Background
Graves’ hyperthyroidism (GH) in pregnancy affects less than 0.5% of pregnant women. Prior to the advent of antithyroid drugs (ATDs) in the 1940s, GH perinatal mortality was as high as 45%. Perinatal mortality has improved significantly and in 2011 was reported at 1.7%. By making an early diagnosis of GH and maintaining euthyroidism during pregnancy, the physician can reduce the risk of complications to the mother, fetus, and newborn.
Pathophysiology and Natural History of Graves’ Hyperthyroidism During Pregnancy
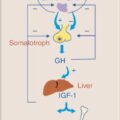
GH is an autoimmune condition in which thyroid-stimulating immunoglobulins (TSI) stimulate the thyroid-stimulating hormone receptor (TSHR), leading to increased thyroid hormone production and thyrotoxicosis. In pregnancy, TSI cross the placenta and act on the fetus’s thyroid. High titers of TSI may lead to fetal thyrotoxicosis. GH may worsen in early pregnancy, possibly secondary to human-chorionic gonadotropin (hCG) stimulation of the thyroid gland and/or elevation in TSI during the first trimester. As pregnancy progresses, changes in the immunologic response lead to a decrease in TSI and improvement in GH.
Clinical Presentation of Graves’ Hyperthyroidism in Pregnancy
Symptoms of hyperthyroidism may include palpitations, tremors, heat intolerance, weight loss, night sweats, moist skin, and loose bowel movements. Signs of GH include ophthalmopathy, diffuse goiter, and pretibial myxedema. In the absence of signs of GH, a diagnosis of hyperthyroidism from clinical symptoms alone may be difficult as many symptoms are present in normal pregnancy.
Etiology of Hyperthyroidism in Pregnancy
The etiology of hyperthyroidism in pregnancy is broad ( Box 23.1 ). GH is typically characterized by the signs and symptoms discussed above, often preceding pregnancy, a suppressed TSH, an elevated thyroxine (T4) outside of the pregnancy reference range, and presence of TSI. Laboratory tests will be discussed further later.
- ■
Graves’ hyperthyroidism
- ■
Painless thyroiditis
- ■
Subacute thyroiditis
- ■
Toxic adenoma
- ■
Toxic multinodular goiter
- ■
Gestational transient thyrotoxicosis (GTT)
- ■
Hyperemesis gravidarum
- ■
Multiple gestations
- ■
- ■
Trophoblastic disease
- ■
Hyperplacentosis
- ■
Hyperreactio luteinalis
- ■
Thyroid-stimulating hormone (TSH)-receptor mutation
- ■
TSH-producing pituitary tumor
- ■
Excessive levothyroxine (LT4) intake
- ■
Drugs
- ■
Amiodarone
- ■
Lithium
- ■
Cysteine proteases (CP) inhibitors
- ■
Interferons
- ■
Iodine
- ■
Adapted with permission from reference 86.
The most common etiology of hyperthyroidism in pregnancy is gestational transient thyrotoxicosis (GTT), which affects 1% to 5% of all pregnant women. TSH is suppressed and T4 is elevated, but unlike GH, TSI is undetectable. GTT is most often associated with hyperemesis gravidarum, which typically occurs between 4 and 8 weeks of gestation and consists of persistent nausea, vomiting, greater than 5% weight loss, dehydration, ketonuria, and electrolyte disturbances that may require hospitalization. Distinguishing between GTT and GH is important because GTT does not require treatment with ATDs and has not been associated with adverse pregnancy outcomes ( Table 23.1 ). The clinical course of GTT parallels the level of hCG, resolving by the end of the first or middle of the second trimester. , Women with a prior history of GTT are at increased risk for a repeat episode.
| GTT | GH | |
|---|---|---|
| Hyperthyroid symptoms prior to pregnancy | − | + |
| Hyperthyroid symptoms during pregnancy | +/− | + |
| Nausea/vomiting | ++ | −/+ |
| Goiter/ophthalmopathy | − | + |
| Thyroid-stimulating immunoglobulins | − | + |
Laboratory Measurements and Considerations in Pregnancy
THYROID-STIMULATING HORMONE
Physiologic changes occur in normal pregnancy that lead to a decrease in TSH. hCG is secreted from the placenta and, due to a shared alpha subunit with TSH, acts on the TSHR. This leads to increased thyroxine hormone production, which feeds back to the pituitary and leads to a decrease in TSH. Consequently, the TSH reference range in pregnancy is shifted down by approximately 0.4 mIU/mL compared to the nonpregnancy reference range. TSH is typically suppressed in GH.
Biotin, a water-soluble B vitamin (B7), is used in several hair and nail supplements. Biotin may interfere with biotin streptavidin immunoassays used in commercial assays to measure TSH and thyroid hormones. Depending on the type of assay, biotin may cause a falsely depressed hormone level (TSH) or elevated free T4 (FT4) level, mimicking hyperthyroid laboratory tests. Withholding biotin for 8 hours may be sufficient to prevent interference with thyroid function laboratory testing. However, biotin is generally withheld for approximately 3 days prior to thyroid function tests (TFTs).
THYROXINE
An increase demand in thyroid hormone requirement in pregnancy is met by an estrogen-mediated increase in hepatic thyroid-binding globulin (TBG) synthesis. Elevation in TBG, along with decreased albumin concentration in the latter half of pregnancy, may contribute to falsely low FT4 immunoassay levels. When using automated immunoassays by nonequilibrium methods, trimester-specific reference ranges should be applied. Alternatively, the FT4 index (FT4I) or total T4 have been shown to be reliable estimations of thyroxine concentration in the latter half of pregnancy. The nonpregnant T4 reference range is adjusted by 1.5 to give the pregnancy reference range (i.e., 4 to 10 µg/dL becomes 6 to 15 µg/dL in pregnancy).
TRIODOTHYRONINE (T3)
T3 is rarely indicated in the assessment of patients with GH. An exception is if the TSH is suppressed in the setting of a normal T4 level, which can be seen in the case of exogenous T3 or autonomous functioning nodule.
THYROID-STIMULATING IMMUNOGLOBLULINS (TSI) AND THYROID-STIMULATING HORMONE RECEPTOR ANTIBODIES (TRAb)
There are two methods to measure antibodies of the TSH receptor: a bioassay, which measures TSI; and a competitive receptor immunoassay, which measures TSHR antibodies (TRAb). The difference between the two assays is that TSI measures only stimulating antibodies, whereas the TRAb assay measures all antibodies to the TSHR including stimulating, neutral, and blocking antibodies. Although TSI is the pathogenetic hallmark of GH, TRAb may also be used in the diagnosis of GH because in a clinically hyperthyroid patient, the majority of the TRAb will be stimulating antibodies.
The TSI bioassay is a functional assay that measures cyclic adenosine monophosphate (cAMP), a downstream product of TSI binding to the TSHR, by radioimmunoassay or chemiluminescent assay. The TRAb assay is a competitive immunoassay that detects antibodies by their capacity to compete for binding of radiolabeled TSH to the TSH receptor. Both assays have sensitivities and specificities greater than 98%. , The TSI bioassays have the advantage of detecting a functional property of the antibodies. The TRAb competitive receptor assay may be more readily available and less expensive ( Table 23.2 ). Going forward in this chapter, the term TSI is interchangeable with TRAb.
| TRAb | TSI | |
|---|---|---|
| Antibodies detected | Stimulating, neutral, blocking | Stimulating |
| Type of assay |
|
|
| Sensitivity and specificity | >98% , | >98% , |
| Considerations |
|
|
TSI should be measured at presentation in pregnant women with active GH with or without ATD treatment, a history of GH previously treated with radioactive iodine ablation (RAIA) or surgery, or a past pregnancy with a fetus/neonate with thyroid dysfunction, because TSI greater than three times the upper limit of normal (ULN) is associated with increased risk of fetal and neonatal hyperthyroidism. Women who are currently euthyroid or hypothyroid who were treated with surgery or RAIA may continue to have elevated TSI because TSI may remain elevated for years, especially after RAIA. Women who are euthyroid secondary to treatment with ATDs at the time of diagnosis of pregnancy are unlikely to have elevated TSI levels.
Management and Treatment Options
The mainstay of treatment of GH in pregnancy is ATDs. The concern with ATDs in the setting of pregnancy is the association of both ATDs, propylthiouracil (PTU) and methimazole (MMZ), with congenital malformations ( Table 23.3 ). , – The maximum sensitivity to teratogenic effects of ATDs is during organogenesis, gestational weeks 6 to 10. If possible, ATDs should be avoided during this time. The physician may consider withholding ATDs at the time of diagnosis of pregnancy in certain patients. Women who have been treated for greater than 6 months, have a prepregnancy TSH within the reference range, and TSI levels less than three times ULN may be able to maintain euthyroidism without ATDs. If ATD is withheld, TFTs should be monitored each week until week 12 then every 2 to 4 weeks as clinically indicated. If hyperthyroidism recurs in the first trimester, PTU should be started.
| Propylthiouracil (PTU) | Methimazole (MMZ) |
|---|---|
| Congenital malformations | |
|
|
| Maternal complications | |
| Skin rash, pruritus, migratory polyarthritis, lupus-like syndrome, cholestatic jaundice, agranulocytosis , | |
| |
If ATDs may not be stopped, PTU is the drug of choice in the first trimester and is dosed at 50 to 150 mg orally every 8 hours, with TSH and T4 checked every 2 to 4 weeks as clinically indicated. Although both PTU and MMZ have similar effectiveness, rates of crossing the placenta, and incidence of congenital malformations (∼2% to 4%), the congenital malformations associated with PTU (e.g., preauricular cysts and urinary tract abnormalities), are considered less severe and surgically correctable compared with those of MMZ (e.g., aplasia cutis, choanal and esophageal atresia, omphalocele, dysmorphic facies, athelia, and ventral septal defects) ( Table 23.2 ). , , – Both PTU and MMZ are associated with agranulocytosis, and patients should be educated to monitor for symptoms such as fever and sore throat.
- ■
Maternal congestive heart failure
- ■
Thyroid storm
- ■
Miscarriage
- ■
Gestational hypertension
- ■
Preeclampsia
- ■
Premature delivery
- ■
Placental abruption
- ■
Premature rupture of membranes
- ■
Intrauterine growth restriction
- ■
Central hypothyroidism in the neonate
- ■
Still birth
- ■
Postpartum bleeding
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree