Chemotherapy-Related Nausea and Vomiting
Ann M. Berger
Rebecca A. Clark-Snow
Nature of the Problem
Currently, there are many efficacious antiemetic regimens for nausea and vomiting produced by chemotherapeutic agents. In a study conducted in 1983, patients with cancer ranked nausea and vomiting as the first and second most severe side effects of chemotherapy, respectively (1). After the emergence of new antiemetic agents and alterations in chemotherapeutic regimens, patients’ perceptions of the most severe side effects were modified. In a 1993 study, 155 patients with cancer, receiving chemotherapy, reported that they experienced an average of 20 physical and psychosocial symptoms: nausea was ranked as the most severe symptom and vomiting as the fifth (2). Therefore, nausea is also an important efficacy parameter when evaluating an antiemetic.
Use of these antiemetic agents has decreased the incidence and severity of nausea and vomiting induced by chemotherapy; however, these agents have not totally prevented the problem. Chemotherapy-induced nausea and vomiting (CINV) continue to remain a concern for patients receiving cancer treatment. Grunberg et al. (3) reported that in spite of the use of modern antiemetics, CINV continues to be a problem for a significant number of patients receiving cancer chemotherapy. It was observed that the frequency of CINV, particularly delayed nausea and vomiting, is underestimated by oncology physicians and nurses (3). The incidence and severity of nausea or vomiting in patients receiving chemotherapy vary, depending on the type of chemotherapy given, dose, schedule, combinations of medications, and individual characteristics. The consequences of not controlling the nausea and vomiting induced by cancer treatment may lead to medical complications, a failure of the patient to comply with the cancer therapy and follow-up, and a diminished quality of life.
The supportive care of patients receiving agents with the potential to cause nausea and vomiting remains an important aspect of effective management of the oncology patient. In every treatment situation, the primary goal is prevention of CINV. However, despite an improved understanding of the pathophysiology associated with this phenomenon, the identification of predictive factors, the definition of emetic syndromes, and the development of evidence-based guidelines that incorporate the most effective antiemetic agents and regimens available to prevent and treat chemotherapy-induced emesis, there continue to be patients for whom achieving complete control (no vomiting or nausea) is problematic. To this end, patients’ quality of life may be significantly compromised in the event of incomplete control.
Pathophysiology of Nausea and Vomiting
The precise mechanisms by which chemotherapy induces nausea and vomiting are unknown; however, it appears probable that different chemotherapeutic agents act at different sites and that some chemotherapeutic agents act at multiple sites. The fact that different chemotherapeutic agents cause nausea and vomiting by different mechanisms, and that one chemotherapeutic agent may induce nausea and vomiting by more than one mechanism, helps clinicians to understand why there is no one antiemetic regimen that is effective all of the time.
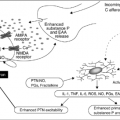
Mechanisms by which chemotherapeutic agents cause nausea and vomiting are activation of the chemoreceptor trigger zone (CTZ) either directly or indirectly, peripheral stimulation of the gastrointestinal (GI) tract, vestibular mechanisms, cortical mechanisms, or alterations of taste and smell. For most chemotherapeutic agents, the most common mechanism is thought to be activation of the CTZ.
The CTZ is located in the area postrema of the brain and can be reached by emetogenic chemicals through the cerebrospinal fluid or the blood. The thought is that the mechanisms of interaction between the CTZ and chemotherapy involve the release of various neurotransmitters that activate the vomiting center. Either one or a combination of these transmitters may induce vomiting. Some of the neurotransmitters located in the area postrema of the brain that may be excited and lead to emesis include dopamine, serotonin, histamine, norepinephrine, apomorphine, neurotensin, angiotensin II, vasoactive intestinal polypeptide, gastrin, vasopressin, thyrotropin-releasing hormone, leucine-enkephalin, and substance P (4). Other enzymes surround the CTZ, such as adenosine triphosphatase, monoamine oxidase, cholinesterase, and catecholamines; however, their role in chemotherapy-induced emesis is unknown.
Until the 1990s, the neurotransmitter that appeared to be the most responsible for CINV was dopamine. Many effective antiemetics are dopamine antagonists that may bind specifically to the D2 receptor. However, there is a high degree of variation in dopamine receptor–binding affinity by these drugs. The action of some drugs that cause nausea and vomiting is affected very little or not at all by dopamine antagonists. It is known that not all the important receptors in the CTZ are dopaminergic, as the effect of dopamine antagonists is not equal to surgical ablation of the CTZ. It has also been noted that the degree of antiemetic activity of high-dose metoclopramide cannot be explained on the basis of dopamine blockade alone.
Histamine receptors are found in abundance in the CTZ; however, H2 antagonists do not work as antiemetics at all. H1 antagonists alleviate nausea and vomiting induced by vestibular disorder and motion sickness but not nausea and vomiting induced by chemotherapy (5).
Knowledge that opiate receptors are found in abundance in the CTZ, as well as the facts that narcotics have mixed emetic and antiemetic effects that are blocked by naloxone and that naloxone has emetic properties, have led to the proposal of opiates or enkephalins as an antiemetic. High doses of naloxone augments emesis induced by chemotherapy, and low doses of narcotics may reduce emesis. Studies to date have shown that opiates can prevent chemotherapy-induced emesis in laboratory animals; however, both butorphanol and buprenorphine have not proved to be effective antiemetics in patients who had received chemotherapy previously. One study by Lissoni et al. (6) did demonstrate the synthetic enkephalin analog Fk-33-824 was more effective as an antiemetic in patients who received cisplatin; however, it was ineffective for patients receiving cyclophosphamide or epirubicin.
Edwards et al. found that arginine vasopressin levels rise to a greater extent in patients who vomit when they receive chemotherapy as compared with those who do not vomit (7). It has been suggested that perhaps arginine vasopressin plays a role in nausea more than in the vomiting induced by chemotherapy. Dexamethasone, which is a known effective antiemetic, may work by reducing arginine vasopressin levels. Another mechanism of action of corticosteroids as antiemetics may be related to modulation of prostaglandin release.
Some evidence suggests that although no one neurotransmitter is responsible for all CINV, it appears that 5-hydroxytryptamine (5-HT) (serotonin) receptors are particularly important in the pathophysiology of acute vomiting, whereas others may be more important in the pathophysiology of nausea and delayed emesis. The role of the 5-HT type 3 (5-HT3) receptor in chemotherapy-induced emesis was recognized by examining the mechanism of action of high-dose metoclopramide in decreasing cisplatin-induced emesis. High-dose metoclopramide, unlike other D2-receptor antagonists, has an exceptionally good capacity to decrease the emesis induced by cisplatin administration. It has been recognized that metoclopramide has pharmacologic effects other than dopamine antagonism. Metoclopramide is a weak antagonist of peripheral 5-HT3 receptors and can stimulate GI motility by increasing acetylcholine release from the cholinergic nerves of the GI tract. To test whether a 5-HT3-receptor blockade would decrease cisplatin-induced emesis, Miner et al. (8) took a substituted benzamide, BRL 24924, which has stimulatory effects on the GI tract and is a 5-HT3-receptor blocker, and demonstrated decreased emesis in ferrets that received cisplatin. This study was repeated with a nonbenzamide selective 5-HT3-receptor blocker MDL 72222, which has no GI-stimulating activity. The study revealed that cisplatin-induced emesis was totally blocked by this compound (8). The same conclusion was reached in another study using a different nonbenzamide, the selective 5-HT3 antagonist ICS 205-930 (9). These studies demonstrated the role of 5-HT3-receptor blockade in chemotherapy-induced emesis.
The precise mechanism of action of the 5-HT3-receptor antagonists is unknown; however, the primary effect appears to be peripheral at the site of the 5-HT3 receptors on the vagal afferent neurons. The GI tract contains approximately 80% of the body’s supply of serotonin, and it has been suggested that perhaps chemotherapy administration causes release of serotonin from the enterochromaffin cells of the GI tract, which then stimulates emesis through both the vagus and greater splanchnic nerve, and stimulates the area postrema of the brain. After cisplatin administration, there is an increase in urinary excretion of 5-hydroxyindoleacetic acid, the main metabolite of serotonin, and this increase parallels the number of episodes of emesis (10). Studies have shown that the 5-HT3-receptor antagonists decrease emesis from several chemotherapeutic agents, including cisplatin, cyclophosphamide, and doxorubicin (11, 12).
An important mechanism whereby chemotherapy may induce emesis is the peripheral effect that is thought to arise from the pharynx and the upper GI tract. It is most likely that chemotherapy does not directly stimulate the peripheral receptors. Rather, neurotransmitters probably are released as a result of local GI irritation or damage. GI tract serotonin, dopamine, opiate, histamine, and cholinergic receptors are most likely involved in the emesis induced by chemotherapy. The peripheral effects may be abolished by vagotomy, indicating that impulses from the GI tract may reach the vomiting center through the vagus and sympathetic nerves.
In addition to serotonin, substance P has recently been identified as an important neurotransmitter involved in CINV. Positron emission tomography imaging of healthy human brains has demonstrated that substance P/neurokinin-1 (NK1) receptors are located centrally in the brain stem (13). Substance P is believed to exert its effect on the emetic reflex primarily through the central mechanism of binding to the NK1 receptors in the midbrain. NK1 receptor antagonists that cross the blood–brain barrier have been shown to inhibit both acute and delayed emesis by cisplatin in animal models and human studies. Hesketh et al. analyzed data from clinical trials for the time course of cisplatin-induced emesis and demonstrated that serotonin-dependent mechanisms appeared to predominate in the first 8–12 hours post-cisplatin, but thereafter, NK1-dependent mechanisms for emesis appeared to have relatively greater importance (14). Specifically, early acute events responsive to 5-HT3-receptor antagonists are likely to be mediated by peripheral serotonin release, whereas later acute and delayed events responsive to NK1 receptor antagonists are more likely to be medicated by substance P acting centrally at the NK1 receptors (14).
Another mechanism that may be involved in chemotherapy-induced emesis could be the therapy’s effect on the vestibular system. It is known that patients who have a history of motion sickness experience a greater severity, frequency, and duration of nausea and vomiting from chemotherapy than patients who do not experience motion sickness. The mechanism by which the vestibular system may lead to chemotherapy-induced emesis is unknown; however, it is postulated that sensory information that is received by the vestibular system is different from information that was expected.
Some investigators believe that taste changes induced by chemotherapy may lead to nausea and vomiting. There are two suggested mechanisms for this. First, taste is thought to inhibit some activities incompatible with eating (e.g., oral pain, gag, nausea, vomiting). Damage to taste such as that produced by some chemotherapy might release that inhibition leading to enhancement of gag, nausea, and vomiting. This is supported by a study showing taste damage in women who have suffered from hyperemesis during pregnancy (15). Second, some chemotherapeutic agents may be tasted. For example, in a study of patients with breast carcinoma who received cyclophosphamide, methotrexate, and 5-fluorouracil, 36% reported a bitter taste in their mouth. One third of the patients thought that the bitter taste caused vomiting (16). The exact mechanism by which taste is changed by chemotherapy is unknown; however, it is thought that while the drugs are in the plasma or saliva, they have a direct effect on the oral mucosa or taste buds. Changes in taste may contribute both to nausea and vomiting as well as to anorexia.
Finally, chemotherapy-induced emesis may be induced by direct or indirect effects on the cerebral cortex. Animal studies have shown that nitrogen mustard partially causes
emesis through direct stimulation of the cerebral cortex. Studies demonstrate that the risk of nausea and vomiting is increased when a patient’s roommate is experiencing nausea and vomiting. It is also known that the amount of sleep had before receiving chemotherapy may influence whether a patient develops chemotherapy-induced emesis. In addition, large differences exist in the severity and incidence of nausea and vomiting from the same chemotherapeutic agents in different countries. These studies indicate that indirect psychological effects can mediate CINV.
emesis through direct stimulation of the cerebral cortex. Studies demonstrate that the risk of nausea and vomiting is increased when a patient’s roommate is experiencing nausea and vomiting. It is also known that the amount of sleep had before receiving chemotherapy may influence whether a patient develops chemotherapy-induced emesis. In addition, large differences exist in the severity and incidence of nausea and vomiting from the same chemotherapeutic agents in different countries. These studies indicate that indirect psychological effects can mediate CINV.
