reviewed at an MDT clinic appointment and it is decided with the family if continuing treatment is appropriate. If the diet is of no benefit it should be discontinued. Some reasons for discontinuing the diet are
- child and family finding the diet too difficult and do not wish to continue
- diet is not effective in controlling seizures or not achieving goals set at baseline
- side effects outweigh benefits of the diet, e.g. poor growth
- non compliance
If the diet continues then 6 monthly reviews are necessary. In the authors’ a trial off diet is suggested at 2 years to determine the ongoing efficacy of the treatment.
Initiating ketogenic diets
Calculating the diet
The diet prescription needs to be adjusted over time depending on the child’s weight, level of ketosis, seizure control and activity. For example if seizure activity decreases, mobility may increase and with it energy requirements (p. 376). Although fasting has been traditionally used to initiate the diet this has now been shown to be unnecessary [52, 53].
Calorie requirements
Calorie intake is carefully controlled on the ketogenic diet as excess or limited calories may compromise ketosis and seizure control. All calculations for the diet are expressed in (kilo) calories as the unit of energy and daily intake is based on the estimated average requirement (EAR) for energy as advised by the UK Department of Health [54]; however, requirements should be individualised.
Protein requirements
Protein requirements are based on safe levels for growth [55]. At the authors’ centre the classical diet includes the protein content of vegetables in the daily allowance so it is important to give a minimum of 1.0–1.2 g protein/kg/day. This may need to be higher for younger children to ensure optimal growth [56].
Diet prescription for the classical diet
The calculation is based on
- calorie requirements
- protein requirements
- ratio of fat : (protein + CHO), e.g. 4:1 = 4 g fat : 1 g (protein + CHO)
Proportions of fat for different classical diet ratios are given in Table 16.5. A worked example of the classical 4:1 diet is given in Table 16.6 and an example meal prescription is given in Table 16.7.
Table 16.5 Proportions of fat for different classical diet ratios
| Ratio | Approximate percentage of calories from fat (remainder from protein and CHO) |
| 4:1 | 90 |
| 3.5:1 | 89 |
| 3:1 | 87 |
| 2.5:1 | 85 |
| 2:1 | 82 |
| 1.5:1 | 77 |
| 1:1 | 69 |
CHO, carbohydrate.
Table 16.6 Worked example for classical 4:1 diet
| A 22-month-old boy with late onset infantile spasms | |
| Weight 12.45 kg (75th centile) Height 86.3 cm (50th centile) | |
| Food diary analysis 1100 kcal/day | |
| Weight centile > height centile therefore plan = 1000 kcal/day | |
| Calculation | |
| 4:1 ratio = 90% kcal from fat | 90/100 × 1000 = 900 kcal |
| 900 kcal/9 kcal per gram fat = 100 g fat | |
| Remainder for protein and CHO | 1000 kcal–900 kcal = 100 kcal for protein and CHO |
| (100 kcal/4 kcal per g protein or CHO) | |
| = 25 g protein and CHO | |
| Safe level protein [55] = 0.97 g/kg Aim: 1.2 g/kg = 15 g protein (60 kcal) CHO = 25 g − 15 g = 10 g (40 kcal) | |
| Daily prescription | |
| 1000 kcal/day | |
| 100 g fat | |
| 15 g protein | |
| 10 g CHO | |
| Use the family’s food diary to estimate calorie distribution for meals and snacks | |
| Work out grams of nutrients per meal/snack | |
| Divide kcal required for meal/total kcal per day | |
| e.g. for a 300 kcal meal: 300/1000 × 15 g protein per day = 4.5 g protein/meal | |
| 300/1000 × 10 g CHO per day = 3 g CHO/meal | |
| 300/1000 × 100 g fat per day = 30 g fat/meal | |
CHO, carbohydrate.
Table 16.7 Example meal prescription for classical 4:1 diet: 1000 kcal diet, 3 meals and 1 snack
| Meal/snack | Amount of fat, protein, CHO |