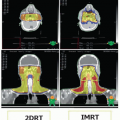
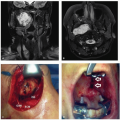
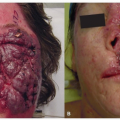
Landmark Trial |
Treatment Arms |
Summary of Results |
Impact |
VA Larynx Trial2 |
Phase 3 randomized control trial comparing neoadjuvant chemotherapy + radiation vs. surgery and postoperative radiation in squamous cell carcinoma of the larynx |
2-year survival was 68% in each arm with 64% of patients in the chemoradiotherapy arm maintaining larynx preservation at early follow-up |
Established chemoradiotherapy as an organ-preserving option for patients with cancer of the larynx |
EORTC HPX Trial3 |
Phase 3 randomized control trial comparing neoadjuvant chemotherapy + radiation vs. surgery and postoperative radiation in squamous cell carcinoma of the larynx and hypopharynx |
3-year survival was 43% in surgical arm and 57% in chemoradiotherapy arm |
Established chemoradiotherapy as an organ-preserving option for larynx and hypopharynx cancer patients |
RTOG 91-114,5 |
Phase 3 randomized control trial comparing (1) neoadjuvant chemotherapy and radiation, (2) concurrent chemotherapy and radiation, or (3) radiation alone |
Survival was similar across all three arms, but laryngeal preservation was highest for the concurrent arm (88%), neoadjuvant (74%), and radiation alone (70%) |
Established concurrent chemotherapy as a nonoperative standard of care with radiation |
MACH Meta-analysis12 |
Meta-analysis of 15 trials (6,515 patients) evaluating altered fractionation vs. conventional fractionation (no chemotherapy) |
Altered fractionation improved survival (3.4% at 5 years) and local-regional control (6.4% at 5 years) |
Established that with radiation alone regimens, altered fractionation is better than conventional fractionation |
RTOG 012929 |
Phase 3 randomized control trial comparing altered fractionation + chemotherapy vs. conventional fractionation + chemotherapy. Post hoc analysis of HPV as prognostic factor |
Arms were equivalent. 3-year overall survival was 82% in the HPV-positive vs. 57% in the HPV-negative patients. OPSCC patients can be categorized into low-, intermediate-, and highrisk groups |
Altered fractionation can be used with concurrent chemotherapy. Treatment of OPSCC patients will be tailored according to risk group |
GORTEC 99-02118 |
Phase 3 randomized control trial of (1) standard fractionation and chemotherapy, (2) hyperfractionation and chemotherapy, and (3) hyperfractionation alone |
Two chemotherapy arms had similar and significantly better outcomes compared to radiotherapy alone. Acute toxicity was worse in the two hyperfractionated arms |
Altered fractionation should be used with caution with concurrent chemotherapy. Altered fractionated radiotherapy alone showed inferior outcomes |
MACH-NC813,14 |
Meta-analysis of 87 trials (16,485 patients) that evaluated the efficacy of definitive chemoradiotherapy |
The addition of chemotherapy to radiation improved survival by 4%-5%. Concurrent chemotherapy had the greatest impact on survival. Neoadjuvant and adjuvant chemotherapy did not significantly affect survival |
Established that concurrent chemotherapy can be given with radiotherapy for the definitive treatment of stage III-IV head and neck squamous cell cancer |
EORTC 22931 and RTOG 950117,18,19 |
2 phase 3 randomized control trials of postoperative radiation vs. postoperative radiation + chemotherapy |
The addition of chemotherapy improved local-regional control and survival in patients with + margins or extranodal extension |
Established that chemotherapy can be given with radiotherapy after surgery in patients with high-risk pathologic features |
Bonner et al.26,27 |
Phase III randomized control trial of radiation vs. radiation + cetuximab. First of its kind study in head and neck cancer, allowing for the evaluation of molecular targeted therapy in combination with radiation |
The addition of cetuximab to radiation resulted in an absolute improvement in survival of 10% without increased toxicities (except acneiform rash) |
Study was the first of its kind in head and neck cancer, allowing for evaluation of molecular targeted therapy in combination with radiation |
RTOG 1016 |
Phase III randomized control trial of radiation + cisplatinum vs. radiation + cetuximab in HPV-associated OPSCC, closed to accrual |
Primary end point is noninferiority and less toxicity in the radiation + cetuximab arm |
If the radiation + cetuximab arm is noninferior and less toxic, this regimen would offer a deintensified option for patients with HPV-associated OPSCC |
ECOG 3311 |
Ongoing phase 2b randomized study of transoral surgery followed by observation (low risk), chemoradiotherapy (high risk), or randomization to 50 Gy vs. 60 Gy (intermediate risk) in HPV-positive OPSCC |
Primary end point is 2-year PFS. Secondary end point is 2-year functional outcome |
Study opened in 2014 |