Apart from there being more than one mechanism by which each chemotherapeutic agent may induce emesis, chemotherapy induces emesis in a manner different from that of other classic emetic agents. Drugs such as apomorphine, levodopa, digitalis, pilocarpine, nicotine, and morphine cause vomiting almost immediately. Nitrogen mustard may also lead to emesis immediately; however, most chemotherapeutic agents and radiotherapy require a latency period before emesis begins. Also, most chemotherapeutic agents do not induce emesis in a monophasic way, as do the classic emetic agents. Chemotherapeutic agents induce emesis with a delayed onset, and the emesis has multiphasic time courses. When managing chemotherapy-induced emesis, one should realize that there is most likely more than one mechanism involved, suggesting that there is not one antiemetic regimen that works for all patients all of the time.
By 1991, >50% of patients received a serotonin antagonist antiemetic (5-HT3) for symptom control; approximately 90% received similar treatment in 1995, with a statistically significant reduction in posttreatment vomiting. Both physicians and nurses acknowledge an improvement in patients’ quality of life and treatment compliance with the use of these agents. However, trends over time have not shown an improvement in the control of nausea. In fact, data confirm that there is a significant increase in the duration of posttreatment nausea and no change in the frequency of posttreatment nausea or anticipatory symptoms (17) (Table 13.1).
Table 13.1 Mechanisms of Nausea and Vomiting After Chemotherapy | ||
|---|---|---|
|
Pharmacogenomics of Chemotherapy-Induced Nausea and Vomiting
The use of genetic profiling to more accurately define the functions and limitations of pharmaceuticals is a relatively recent development of modern biomedicine. The pharmacogenomic approach has proved fruitful in the clinical application of drugs targeted toward CINV (18). Several studies have used a standard approach of sequencing candidate genes associated with known emetic pathways to discover possible associations with the differentiated response among patients to treatment of CINV. Polymorphisms in the serotonin receptor and the cytochrome P (CYP)-450 system are each known to be associated with patient response to therapy.
One group sequenced the gene encoding the 3B subunit of the serotonin (5-HT3) receptor (19). They discovered 13 polymorphisms, of which one, a deletion of AAG, was associated with response to ondansetron and tropisetron. There was also a clear gene dosage effect: homozygosity for the trinucleotide deletion greatly increased postchemotherapeutic emesis over heterozygosity for the same mutation, both in the initial 4 hours after chemotherapy and the following 5–24 hours.
The same group studied gene dosage effects in the CYP-450 enzyme family, particularly the CYP-2D6 enzyme (20). The CYP-450 enzymes metabolize serotonin receptor antagonists, thereby reducing their biological half-life and their value as antiemetic therapeutics. Several alleles of the CYP-2D6 gene decrease the effectiveness of the enzyme versus the wild-type allele. Individuals with three copies of the wild-type allele, which increases serotonin antagonist metabolism, had the greatest number of acute postchemotherapeutic emetic episodes under treatment with ondansetron or tropisetron, whereas individuals with three copies of the inactive variant alleles had fewer emetic episodes. Heterozygosity at the locus produced intermediate effects.
Further studies will focus on other candidate genes in emetic pathways, including the NK1 (substance P) receptor, dopaminergic receptors, and tyrosine hydroxylase. At this time, routine genetic testing of patients receiving emetogenic cancer chemotherapy is not indicated; however, it may prove useful in patients demonstrating unusually impaired response to standard antiemetic therapy. Future testing will more definitively establish the relationship between genotype and antiemetic phenotype and may bring genetic profiling into the realm of standard clinical practice.
