• Graves’ disease (95% of cases)
• Thyroiditis
– Chronic lymphocytic thyroiditis (Hashimoto’s thyroiditis)—transient
– Subacute thyroiditis
• Autoimmune neonatal thyrotoxicosis (placental passage of maternal thyroid hormone receptor antibodies)—transient
• Autonomous functioning lesions
– Toxic thyroid adenoma
– Activating mutation of the TSH receptor gene
– Activating mutation of Gsα (McCune–Albright syndrome)
– Hyperfunctioning papillary or follicular carcinoma
• Isolated pituitary resistance to thyroid hormone
• TSH secreting pituitary adenoma
• Exogenous causes
– Excessive thyroid hormone replacement therapy
– Iodine induced hyperthyroidism
Graves’ disease is caused by thyroid stimulating immunoglobulins that activate TSH receptors on thyroid follicular cells resulting in thyroid hormone overproduction. Note that the clinical aspects of this syndrome, including the goitre, ophthalmopathy, and palpitations were described in the late eighteenth and early nineteenth centuries independently by several physicians from Great Britain and continental Europe including Parry, Graves, Basedow and Flajani. All of their names, either singly or in various combinations, have been attached to this common clinical condition. We will use the name “Graves’ disease” because it is commonly recognised in the English medical literature.
Although Graves’ disease is common in adults it is uncommon in children with a frequency of 0.1–3 per 100,000 children [10]. The incidence increases throughout childhood, with the peak incidence in children aged 10–15 years. The majority (60%) of patients being postpubertal [11]. As with most autoimmune diseases, Graves’ disease in children is more common in girls than boys, at a ratio of 5–6:1 [12]. Other causes of hyperthyroidism do not show a male or female preponderance. For example, the hyperthyroidism of McCune–Albright syndrome is not more common in girls even though the associated precocious puberty is more common in girls than in boys [13].
Graves’ disease is often associated with other autoimmune diseases and there is often a strong family history of thyroid and non-thyroid autoimmune problems [14] such as systemic lupus erythematous, rheumatoid arthritis, myasthenia gravis, vitiligo, immune thrombocytopenic purpura, and pernicious anaemia. In addition, there are several lines of evidence suggesting a genetic component or predisposition to the disease. Some studies suggest that up to 80% of the susceptibility to Graves’ is determined by genetic factors [9]. Graves’ disease has been linked with human leukocyte antigen (HLA)-B8 and (HLA)-DR3 and with abnormalities in chromosomes 6p21 and 2q33 [15]. It is also more common in children with Trisomy 21 [16]. Overall, genetic susceptibility is thought to have a polygenic inheritance, although monozygotic twin studies suggest interplay between environmental and genetic factors.
Evaluation
The diagnosis of hyperthyroidism is often challenging due to the large spectrum of physical and psychological complaints [17]. However, a focused history and physical examination can usually select patients for lab tests and imaging studies that will confirm the diagnosis. The following discussion of the evaluation of patients with hyperthyroidism will focus on Graves’ disease and only briefly mention the other rare causes of hyperthyroidism in children.
History
The symptoms of Graves’ disease in children and adolescents can develop insidiously over months but can also have an abrupt onset. The most common symptoms are behaviour changes such as increased anxiety and hyperactivity. These symptoms are often similar to those of attention deficit hyperactivity disorder (ADHD), [18] and a high degree of clinical suspicion is necessary to pursue the diagnosis of Graves’ disease in this situation. Individuals with Graves’ disease invariably have an altered mental status with increased irritability, emotional lability, and outbursts which understandably create distress for themselves and their families. Other early symptoms of hyperthyroidism in children include deterioration of school performance and changes in handwriting.
Children with hyperthyroidism may fatigue easily and this usually manifests as exercise intolerance. In severe cases they may have difficulty climbing up stairs as a result of muscle weakness. Sleep disturbances are common as is weight loss despite a good appetite. Hyperthyroidism can also cause idiopathic intracranial hypertension (also known as benign intracranial hypertension or pseudotumor cerebri) in children and present with headache and even nausea and vomiting [19]. Symptoms of hyperthyroidism caused by autonomic nervous system disturbances include tremor, heat intolerance, sweating, diarrhoea and palpitations. In girls, menstrual cycle irregularities, including amenorrhoea, are common symptoms. Finally, parents may report that the child has a growth spurt that results from the increased height velocity associated with hyperthyroidism [20].
In addition to hyperthyroidism, Graves’ disease may be associated with eye abnormalities that are the result of autoimmune attack of soft tissue in the orbit. The full array of signs and symptoms in Graves’ ophthalmopathy describes the triad of exophthalmos, chemosis and diplopia. Patients may complain of persistent visual blurring secondary to optic neuropathy or severe eye pain secondary to corneal ulceration. When these symptoms of congestive ophthalmopathy are present then urgent referral to an ophthalmologist for an eye assessment is necessary. Compared to adults, thyroid eye disease is usually mild in children.
Physical Examination
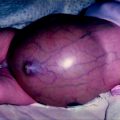
Vital signs of children with hyperthyroidism usually reveal tachycardia and less commonly hypertension. Height and weight are important to evaluate because hyperthyroidism may cause increased linear growth velocity and weight loss. General examination may reveal sweatiness, facial flushing, a tremor or, rarely, choreiform movements [21]. Further to the symptoms described above the eye signs such as eyelid retraction, proptosis, periorbital oedema, optic neuropathy, corneal ulceration, and rarely ophthalmoplegia may be present in Graves’ disease (Fig. 2.1). Importantly, the eye changes sometimes present before signs of thyrotoxicosis. All children with Graves’ disease should be referred to ophthalmology for formal assessment since the eye findings may be subtle but potentially serious.


Fig. 2.1
A 14-year-old girl with Graves’ disease who had diffuse goitre and asymmetrical proptosis (From Bhansali et al. [47], with permission.)
Pubertal stage should be assessed as part of the evaluation of associated menstrual irregularity and to evaluate the uncommon possibility of McCune–Albright syndrome which could present with precocious puberty and cafe au lait pigmentation. Additional skin signs of Graves’ disease include the uncommon finding of pretibial myxoedema which represents an unusual autoimmune dermopathy.
The examination of the neck for thyroid abnormalities is essential in the assessment of hyperthyroidism. A goitre or diffuse enlargement of the thyroid is almost always present in Graves’ disease even though it may not have been noticed by the patient or family. Thyroid nodules are less commonly present. Examination of the thyroid is usually done with the neck slightly extended. Swallowing will elicit movement of the thyroid and potentially clarify the identity of a midline neck swelling. Examination of the thyroid is often best performed by standing behind the patient. As with any swelling or mass, evaluation and description of a thyroid goitre or nodule’s size, site, symmetry, consistency and tenderness is essential. The increased blood flow to a hyperfunctioning thyroid sometimes results in a thyroid bruit.
Laboratory Tests
In the evaluation of hyperthyroidism the pivotal investigation is to measure thyroid function tests—particularly the TSH, fT4 and fT3. In primary hyperthyroidism, when the thyroid gland is producing excessive thyroid hormones, TSH is suppressed and fT4 and fT3 are elevated. In addition, there is preferential conversion of fT4 to fT3 so usually the fT3 will be relatively more elevated. In contrast to the low TSH in primary or thyroid hyperthyroidism, the TSH is elevated in secondary or pituitary hyperthyroidism when there is production of TSH from the pituitary that is not responsive to the normal control mechanisms. When evaluating and managing hyperthyroidism fT3 and fT4 are usually better tests than total T3 and total T4 because total T3 and total T4 measure the much larger pool of protein-bound thyroid hormones in addition to the unbound, free forms. Therefore measurement of total T3 and T4 may be misleading in situations with abnormal serum protein levels or when there are changes in protein binding. Free T3 is relatively more important than free T4 in the diagnosis of hyperthyroidism since fT3 rises before fT4 in thyrotoxicosis. Rarely the fT4 levels can be normal but the fT3 is raised in “T3-toxicosis”.
Other thyroid tests can confirm the aetiology of hyperthyroidism. The diagnosis of Graves’ disease can be confirmed by the detection of stimulatory TSH receptor antibodies. Thyroid peroxidase antibodies are elevated in autoimmune thyroiditis that may present with hyperthyroidism as well as the more common presentation of hypothyroidism. Graves’ disease may also have thyroid ophthalmological immunoglobulin associated with eye signs and thyroid growth immunoglobulins associated with thyroid enlargement.
Other Investigations
Although history, physical examination and thyroid function tests are the critical components of the evaluation of hyperthyroidism other imaging investigations are occasionally useful. Thyroid ultrasound can be useful to confirm the suspicion of a diffuse thyroid goitre and screen for thyroid nodules. See Chap. 3 for further discussion of the evaluation and management of thyroid nodules.
Radioactive iodine (RAI) uptake and scanning may be used when the aetiology of hyperthyroidism is still unclear after laboratory evaluation. In Graves’ disease, RAI uptake is elevated and the scan shows diffuse uptake.
Management
Current treatment options include antithyroid medications that block thyroid hormone production and radioactive iodine and thyroidectomy that eliminate thyroid hormone producing follicular cells. None of the current treatment options are ideal and there is controversy regarding the best therapy for children with hyperthyroidism. In view of the lack of clinical trials to guide treatment decisions, our current opinion is to tailor treatment based on the individual patient’s needs while considering the risks and benefits of each treatment [22]. Medical treatment is the first option for treating hyperthyroidism and maybe the only treatment necessary. It is rare that children require hospital admission at diagnosis, however, if there are marked hyperdynamic cardiovascular symptoms then admission maybe necessary until beta-blockers have controlled these symptoms. Two-thirds of patients relapse following medical treatment and in these individuals, radioiodine or surgery are the second-line options [21]. The outcome from both is excellent. Although the patient will be hypothyroid and need lifelong thyroxine treatment this is easy to manage and results in clinically euthyroid individuals. The risk of relapse decreases with a longer primary course of antithyroid medication [9].
Despite the management challenges, prognosis in the majority of children with hyperthyroidism is good when treatment is timely and appropriate [23]. It is important to remember that long-term consequences of hyperthyroidism can result from both the disease and the treatment used [2]. Stratifying patients according to risk of relapse after medical management with antithyroid medication by identifying predictive factors early has led to improvement in overall management. Factors include young age, severity of hyperthyroidism at diagnosis and the presence or absence of other autoimmune conditions are important to consider.
Medical Management
If the individual is very symptomatic then propranolol (or another beta blocker) is used to control the symptoms of sympathetic overdrive, such as tremor and tachycardia, until definitive therapy. Propranolol is usually begun orally at 0.25–0.5 mg/kg/dose, two to four times daily and the dose is titrated for symptom relief. Propranolol can be stopped when the thyroid hormone levels fall into the normal range. Propranolol should be avoided in patients with asthma but a more selective beta-blocker can be used in this circumstance.
Antithyroid drugs are usually considered first-line treatment of hyperthyroidism in children [24]. Antithyroid drugs are known as thionamides and include propylthiouracil (PTU), carbimazole and the active metabolite methimazole. They inhibit thyroid hormone synthesis by interfering with the organification process by which iodine attaches to the tyrosine moieties in thyroglobulin, i.e. the thyroid peroxidase mediated iodination. PTU and methimazole are available in the United States and much of the rest of the world. Methimazole is preferred to PTU because PTU is associated with a risk of severe liver damage (PTU induced hepatitis) and production of cytoplasmic anti-neutrophil autoantibodies. Antibody-positive vasculitis is exceptionally rare. PTU can block the conversion of T4 to T3 whereas methimazole cannot [9, 25]. The United States Food and Drug Administration (FDA) recommend that PTU should be used in children only if other treatment options are unavailable. Another antithyroid drug is carbimazole. Carbimazole is metabolised by the patient into methimazole. Carbimazole is widely used in the United Kingdom, Europe, and the countries of the former British Commonwealth. Carbimazole, methimazole and propylthiouracil have side effects that range in severity from a transient rash to agranulocytosis and neutropenia. Allergic-type reactions with fever, rash, urticaria, gastrointestinal symptoms and arthralgia occur in 1–5% of patients. These reactions are usually transient and can be treated with antihistamines without discontinuing therapy. Agranulocytosis and neutropenia is a rare but serious side effect. Although it is not necessary to check regular complete blood counts it is important to communicate to the family and child that they should immediately report symptoms of easy bruising, sore throat, mouth ulcers, or fever and a complete blood count performed urgently to ensure immunocompetency. It is important to provide the family with verbal and written instructions that specify what symptoms mandate urgent reporting and the need to stop medications. Clear documentation of these discussions and instructions is essential. Even when the patient suffers a serious side effect of an antithyroid drug it is often possible to switch to an alternative antithyroid drug with careful monitoring.
The recommended dose of carbimazole for children less than 12 years is 0.25 mg/kg/dose three times per day. For children between 12 and 18 years the dose is 10 mg three times per day. The dose for propylthiouracil is 25 mg three times per day for children 1–5 years, 50 mg three times per day for children 5–12 years and 100 mg three times per day for adolescents 12–18 years. The conversion between these two antithyroid drugs is 1 mg of carbimazole equates to approximately 10 mg of propylthiouracil.
There are two main antithyroid drug treatment strategies, (1) block and replacement or (2) dose titration. In block and replacement the thyroid gland is first “blocked” or “switched off” with antithyroid drugs, suppressing T4 and T3 and rendering the patient virtually hypothyroid. The “block” is accomplished using relatively higher doses of any of the antithyroid drugs. With this strategy it usually takes 2 weeks or more of treatment before the patient becomes euthyroid. When T4 and T3 levels have fallen and stimulate the patient’s TSH into the normal range then “replacement” with exogenous thyroxine commences. Some of the possible advantages of the “block and replace” regimen include having less blood tests, improved stability and possibly higher remission rates [5, 26–28]. The higher doses of antithyroid drugs used in “block and replacement therapy” provides better control of thyroid hyperfunction and may be responsible for a higher remission rate. Compliance with taking the needed thyroxine replacement can be increased by direct observation of treatment by parents or providing a pill box. The best routine with the most effective pharmacokinetic action is to take thyroxine in the morning on an empty stomach.
The second antithyroid drug strategy is “dose titration therapy” which uses lower doses of antithyroid drugs so that within 1–2 months of starting antithyroid treatment the fT4 and fT3 fall and the TSH remains suppressed. When fT3 and fT4 are within the lower half of the normal range, which can take up to 3 months with this option, the antithyroid drug dose is reduced and “titrated” to keep the fT3 and fT4 in the normal range without the need for exogenous thyroxine treatment. A potential advantage of the “dose titration” strategy may be better compliance with the monotherapy of an antithyroid drug alone rather than an antithyroid drug and thyroid hormone replacement. In addition, the lower doses of antithyroid drugs may result in fewer side effects. More blood tests may be necessary in the “titration” phase and this is a disadvantage of the dose titration therapy, especially in children.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree