Hot Flashes
Domingo G. Perez
Charles L. Loprinzi
Nearly* three fourth of menopausal women experience hot flashes (1), which are usually described as a sudden and disturbing sensation of intense warmth that starts in the chest and then progresses to the neck and face. Red blotches can appear on the skin, and the increase in skin temperature can lead to profuse sweating. This feeling of intense warmth is often accompanied by palpitations and anxiety. Hot flashes usually last about 4 minutes, but they can last for as little as a few seconds or for 10 minutes or longer. In some women, hot flashes occur every 20 minutes, while in others they occur only once a month. Hot flashes typically start 1 or 2 years before menopause and continue to occur for 1 to 5 years, but in some women they can occur for a longer period of time. Hot flash symptoms can have serious detrimental effects on a woman’s work, recreation, sleep, and general perception of quality of life (2).
Hot flashes can be a significant problem for patients with cancer. In many premenopausal women with breast cancer and other gynecologic malignancies, the precipitation of menopause by oophorectomy, chemotherapy, radiotherapy, or hormonal manipulation can lead to the rapid onset of hot flash symptoms that are more frequent and severe than those associated with natural menopause (3). Discontinuation of estrogen replacement therapy in women newly diagnosed with breast or uterine cancer often results in hot flash symptoms. In addition, tamoxifen and the aromatase inhibitors, the most commonly prescribed agents for the treatment of breast cancer, are associated with an increased risk of hot flashes (4). Postmenopausal women with a history of hot flashes are more likely to have hot flashes when exposed to tamoxifen.
Hot flashes are also a common problem in men undergoing androgen deprivation therapy for prostate cancer. Hot flashes have been reported to occur in up to 70% of men after orchiectomy, 80% of men receiving neoadjuvant hormonal therapy before radical prostatectomy, and 70–80% of men receiving long-term androgen deprivation therapy (5, 6, 7).
Pathophysiology of Hot Flashes
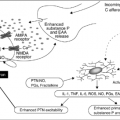
In humans, perspiration and vasodilatation, the classic mechanisms of heat loss that are activated during hot flashes, are centrally regulated by the thermoregulatory nucleus in the medial preoptic area of the hypothalamus (8). The thermoregulatory nucleus activates perspiration and vasodilatation to keep core body temperature within a tightly regulated range known as the thermoregulatory zone. In menopausal women with hot flashes, the thermoregulatory zone is shifted downward and is narrower than in menopausal women who do not have hot flashes (9). Therefore, in women with hot flashes, small changes in body temperature (as low as 0.01°C) may trigger the mechanisms of heat loss that lead to hot flash symptoms (10).
The dramatic decrease in sexual hormone levels that occurs in menopausal women and in men receiving androgen deprivation therapy is thought to be responsible for lowering and narrowing the thermoregulatory zone. However, sexual hormones have profound effects on multiple neuroendocrine pathways (11) and the exact mechanisms by which they affect the thermoregulatory zone are poorly understood. Therefore, when discussing the pathophysiology of hot flashes and the possible mechanism of action of any of the nonhormonal agents currently in use for the treatment of this problem, only an educated guess can be made.
Since estrogen withdrawal results in decreased central serotonergic activity (12) and some of the newer antidepressants (i.e., venlafaxine and paroxetine) have been shown to relieve hot flashes in placebo-controlled, randomized clinical trials (vide infra), serotonin (5-HT) is thought to play an important role in mediating the thermoregulatory effects of estrogen. In particular, the 5-HT2A receptor has been closely associated with thermoregulation in mammals. Multiple animal and human studies have shown that central expression of the 5-HT2A receptor decreases after estrogen withdrawal and that estrogen treatment reverses this change in estrogen-deficient animals and women (12, 13). In addition, tamoxifen has been shown to block the positive effects of estrogen on central 5-HT2A receptor expression in ovariectomized rats (14). Since estrogen withdrawal and tamoxifen treatment result in decreased central expression of 5-HT2A receptors, it is possible that the efficacy of the newer antidepressants against hot flashes is due, at least in part, to their ability to cause a “compensatory” increase in central 5-HT2A signaling (15).
Norepinephrine has also been implicated in the pathophysiology of hot flashes. Estrogen withdrawal leads to increased norepinephrine levels in the hypothalamus (9), which are thought to contribute to the lowering and narrowing of the thermoregulatory zone. In keeping with this theory, several placebo-controlled, randomized clinical trials have shown that clonidine, a centrally acting α2-adrenergic receptor agonist, is moderately effective in decreasing hot flashes (vide infra).
The neuroendocrine pathways that govern thermoregulation in mammals are extraordinarily complex and, as yet, incompletely understood. There is a clear need for further studies to clarify the pathophysiology of hot flashes and to guide the clinical development of more targeted nonhormonal treatments for this problem.
Treatment of Hot Flashes
Nonpharmacologic Interventions
Multiple nonpharmacologic interventions have been claimed to be of help in alleviating hot flashes. These include the use of fans, air conditioners, cold water, special diets, exercise programs, acupuncture, meditation, relaxation techniques, paced respiration, biofeedback, and so on. However, none of these strategies has been tested in controlled, randomized clinical trials and the placebo effect likely plays a significant role in their apparent efficacy. Indeed, placebo-controlled, randomized clinical trials in women with hot flashes have consistently shown a 20 to 30% reduction in hot flash frequency and severity in women in the placebo group over a 4-week period (vide infra). This must be kept in mind when evaluating anecdotal evidence or pilot studies.
Pharmacologic Interventions
Hormonal Therapy
Estrogen
Estrogen reduces hot flashes by 80 to 90% in women with this problem (16). However, there are many situations in which estrogen treatment is contraindicated. For instance, women with a history of coronary artery disease, venous thromboembolism, and uterine cancer should not take estrogen. The use of estrogen in women with a history of breast cancer is controversial. Several prospective and retrospective studies suggest that at least some breast cancer survivors (women with small tumors, negative lymph node status, long disease-free survival, or estrogen receptor–negative tumors) can be safely treated with estrogen replacement (17). However, in the absence of data from large, well-designed randomized clinical trials proving the safety of estrogen treatment in women with a history of breast cancer, most physicians will avoid using estrogen in this patient population. Moreover, the results of the Women’s Health Initiative study (18) and other recent studies suggest that long-term estrogen treatment should not be recommended for most women for a variety of reasons.
Progestational agents
Several pilot studies in the 1970s and 1980s suggested that medroxyprogesterone decreased hot flashes. Subsequently, a placebo-controlled, randomized clinical trial of megestrol 40 mg daily in 97 women with history of breast cancer and 66 men receiving androgen deprivation therapy for prostate cancer showed a reduction in hot flashes of 75–80% in the treatment group compared to 20–25% in the placebo group (19). Three years after the completion of the study, one third of the women were still taking megestrol, and this group of women reported having less hot flashes that women who had stopped taking the medication (20). The intramuscular long-acting progestational agent depomedroxyprogesterone acetate (DMPA) has also been shown to be useful for the treatment of hot flashes. In a randomized clinical trial of DMPA versus megestrol acetate, both agents were found to have similar efficacy (21). A placebo-controlled, randomized clinical trial of a progesterone cream in 102 postmenopausal women showed a reduction in hot flashes of 83% in the treatment group compared to 19% in the placebo group, after 4 weeks of therapy (22). This benefit was still present after 12 months with continued use of the progesterone cream. Adverse effects of progestational agents include vaginal bleeding upon discontinuation of the medication, weight gain, bloating, and thromboembolic phenomena.
Despite the proved efficacy of progestational agents for the treatment of hot flashes, many physicians are wary of using hormonally active agents in patients with a history of breast cancer. Though there is some evidence that progestational agents are active against breast cancer (23), in vitro data suggest that they can increase epithelial cell proliferation, a potentially undesirable effect in patients with a history of breast cancer (24). In addition, megestrol was reported to increase the prostate specific antigen (PSA) level in a patient with prostate cancer who was being treated with this agent for hot flashes (25). Given the ongoing debate on this issue, patients need to be counseled before starting a progestational agent if they have a history of breast or prostate cancer.
Nonhormonal Therapy
The reluctance to use hormonally active agents in patients with a history of breast cancer provided the impetus for finding nonhormonal agents that could help alleviate this problem. The following is a brief summary of their clinical development.
Newer antidepressants
In the 1990s, several authors reported reductions in hot flash frequency and severity in postmenopausal women who were taking several of the newer antidepressants for other reasons. Since then, the results of 18 prospective studies of several newer antidepressants for the treatment of hot flashes have been reported. These studies have been reviewed in detail elsewhere (26). Given the reluctance to use estrogen or even a progestational agent in women with a history of breast cancer, many of these studies were done in this patient population, but some studies have been done in noncancer patients and in men with prostate cancer. Self-completed daily hot flash diaries were used to document the frequency and severity of hot flashes. Data on toxicity, quality of life and mood status were commonly obtained. The main efficacy measures used in most studies were the change from baseline in the weekly average number of daily hot flashes and average hot flash score (defined as the number of mild hot flashes plus twice the number of moderate hot flashes plus three times the number of severe hot flashes plus four times the number of very severe hot flashes during that week).
Venlafaxine
Venlafaxine selectively inhibits serotonin, norepinephrine, and dopamine reuptake, in order of decreasing potency. The efficacy of venlafaxine for the treatment of hot flashes was first studied in 1997 in a small pilot study (27). This study included women with a history of breast cancer and men receiving androgen deprivation therapy for prostate cancer. Patients were treated with venlafaxine 12.5 mg twice a day for 4 weeks. Of the 31 patients originally enrolled, 25 completed the study. Eighty-two percent of the patients were women. The average number of daily hot flashes decreased from 6.6 at baseline to 4.3 during the last week of the study. The average hot flash score decreased by 55%. Patients reported significant improvement in fatigue, sweating, and difficulty sleeping, and, at the completion of the study, 64% of the patients chose to continue venlafaxine.
Subsequently, the same investigators conducted a placebo-controlled, double-blind, randomized clinical trial to assess more definitely the efficacy and toxicity of venlafaxine in breast cancer survivors with hot flashes (28). Women were randomized to four treatment arms:
Placebo (n = 56)
Venlafaxine extended release (ER) 37.5 mg daily for 4 weeks (n = 56)
Venlafaxine ER 37.5 mg daily for 1 week, followed by 75 mg daily for 3 weeks (n = 55)
Venlafaxine ER 37.5 mg daily for 1 week, followed by 75 mg daily for 1 week, followed by 150 mg daily for 2 weeks (n = 54).
Sixty-nine percent of the women were taking tamoxifen. Complete information was available for 191 of the 221 women originally enrolled onto the study. After 4 weeks of treatment, the median frequency of hot flashes decreased 19, 30, 46 and 58% in women in groups 1, 2, 3 and 4, respectively, (p >.001 for all groups compared to placebo). Hot flash activity decreased more than 50% in 20, 45, 63 and 55%, respectively. Beck Depression Inventory (BDI) scores improved by a mean of 1.6, 2.4, 4.8 and 3.2 points, respectively. Overall quality of life decreased by 3 points in the placebo group compared to an average increase of 3 points in patients taking venlafaxine ER (p = .02). Dry mouth, nausea, constipation, and decreased appetite were significantly more common in patients taking venlafaxine. Venlafaxine ER 150 mg daily was associated with significantly more adverse effects. Efficacy was similar whether tamoxifen was being used or not.
At the completion of the study, treatment was unblinded and women were offered the option of participating in an 8-week, open-label, continuation study (29). Of the 221 women originally enrolled onto the double-blind study, 157 participated in the continuation study. At the time of data analysis, complete information was available for 102 women. Sixty-six percent of women were taking tamoxifen. At the completion of the study, 26 women were taking venlafaxine ER 37.5 mg daily, 35 were taking 75 mg daily, 6 were taking 112.5 mg daily, and 34 were taking 150 mg daily. Among women initially randomized to placebo, the mean hot flash score decreased 62% at the end of the 8-week period. An additional mean reduction in hot flash score of 26% was reported for women originally randomized to venlafaxine 37.5 mg daily. For the 75 mg and 150 mg groups, the 60% reduction in hot flash score achieved in the double-blind study was maintained throughout the continuation phase. Side effects, including mood changes, trouble sleeping, abnormal sweating, and fatigue were reported by fewer women during the continuation phase than during the baseline week.
The long-term efficacy and toxicity of venlafaxine in women with hot flashes was evaluated in two recent randomized clinical trials. In the first trial (30), 80 healthy postmenopausal women with hot flashes were randomized to placebo (n = 40) or venlafaxine ER (n = 40). Women received venlafaxine 37.5 mg daily for 1 week, followed by 75 mg daily for 11 weeks. Sixty-one women completed the study (32 in the placebo group and 29 in the treatment group). After 12 weeks of treatment, there were no statistically significant differences between the two groups in terms of hot flash frequency and severity. Dryness of the mouth, insomnia, and decreased appetite were significantly more common in the venlafaxine group. However, mental health, and vitality were significantly improved in the venlafaxine group, and 93% of women in this group chose to continue venlafaxine at the conclusion of the study. In the second trial (31), which has been published in abstract form only, 60 healthy postmenopausal women with hot flashes were randomized to placebo (n = 20), venlafaxine 37.5 mg daily (n = 22) or venlafaxine 75 mg daily (n = 18). After 3 weeks of therapy, hot flashes increased by 10% in the placebo group, while they decreased by 65 and 51% in women taking venlafaxine 37.5 mg and 75 mg, respectively. From this point on, all women in the venlafaxine groups (except six women in the 75 mg group who withdrew from the study) received 37.5 mg daily. After 4 months of treatment, hot flashes decreased by 78% in the venlafaxine group compared to 2% in the placebo group. Nausea and dryness of the mouth, the most frequent side effects, were more common in the venlafaxine groups and were dose related.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree