Author, reference, country
Analytical system
Invitation strategy
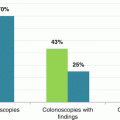
Number with faecal haemoglobin concentration and endoscopy data
Recruitment from primary/secondary care
McDonald et al., [51], Scotland
OC-Sensor
Phone call of invitation by research nurse after referral from primary care for endoscopy
280
0/100
Godber et al., [52], Scotland
HM-JACKarc
Request for FIT included with bowel preparation materials on invitation to, and appointment for, endoscopy.
484
0/100
Mowat et al., [53], Scotland
OC-Sensor
Request for FIT analysis made in primary care along with documentation of symptoms and referral for endoscopy
755
100/0
Cubiella et al., [54], Spain
OC-Sensor
Consecutive patients with gastrointestinal symptoms referred for colonoscopy from primary and secondary healthcare.
787
Both—but percentages not documented.
Auge et al., [55], Spain
HM-JACKarc
Consecutive patients who attended for colonoscopy for the investigation of symptoms or polyp surveillance.
208
Not documented. However, tests requested on attendance at secondary care clinic.
Rodriguez-Alonso et al., [56], Spain
OC-Sensor
Patients referred for diagnostic colonoscopy after consultation in which an exhaustive interview was performed by a gastroenterologist.
1003
66.3/33.7
Widlak et al., [57], England
HM-JACKarc
Patients recruited from “two week wait” clinics after primary healthcare referral.
430
0/100
3.4.3.1 The Study of McDonald et al.
This study seems to be the first aimed at determining whether f-Hb found by a quantitative FIT can assist in deciding which patients with lower abdominal symptoms would benefit from endoscopy [51]. Among 739 invited patients, FIT and endoscopy were completed by 280 (median age 63 years, 59.6% women). Six (2.1%) participants had CRC, 23 (8.2%) had AA, 31 (11.1%), non-advanced adenoma (NAA) and 26 (9.3%) IBD as the most serious diagnosis. As with findings in screening, f-Hb was clearly related to severity of colorectal disease since those with CRC had a median f-Hb of >200 μg Hb/g faeces and those with CRC + AA + IBD having a median f-Hb of 15 μg Hb/g faeces, which were both significantly higher than that of all remaining participants without SCD. The NPV using a cut-off f-Hb of 10 μg Hb/g faeces were 100%, 94.4% and 93.9% for CRC, AA and IBD respectively. The AUC for all four clinical groups examined, with the more important colorectal diseases classified as positive, ranged from 0.734 to 0.671; these AUC suggest only a “fair” to “poor” rule-in diagnostic test. However, the high NPV, particularly for the group that is considered of most clinical interest, that is SCD (CRC + AA + IBD), was 88.1%. In consequence, a negative FIT result with f-Hb <10 μg Hb/g faeces means that SCD is unlikely to be present and, when used as a rule-out test, could facilitate patient reassurance of patients and saving of colonoscopy resources.
3.4.3.2 The Study of Godber et al.
This study followed up that described above in a different part of Scotland and with a different FIT system [52]. Again designed to determine whether patients with lower abdominal symptoms can be investigated quickly using f-Hb, 909 consecutive patients referred from primary care for colonoscopy were invited: 507 submitted samples for f-Hb and a colonoscopy was completed in 484 patients. As with the study of McDonald et al. [51], those with CRC, AA or IBD had higher f-Hb than the group of 243 with normal colonoscopy plus the 196 patients with less significant clinical findings. Again, using a f-Hb cut-off of 10 μg Hb/g faeces, for the group with SCD, the test was a poor rule-in test with low sensitivity (68.9%) and PPV (26.3%,), but the NPV of 96.2% confirmed that FIT was a good rule-out test for SCD. The clinical characteristics at different f-Hb cut-offs were examined and, as expected, as the cut-off f-Hb was increased, sensitivity fell and specificity rose; while the PPV rose, the NPV remained very high at all f-Hb cut-offs. Interestingly, the 11 (2.2%) patients with CRC all had f-Hb >190 μg Hb/g faeces and sensitivity was therefore 100% for CRC, as found by McDonald et al. [51]. It was suggested that it might be that FIT could be of considerable value as either a rule-in for CRC on its own or a rule-out investigation for SCD.
3.4.3.3 The Study of Mowat et al.
This investigation studied the diagnostic accuracies of f-Hb and f-C in a group of symptomatic patients [53]; only the f-Hb component of this work will be addressed in this section of the Chapter. The PPV of referral symptoms for a diagnosis of CRC were not high and the values for specific symptoms were: palpable mass: 50.0%, weight loss: 14.3%, anaemia: 9.0%, rectal bleeding: 4.3%, abdominal pain: 3.6%, diarrhoea: 2.4%, and altered bowel habit: 2.2%. Rectal bleeding had a PPV of 21.0% for any SCD, but only one-third of these had undetectable f-Hb. As expected from the known overlap of symptoms in those with and without SCD, the most common findings at colonoscopy were: normal in 241 (33.2%), diverticular disease in 190 (25.2%), haemorrhoids in 98 (13.0%), NAA in 65 (8.6%), AA in 41 (5.4%), IBD in 34 (4.5%) and CRC in 28 patients (3.7%). In total, 1043 patients returned samples and f-Hb was detectable, that is greater than 0 μg Hb/g faeces, in 57.6%; 755 patients (median age 64 years, 54.6% women) returned faecal samples and completed colonic investigations and 103 patients were found to have SCD. Using a cut-off of “detectable” f-Hb, NPV were 100%, 97.8% and 98.4% for CRC, AA and IBD, respectively. It was concluded that undetectable f-Hb is a good rule-out test for SCD and could guide who requires further investigation. However, in laboratory medicine, the requirement is to fully conform to ISO 15189—“medical laboratories—particular requirements for quality and competence”—means that only results greater than the lower limit of the analytical working range laid down by the manufacturer, best described as the “limit of quantitation”, can be reported as concentrations [58]. Thus, while interesting for research studies, a cut-off of detectable f-Hb cannot be used in clinical practice, and only results for the OC-Sensor of <10 μg Hb/g faeces can be considered undetectable and only those ≥10 μg Hb/g faeces can be reported as numerical data. However, Mowat et al. also give comprehensive data using 10 μg Hb/g faeces as the cut-off. NPV for CRC, AA, IBD and SCD were 99.5%, 96.5%, 98.4% and 94.4%.: in contrast to previous studies in Scotland [51, 52], cases of CRC (3 of 28) would be missed using this cut-of as would 20 of 40 of AA and 9 of 34 of IBD, that is, 32 of 102 of those with SCD. Most interestingly, the three missed cases of CRC were all women (personal communication): this may reflect that fact that f-Hb is lower in women than in men [44, 45], possibly due to slower transit time of faeces passing through the colon [59].
3.4.3.4 The Study of Cubiella et al.
In this study [54], a f-Hb cut-off of 20 μg Hb/g faeces was used, rather higher than that in the three studies done in Scotland [51–53]; the rationale probably being that this is the most common f-Hb cut-off used in asymptomatic screening programmes. The novel aspect of this study is that FIT to detect CRC was compared with the then current NICE [14] and SIGN [15] referral guidelines. In 787 symptomatic patients referred for colonoscopy, patients were assessed to see if they met NICE and SIGN referral criteria and all patients collected one sample for an estimate of f-Hb. CRC was detected in 97 (12.3%) patients; 241 (30.6%) had f-Hb ≥20 μg Hb/g faeces and 300 (38.1%) and 473 (60.1%) met NICE and SIGN referral criteria. As shown in other studies discussed earlier, the median f-Hb was found to be statistically significantly higher in individuals with a CRC than the others. The important finding here is that f-Hb had a higher sensitivity for CRC (87.6%) than NICE (61.9%) and SIGN criteria (82.5%). The specificity of f-Hb was also higher than NICE and SIGN criteria (77.4%, 65.2% and 42.7%). It was concluded that f-Hb is more accurate for the detection of CRC than the then current NICE and SIGN referral criteria in symptomatic patients referred for colonoscopy. In view of the overlap of symptoms in patients with and without SCD, this is probably hardly surprising, since NICE and SIGN referral criteria are based on age and symptoms and other clinical information rather than f-Hb. However, combinations of symptoms and f-Hb might have advantages and this will be discussed later in this Chapter. Finally, the authors of this study concentrated on the use of f-Hb as a rule-in test for CRC, but there are data on the NPV of f-Hb and NICE and SIGN referral criteria for both CRC and advanced neoplasia (AN), defined as CRC plus AA: these were 97.8%, 92.4% and 94.5% respectively for CRC and 90.8%, 82.1% and 83.6% for AN respectively. Thus, it was actually demonstrated that f-Hb is a good rule-out test for AN.
3.4.3.5 The Study of Auge et al.
Auge et al. [55] provided a unique perspective on use of FIT in assessment of the symptomatic since two samples were obtained on each patient for FIT. The aims of this study were to evaluate the diagnostic yield for what was termed advanced colorectal neoplasia (ACRN), equivalent to CRC plus AA, in symptomatic patients, assessing the f-Hb of the first of two samples (FIT/1) and the higher concentration of two FIT samples (FIT/max). Samples were collected from two consecutive bowel motions from 208 symptomatic patients who required colonoscopy. Patients were categorised into two groups: patients with any ACRN and those with other diagnoses or normal colonoscopy. Colonoscopy detected ACRN in 29 patients. FIT/1 and FIT/max were significantly higher than in patients with NAA, other findings and normal colonoscopy, confirming the now well-established relationship between f-Hb and colorectal disease severity, This study also showed that higher f-Hb are found in men with lower abdominal symptoms compared with women, consistent with other studies [44–47], Similarly, a higher clinical sensitivity and PPV for ACRN was observed in men than in women and the NPV was lower in men, which perhaps implies that different f-Hb cut-offs should be used for the different sexes. The AUC of FIT/1 and FIT/max were 0.71 and 0.69, respectively, again showing that FIT are less than good tests for detection of disease. However, undetectable FIT/1 gave NPV of 95.0%, but increasing the FIT/1 cut-off to 10 μg Hb/g faeces decreased the NPV to 89.2%. Similar results were obtained using FIT/max with a cut-off of 20 μg Hb/g faeces, providing NPV of 89.3% and 89.0%. The authors concluded that undetectable FIT is a good strategy to rule out ACRN in symptomatic patients, but the problems of using this and complying with ISO 15189 standards in routine practice have already been discussed. Considering which of one or two samples is better, it was shown that the diagnostic yield for a rule-in test of collecting two samples for FIT can be achieved with one sample, but a lower faecal f-Hb cut-off is required. Interestingly, the FIT/max result was not concordant with the FIT/1 result in 39.2% of patients: this might indicate that f-Hb has considerable within-subject variation and this would be very worthy of investigation as has been done recently for f-C [60].
3.4.3.6 The Study of Rodríguez-Alonso et al.
This was the second study done comparing quantitative FIT with NICE [14] and SIGN [15] guidelines for referral [56]. The authors prospectively studied 1054 symptomatic patients referred for colonoscopy from both primary and secondary healthcare. NICE and SIGN guidelines detected 46.7% and 43.3% of cases of CRC while f-Hb ≥15 μg Hb/g detected 96.7% of cases. Male sex, age and f-Hb ≥10 μg Hb/g were independent predictive factors of advanced neoplasia. As found in all studies, symptoms often used as referral criteria were poor predictors of both CRC and AN. The performance characteristics of NICE, SIGN and f-Hb concentration ≥ 15 μg Hb/g for the detection of AN were: sensitivity: 38.3%, 36.1% and 57.1% and specificity: 71.8%, 69.5 and 86.6%, respectively. This study explored the known relationship between f-Hb and severity of disease in considerable detail. AUC for f-Hb in the detection of CRC and AN were 0.94 and 0.76 and the optimal f-Hb cut-off for CRC and AN detection were ≥15 μg Hb/g and ≥10 μg Hb/g, respectively. If f-Hb ≥ 15 μg Hb/g was used as the criterion for referral, only 19.2% of the patients would need urgent assessment for detection of 96.6% of CRC, one of 30 patients being missed. At a cut-off f-Hb of ≥10 μg Hb/g, 22.5% of the patients would need urgent assessment for detection of 61.6% of the AN. It was concluded that a FIT-based strategy performs better than the then current high-risk symptoms-based approaches for fast-tracking referrals for investigation of suspected cancer referrals. The authors also considered that, with NPV of 99.9% for CRC and 93.0% for AN at a f-Hb cut-off of 10 μg Hb/g faeces, f-Hb could also provide a single and excellent rule-out test, thus sparing those with lower abdominal symptoms who do not have SCD from undergoing a colonoscopy. This would save many referrals to colonoscopy and would allow fast-track referrals to be seen more quickly. For the first time, risk-scoring involving f-Hb in assessment of the symptomatic, along with sex and age, was investigated and it was shown that this could accurately estimate the risk of AN. Risk-scoring will be discussed later in this Chapter.
3.4.3.7 The Study of Widlak et al.
Similar to Mowat et al. [52], the aim was to investigate f-Hb and f-C to identify patients for referral for colonic investigation who are most likely to have CRC; here, only the data on f-Hb will be discussed. This study involved patients recruited via the “two-week wait” clinics rather than directly in primary healthcare. Of 430 study participants, median age 67 years, 176 (64.1%), had change in bowel habit, 185 (43.0%) rectal bleeding, 129 (30.0%) abdominal pain, 74 (17.2%) anaemia, 68 (15.8%) weight loss and 95 (22.1%) family history of CRC. Twenty-four patients with CRC and one patient with high grade dysplastic adenoma were grouped together as cancer. At a cut-off of 7 μg Hb/g faeces, f-Hb returned a sensitivity of 84% and specificity of 93% for CRC detection with an AUC of 0.94 and NPV of 99%. For adenoma detection, with no other pathology on colonoscopy or CT, f-Hb had a sensitivity of 69% and specificity of 56% with AUC of 0.70 and NPV of 94%. As found in all other studies described here, the evidence was that f-Hb alone has considerable potential for use in risk stratifying symptomatic patients.
3.4.4 Conclusions from Studies on Quantitative FIT
It would be of significant value for a meta-analysis to be performed on quantitative FIT in assessment of the symptomatic, but that is beyond the scope of this Chapter. However, the results of the seven published studies known to us to date provide evidence that unequivocally demonstrates the following:
in patients with lower abdominal symptoms, f-Hb is higher in CRC than in those with AA and IBD, and f-Hb in these is higher than in those with less significant colorectal disease such as NAA, haemorrhoids and simple diverticular disease and those with no abnormalities found on colonoscopy,
high f-Hb is not only found in patients with SCD but in some who have less significant pathology and no abnormality, and so the PPV is not optimal,
the higher the f-Hb cut-off used for referral for colonoscopy, the lower is the sensitivity, positivity rate and the NPV, and the higher the specificity and PPV,
f-Hb cut-off at the limit of quantitation documented by the manufacturers of the FIT system used should be applied to give highest sensitivity for detection of SCD and highest NPV, albeit at the expense of specificity and PPV,
at such f-Hb cut-offs, few CRC and some AA and IBD will be missed and safety-netting, which will be discussed later in this Chapter, is mandatory,
f-Hb provides a good rule-in test for CRC and a patient with a f-Hb above the lower limit of the analytical working range should be referred for urgent colonoscopy,
f-Hb provides a good rule-out test for SCD: a result below the cut-off means that SCD is unlikely and the patient can be reassured regarding the absence of disease and not referred immediately for colonoscopy, although other strategies are options, as discussed later,
f-Hb is better at detection of CRC than some guidelines based upon symptoms and age and other factors for referral from primary care when CRC is suspected,
one sample is sufficient for detection of most SCD,
men and women with symptoms have different clinical outcomes at a single f-Hb cut off,
one study using two samples shows that there may be considerable within-subject variation of f-Hb from day to day and this is worthy of exploration as has been recently done for f-C [60] and
ubiquitous use of f-Hb as the initial investigation in primary (and secondary) healthcare could undoubtedly help direct colonoscopy resources to those who would benefit most.
It might appear that use of central laboratory-based quantitative FIT would cause some, albeit small, delay in reporting of f-Hb results as compared to POCT qualitative FIT. This might delay diagnosis and, more importantly, might cause some drop out and loss of patients from pathways after presentation since they might not wish to undertake faecal collection, in spite that quantitative FIT have easy to use, hygienic collection devices and only one sample is needed. It would therefore be of interest to undertake further research comparing qualitative and quantitative FIT. Further, in view of the contentious issue of application of tests for occult blood in faeces in patients presenting in primary care, NICE established a Diagnostics Assessment Committee on quantitative faecal immunochemical tests to guide referral for colorectal cancer in primary care; the final guidance (DG30) has been published very recently [61]: The recommendations include the following:
- 1.
Quantitative faecal immunochemical tests are recommended for adoption in primary care to guide referral for suspected colorectal cancer in people without rectal bleeding who have unexplained symptoms but do not meet the criteria for a suspected cancer pathway referral outlined in NICE’s guideline on suspected cancer (NG12).
- 2.
Results should be reported using a threshold of 10 μg of haemoglobin per gram of faeces. Companies should provide advice about the performance characteristics of the assays to laboratories, and ensure standardisation of results.
It is pleasing to see that, in general, the guidelines are in accord with the material in this Chapter.
3.5 Comparison of Faecal Haemoglobin, Faecal Calprotectin and M2-PK
3.5.1 Faecal Calprotectin
Calprotectin is one of the S100 family of proteins which occurs in large amounts in neutrophil granulocytes, where it accounts for 5% of total proteins and 60% of cytoplasm proteins: f-C is released into the gut lumen in the presence of inflammation. Thus, when f-C is detected, a patient might have IBD such as Crohn’s disease or ulcerative colitis (UC). These diseases present symptoms similar to those seen in irritable bowel syndrome (IBS). But, in view of the major clinical differences, it is important to distinguish between IBD and IBS.
Testing using f-C is recommended by NICE as an option to help distinguish between IBD and IBS [62], the rationale being that many with IBS have unnecessary colonoscopy before the true nature of their problem is diagnosed. The guideline states that using f-C will mean that most people with IBS will be diagnosed without the need for these investigations. The NICE guideline is that: f-C testing is recommended as an option to support clinicians with the differential diagnosis of IBD and IBS in adults with recent onset lower gastrointestinal symptoms for whom specialist assessment is being considered, with the important caveat: only when cancer is not suspected, having considered the risk factors (for example, age) described in the then current NICE referral guidelines for suspected CRC [14].
Historically, f-C has often been suggested as a useful investigation for CRC asymptomatic screening [63], but here, only the use in assessment of SCD in the symptomatic will be discussed. It is a timely topic, since, in a very recent study [64], it was suggested that there would be a sufficient inflammatory component in patients with symptomatic CRC resulting in a raised f-C. In 654 patients (median age 69 years, female 56%), who completed f-C and evaluation of the colon using a variety of approaches, the NPV for CRC and AN were 98.6% and 97.2%. By altering the cut-off f-C to have an NPV of 97.0%, the PPV for CRC increased from 8.7% to 13.3%. It was concluded that f-C has a high NPV for CRC and AA in patients with suspected CRC and the 27.8% of patients who had a normal f-C could safely have been spared a two-week wait referral. It was proposed that, in part since f-C was available and there was current confusion regarding the use of faecal tests for occult blood [17–20], the addition of f-C into current symptom-based assessment has potential to increase CRC detection and be both clinically and cost effective. However, studies on f-C as compared to f-Hb are more germane to this Chapter.
3.5.2 Comparison of Faecal Haemoglobin with Faecal Calprotectin
Two studies performed in The Netherlands described in detail earlier investigated POCT f-Hb and f-C to distinguish those with from those without SCD [36, 37]. It was concluded that a diagnostic strategy with routine clinical data and f-Hb alone might safely rule out SCD and prevent unnecessary referral for colonoscopy in approximately one-third of patients.
Mowat et al. [54] showed that using a cut-off f-C of 50 μg/g, as often used in differentiating possible IBD from IBS, the positivity rate was 62.0%, the PPV for SCD was 16.9% and the PPV for IBD was 6.4%. The NPV for IBD was 98.9%. However, 25 cases of SCD (5 CRC, 17 AA and 3 IBD) had f-C below this cut-off and would have been missed if f-C alone was used. This study examined combinations of f-C at cut-offs of 50 μg/g and 200 μg/g and undetectable f-Hb. The conclusion was that faecal tests requested in primary care can provide a reliable prediction of the absence of SCD and, importantly, that f-Hb is superior to f-C and enables, with a single faecal test, an objective assessment of the need for, and urgency of further investigation.
Similarly, Widlak et al. [57] investigated f-Hb and f-C alone or combined as a method to identify those patients for referral for colonic investigation who were most likely to have CRC. As discussed earlier, it was stated that the diagnostic accuracy of f-Hb indicated that undetectable f-Hb is a good rule-out test for CRC, with a NPV of 99%, and was also judged as being far more accurate than the current referral pathway based on symptoms alone. Both f-Hb and f-C also offered a good rule-out option for IBD and microscopic colitis with NPV of 100% and 99% respectively. It was said that this means that treatable (benign) symptomatic colonic pathology would not be missed either; however, adding f-C to f-Hb showed no additional benefit for the added cost of analysing two tests. The conclusion was that an undetectable f-Hb was sufficiently sensitive to exclude CRC, with higher concentrations in left-sided lesions, while f-C in combination did not appear to provide additional diagnostic information.
As discussed earlier, in a recent study from Sweden [38], a qualitative FIT and a f-C test were compared for detecting CRC, AA and IBD in primary care, along with assessment of the value of combining these with anaemia and iron-deficiency tests. The sensitivity of f-C for detection of CRC and IBD was stated to be too low, at the cut-off of 100 μg/g recommended, for use as a diagnostic aid on its own. With a cut-off f-C of 50 μg/g, the performance of f-C and FIT was similar, but four cases of AA were missed when compared to the FIT. It was concluded that combining f-C and FIT showed no improvement over the FIT alone, which detected a significantly larger proportion of CRC, AA and IBD than f-C (0.92 versus 0.46).
There have now been studies on the use of f-Hb in assessment of UC. It has been suggested that consecutive measurements of f-Hb in quiescent UC patients who achieved mucosal healing (MH) with negative f-Hb results would help identify patients with clinical relapse whose symptoms had not yet presented [65]. It has also been shown in a study assessing the risk of relapse in UC patients in clinical remission using mucosal status and f-Hb, negative f-Hb results one year or more after remission induction correlate with complete MH and better prognosis; it was considered that performing FIT one year after remission induction might be useful for evaluating relapse risk [66]. Further, a comparison of the predictive ability of f-Hb and f-C for MH in UC showed that both f-Hb and f-C were significantly correlated with the Mayo endoscopic sub-score [67]; it was concluded that f-Hb and f-C can both efficiently predict MH in UC, but f-Hb appears to be more sensitive than f-C for predicting a Mayo endoscopic sub-score of 0.
These early data suggest that f-Hb is superior to f-C in detection of SCD and more useful in UC. Consequently, it has been suggested [25] that, perhaps in the near future, when quantitative f-Hb becomes more widely available for triage of symptomatic patients, f-C will be used mainly in the monitoring of patients with known IBD rather than in the diagnostic setting. Further research seems warranted to compare f-Hb and f-C and in a wide range of GI disorders.
3.5.3 M2-PK and Use with Faecal Haemoglobin
M2-PK is the abbreviation used for the dimeric form of the pyruvate kinase isoenzyme type M2, a key enzyme in metabolism of tumours. Although M2-PK can be raised in many tumour types, increased faecal M2-PK has been investigated as a method of screening for CRC [63].
There appears to be only one study on use of M2-PK in assessment of patients with symptoms. Parente et al. explored a combination of f-Hb, obtained using the HM-JACK system (Kyowa-Medex), M2-PK and f-C [68]. All tests were performed on a single faecal sample from 280 patients aged 50–80 years, without any dietary restriction, before colonoscopy; 47 had CRC and 85 patients had one or more AA. CRC was associated with a highly significant increase in M2-PK which correlated with Dukes’ staging. For CRC detection, f-Hb was the test with the highest specificity and PPV (89% and 53%), whereas M2-PK had the highest sensitivity and NPV (87% and 96%); f-C showed performance similar to M2-PK in terms of sensitivity and NPV, but had lower specificity. It was suggested that the best combination to predict the risk of CRC was f-Hb plus M2-PK. Further studies comparing f-Hb with M2-PK, alone or in combination, would be useful.
3.6 Risk Prediction Models
As well described in a review by Vega et al. [69] on “CRC diagnosis: pitfalls and opportunities”, many factors affect incidence and mortality of CRC, including sex, age, socio-economic status, diet, alcohol consumption and tobacco use. Since the same spectrum of symptoms occur in those with and without SCD, it is attractive to think that risk-scoring systems which include both factors affecting incidence and mortality and/or symptoms might be effective. Williams et al. recently reviewed risk prediction models which combine multiple risk factors and symptoms that have the potential to improve timely diagnosis [70]. The aim was to systematically identify and compare the performance of models that predict the risk of primary CRC among symptomatic individuals; 18 papers describing 15 risk models were included. Models with good discrimination had been developed in both primary and secondary care populations and most contained variables which are easily obtainable in a single consultation. It was suggested that further research was needed to assess clinical utility before incorporation into practice. Some include results of gFOBT but, in this Chapter, only the few models to date which include f-Hb are discussed.
The study from The Netherlands using POCT FIT that evaluated the diagnostic value of history taking, physical examination, and simple blood testing and, subsequently, the added value of the POCT FIT, in that order, has been discussed previously [37], but models using quantitative estimates of f-Hb are of more current interest.
A very simple model was developed by Rodríguez-Alonso et al. [56] to provide a risk score for AN. The model incorporates age in five groups, sex and f-Hb as <10 μg Hb/g faeces or ≥10 μg Hb/g faeces. The risk score has a range of 0–11 points based on the sum of the scores according to the presence or absence of risk factors (see Table 3.2). The AUC for the risk score was 0.79. The internal validity of the model was assessed by a split sample procedure with 680 individuals (67.8%) in the training set, of whom 91 had AN, and 323 individuals (32.2%) in the validation set, of whom 42 had AN. Good agreement was found between the risk of AN predicted by the model and the observed prevalence of AN. The optimal cut-point for the score derived from the whole study population was ≥5, with 75.9% sensitivity and 72.0% specificity. Identical values were found for the score derived from the training set. The corresponding values in the validation set were 88.1% sensitivity and 63.3% specificity.
Table 3.2
Pocket chart for risk score calculation
Risk factor | Points |
|---|---|
Age
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

| |