Emetic Syndromes
Patients undergoing therapy for the treatment and possible cure of cancer with chemotherapy are often faced with the distressing side effects of nausea and vomiting. The goals of antiemetic therapy are as follows:
To achieve complete control in all settings
To provide maximum convenience for patients and staff
To eliminate potential side effects of the agents
To minimize the cost of treatment with antiemetic agents and drug administration
As a result of antiemetic investigations, three major, related emetic syndromes have been identified: acute, delayed, and anticipatory emesis. Traditionally, acute emesis is defined as occurring within the first 24 hours of administration of chemotherapy (usually within 1–2 hours) and is generally most severe during the initial 4–6 hours. Delayed emesis has been arbitrarily defined as occurring 24 or more hours after chemotherapy (range of 16–24 hours), with maximal risk at 48 hours. It is most commonly associated with the administration of cisplatin, carboplatin, cyclophosphamide, and doxorubicin. A study that outlines the natural history of delayed emesis concluded that although the emesis associated with this dilemma is less severe than that which is seen in the acute phase, it still poses significant problems with nutrition, hydration, and possibly a prolonged hospital course (21).
Table 13.2 Factors Associated With an Increased Incidence of Anticipatory Nausea and Vomiting | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Initial studies revealed that delayed emesis could be controlled with a regimen of metoclopramide and dexamethasone. Because of the possibility of extrapyramidal side effects such as anxiety, akathisia, restlessness, torticollis, or oculogyric crisis, with metoclopramide, patients should be given a prescription for diphenhydramine to be taken at the first sign of an extrapyramidal symptom. In the younger patient, diphenhydramine should be given prophylactically.
Early trials addressing the treatment of delayed emesis with the single-agent serotonin antagonist ondansetron were discouraging and labeled the serotonin antagonists as having low activity. Two randomized studies, one with ondansetron and one with granisetron, indicated efficacy of the serotonin antagonists for delayed emesis in patients receiving chemotherapy of intermediate emetogenicity (22, 23). New antiemetic agents that are beneficial in both the prevention and treatment of delayed emesis have been recently identified and approved for use. These agents are discussed later in the section New Agents.
Preventive therapy is imperative for patients to achieve the best outcome. The risk defined for acute emesis is a good predictor of delayed emesis. Patients who do not receive preventive therapy have a 70–90% incidence of delayed emesis with high-risk agents and a 30–60% risk with moderate-risk agents.
Anticipatory emesis (Table 13.2) is a learned or conditioned response that typically occurs before, during, or after the administration of chemotherapy. In this instance, patients may be responding to a variety of stimuli that in most instances were associated with a prior experience when there was inadequate control of emesis. The corresponding psychological mechanism for anticipatory emesis is unknown and is secondary to the direct administration of the chemotherapy agent itself. Therefore, patients must be given the opportunity to receive the optimal antiemetic regimen with their initial course of chemotherapy to prevent acute and delayed emesis, and, consequently, anticipatory emesis. Treatment for the occurrence of anticipatory emesis may include the use of benzodiazepines in addition to antiemetics before and during chemotherapy. Relaxation techniques, guided visual imagery, desensitization, and hypnosis techniques may also be effective (24, 25, 26, 27).
In addition to hypnosis, relaxation, imagery, and desensitization, acupuncture is a nonmedicinal complementary therapy that has been shown to have benefits in chemotherapy-related nausea and vomiting. An initial trial was done with 130 patients who had a history of distressing emesis in prior chemotherapeutic regimens. Emesis was reduced in 97% of the subjects (28). A National Institutes of Health consensus trial concluded that acupuncture was effective in reducing chemotherapy-induced emesis; however, placebo effect was a concern (29). A subsequent trial that addressed the issue of placebo effect was done with women with breast cancer receiving high-dose cyclophosphamide, cisplatin, and carmustine. One hundred four women were randomly assigned to receive no needling, minimal needling at control points with mock electrostimulation, or classic antiemetic electroacupuncture once daily for 5 days. The number of emesis episodes were lower in the first 5 days for those receiving electroacupuncture compared with those receiving minimal needling at control points or no needling (p <.001). The effect appeared to be of limited duration in that there were no significant differences during the 9-day follow-up (30). Clearly the data are promising; however, additional research is needed in this area.
Control of Emesis and Risk Factors
The methodology used in antiemetic trials has identified useful patient characteristics and prognostic factors that may affect antiemetic control. These indicators become important for tailoring antiemetic regimens as well as designing antiemetic trials. Careful studies have identified patient-related risk factors to include prior experience with chemotherapy, alcohol intake history, age, and gender as influencing patient outcomes.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree