Agent
Mechanism of action
Toxicities
Clinical pearls
Antimicrotuble agent; stabilizes microtubules inhibiting interphase and mitosis
•Myelosuppression
•Nausea/vomiting (low risk)
•Peripheral neuropathy
•Arthralgia/myalgia
•Hypersensitivity
•Alopecia
•Vascular irritant
•Should be administered prior to platinum derivatives (to avoid toxicity due to decreased paclitaxel clearance)
•Premedicate with corticosteroid, H2 and H1 antagonist
•Infuse through 0.22 μm in-line filter and nonsorbing administration set
•Myelosuppression increased with higher doses, more frequent doses, and longer infusion times
•Peripheral neuropathy increased with more frequent dosing and shorter infusion times
•Dose adjust for hepatic toxicity
•Drug interactions
1.Anthracyclines
2.CYP2C8, CYP3A4 inducers and inhibitors
3.P-glycoprotein inducers/inhibitors
Antimicrotubule agent, stabilizes microtubules inhibiting interphase and mitosis
•Myelosuppression
•Nausea/vomiting (low)
•Peripheral neuropathy
•Arthralgia/myalgia
•Oncholysis, Alopecia
•Hypersensitivity reactions
•Fluid retention
•Vascular irritant
•Should be administered prior to platinum derivatives (to avoid toxicity due to decreased clearance)
•Premedicate with corticosteroid for 3 days starting the day prior to treatment to help with edema
•Infuse with nonsorbing polyethylene lined (non-DHEP) tubing
•Dose adjust for hepatic toxicity
•Drug interactions
1.Anthracyclines
2.CYP3A4 inducers and inhibitors
3.P-glycoprotein inducers/inhibitors
Alkylating agent, forms DNA intrastrand and interstrand cross-links inhibiting DNA function and synthesis
•Nausea/vomiting (high risk)
•Nephrotoxicity
•Electrolyte depletion (potassium, magnesium)
•Ototoxicity
•Peripheral neuropathy
•Myelosuppression
•Vesicant at higher concentrations (> 0.4 mg/mL)
•Hypersensitivity may develop with prolonged (> 6 cycles) or prior exposure
•Administer after taxanes
•Adequate hydration prior to (1–2 L) and post infusion is recommended to prevent nephrotoxicity
•Do not administer with aluminum needles or IVs
•Dose adjust for renal impairment
•Drug interactions
1.Aminoglycosides
2.Renal eliminated drugs
3.Loop diuretics
Alkylating agent, forms DNA interstrand cross-links inhibiting DNA function and synthesis
•Nausea/vomiting (moderate risk)
•Peripheral neuropathy
•Myelosuppression
•Electrolyte depletion (potassium, magnesium)
•Nephrotoxicity
•Hypersensitivity may develop with prolonged (>6 cycles) or prior exposure
•Administer after taxanes
•Do not administer with aluminum needles or IVs
•If dose calculated using estimated GFR consider capping estimated GFR at 125 mL/min to avoid toxicity
•Doses calculated by target AUC via the Calvert Formula [Total dose = Target AUC × (GFR + 25)]
•Drug interactions
1.Aminoglycosides
2.Renally eliminated drugs
Alkylating agent, forms DNA intrastrand and interstrand cross-links inhibiting DNA function and synthesis
•Nausea/vomiting (moderate risk)
•Myelosuppression
•Peripheral neuropathy (acute and chronic)
•Fatigue
•Vesicant
•Hypersensitivity may develop with prolonged (>6 cycles) or prior exposure
•Must be administered via central vein
•Do not administer with aluminum needles or IVs
•Counsel patients to avoid cold to prevent worsening of acute neuropathies/paresthesia
Topoisomerase I inhibitor, reversibly binds to topoisomerase I stabilizing the cleavable complex resulting in double-strand DNA breaks and cell death. S-phase specific
•Fatigue
•Nausea/vomiting (moderate risk)
•Myelosuppression
•Diarrhea (early and late)
•Cholinergic toxicity
•Alopecia
•Hyperbilirubinemia
•Cholinergic symptoms and early diarrhea may be treated/prevented with atropine 0.25–1 mg
•Treat late diarrhea with loperamide 4 mg PO at onset followed by 2 mg every 2 h until no bowel movement for 12 h
•Patients with homozygous UGT1A1*28 allele are at increased risk for toxicity
•Dose adjust for hepatic impairment
•Drug interactions
1.Azole antifungals
2.Carbamazepine
3.CYP2B6, CYP3A4 inducers/inhibitors
4.Conivaptan
5.Fosphenytoin/phenytoin
6.Grapefruit juice
7.P-glycoprotein inducers/inhibitors
8.St. John’s Wort
Topoisomerase I inhibitor, binds to topoisomerase I and stabilizing the cleavable complex resulting in single-strand DNA breaks. S-phase cell cycle specific
•Myelosuppression
•Alopecia
•Fatigue
•Nausea/vomiting (low risk)
•Constipation
•Diarrhea
•Stomatitis
•Vascular irritant
•Dose adjust for renal impairment
•Drug interactions
1.Clozapine
2.Fosphenytoin/phenytoin
3.P-glycoprotein inhibitors
Topoisomerase II inhibitor; intercalates between DNA base pairs causing disruption of topoisomerase II. Chelates with iron forming complex which can produce free radicals that cause damage to DNA and cell membranes. Liposomal formulation is pegylated which increases blood circulation time
•Palmar-plantar erythrodysesthesia (Hand-foot syndrome)
•Nausea/vomiting (Low risk)
•Stomatitis/mucositis
•Diarrhea
•Myelosuppression
•Discolored urine/body fluids
•Infusion reaction
•Vascular irritant
•Do not infuse with in-line filter
•Associated with less cardiotoxicity than doxorubicin but cumulative lifetime dose should be considered
•Monitor cardiac function (LVEF) at baseline and periodically during treatment
•May cause radiation recall
•Dose adjust for hepatic impairment
•Drug interactions
1.Clozapine
2.CYP2B6 substrates
3.CYP2D6, CYP3A4 inhibitors/inducers
4.Taxanes
Topoisomerase II inhibitor; intercalates between DNA base pairs causing disruption of topoisomerase II. Chelates with iron forming complex which can produce free radicals that cause damage to DNA and cell membranes
•Cardiotoxicity (acute and delayed)
•Myelosuppression
•Nausea/vomiting (moderate risk)
•Diarrhea
•Mucositis
•Alopecia
•Discolored urine/body fluids
•Photosensitivity
•Radiation recall
•Infertility
•Secondary malignancy
•Vesicant
•Increased risk of cardiotoxicity with lifetime cumulative dose >500 mg/m2
•Baseline and periodic LVEF monitoring is recommended
•Prolonging infusion time decreases risk of cardiotoxicity
•Dexrazoxane may be used at a 10:1 ratio to decrease risk of cardiotoxicity
•If administering continuous infusion use of a central venous line is recommended
•Dose adjust for hepatic impairment.
•Drug interactions
1.Clozapine
2.CYP2B6 substrates
3.CYP2D6, CYP3A4 inhibitors/inducers
4.Taxanes
Topoisomerase II inhibitor; intercalates between DNA base pairs causing disruption of topoisomerase II. Chelates with iron forming complex which can produce free radicals that cause damage to DNA and cell membranes
•Nausea/vomiting (moderate risk)
•Cardiotoxicity
•Alopecia
•Myelosuppression
•Infertility
•Mucositis
•Secondary malignancy
•Vesicant
•Increased risk of cardiotoxicity with lifetime cumulative dose >900 mg/m2
•Baseline and periodic LVEF monitoring is recommended
•Dose adjust for hepatic impairment and severe renal impairment
•Drug interactions
1.Cimetidine
2.Taxanes
Pyrimidine antimetabolite inhibits DNA synthesis in S-phase. Phosphorylated intracellularly to its active metabolites, gemcitabine diphosphate and gemcitabine triphosphate
•Myelosuppression
•Nausea/vomiting (low risk)
•Rash
•Diarrhea
•Alopecia
•Transient flu-like symptoms
•Hemolytic uremic syndrome
•Increasing infusion time >60 min increases toxicity
•Drug interactions
1.Bleomycin
2.Fluorouracil
Antimicrotubule agent; inhibits the formation of microtubules preventing cell replication. M phase specific
•Myelosuppression
•Peripheral neuropathy
•Constipation
•Paralytic ileus/intestinal obstruction
•Fatigue
•Nausea/vomiting (minimal risk)
•Alopecia
•Vesicant
•Consider placing patient on bowel regimen
•Dose adjust for hepatic impairment
•Drug interactions
1.Substrates of CYP3A4
2.Azole antifungals
3.Phenytoin
Antimicrotubule agent; inhibits the formation of microtubules preventing cell replication. M phase specific
•Myelosuppression
•Hypertension
•Alopecia
•Constipation
•Peripheral neuropathy
•Nausea/vomiting (minimal risk)
•Vesicant
•Dose adjust for hepatic impairment
•Consider placing patient on bowel regimen
•Drug interactions
1.CYP3A4 and P-glycoprotein substrates
2.Induces P-glycoprotein
3.Itraconazole
4.Voriconazole
5.Erythromycin
Antimicrotubule agent; inhibits the formation of microtubules preventing cell replication. M and S phase specific
•Alopecia
•Myelosuppression
•Nausea/vomiting (minimal risk)
•Cranial nerve dysfunction
•Constipation
•Paralytic ileus/intestinal perforation
•Peripheral neuropathy
•Foot drop/gait changes
•Vesicant
•Consider placing patient on bowel regimen
•Drug Interactions
1.CYP3A4 and P-glycoprotein substrates
2.Itraconazole
3.Voriconazole
Folate antimetabolite; binds to and inhibits dihydrofolate reductase decreasing formation of reduced folates and thymidylate synthetase inhibiting DNA synthesis, repair and cell replication. S phase specific
•Myelosuppression
•Alopecia
•Nausea/vomiting (low to minimal risk)
•Mucositis
•Leucovorin given with some regimens to mitigate hematologic and gastrointestinal side effects
•Elimination reduced in patients with ascites and/or pleural effusions
•Dose adjust for renal and hepatic impairment
•Drug interactions
1.Substrates of P-glycoprotein
2.Probenecid
3.Salicylates
4.Hydantoin anticonvulsants
5.Nonsteroidal anti-inflammatory drugs (NSAIDs)
Binds to guanine DNA base inhibiting DNA, RNA, and protein synthesis
•Alopecia
•Nausea/vomiting (moderate risk)
•Increased pigmentation
•Rash
•Myelosuppression
•Vesicant
•Adjust dose for hepatic impairment
•Drug interactions
1.NSAIDs
2.Salicylates
Alkylating agent; forms cross-links between strands of DNA inhibiting DNA synthesis. Prodrug that requires activation by liver
•Nausea/vomiting (moderate risk)
•Alopecia
•Secondary malignancy
•Infertility
•Hemorrhagic cystitis (rare at doses use for gynecologic malignancy)
•Myelosuppression
•Increase fluid intake during treatment to prevent bladder toxicity
•Mesna may be given with higher doses (≥1,000 mg/m2) to prevent/treat hemorrhagic cystitis
•Oral doses should be taken in morning with plenty of fluid
•Drug interactions
1.CYP2B6 substrates
2.Nalidixic acid
Alkylating agent; not fully characterized
•Myelosuppression
•Peripheral neuropathy
•Nausea/vomiting (moderate risk)
•Available as 50 mg capsule
•Drug interactions
1.Monoamine oxidase inhibitors
2.NSAIDs
3.Pyridoxine
Pyrimidine antimetabolite; Prodrug of fluorouracil, activated by liver and tissue to active form which inhibits thymidylate synthetase inhibiting DNA and RNA synthesis. G1 and S phase specific
•Nausea/vomiting (low risk)
•Myelosuppression
•Mucositis
•Diarrhea
•Palmar plantar erythrodysesthesia (Hand-foot syndrome)
•Photosensitivity
•Take within 30 min of meal
•Swallow tablets whole
•Contraindicated in known deficiency of dihydropyrimidine dehydrogenase (DPD)
•Not recommended for CrCl <30 mL/min
•Dose adjust for renal and hepatic impairment
•Drug interactions
1.CYP2C9 inhibitor
2.Warfarin
3.Folic acid
4.Leucovorin
Pyrimidine antimetabolite; inhibits thymidylate synthetase inhibiting DNA and RNA synthesis. G1 and S phase specific
•Vascular irritant
•Palmar-plantar erythrodysesthesia
•Nausea/vomiting (Low risk)
•Photosensitivity
•Myelosuppression
•Contraindicated in known deficiency of dihydropyrimidine dehydrogenase (DPD)
•Drug interactions
1.Inhibits CYP2CP
2.Dapsone
3.Trimethoprim
4.Hydantoin anticonvulsants
5.Levamisole
Alkylating agent; cross-linking strands of DNA inhibiting protein and DNA synthesis
•Hemorrhagic cystitis
•Alopecia
•Nausea/vomiting (High to moderate risk)
•Myelosuppression
•Encephalopathy (confusion, somnolence, dizziness)
•Infertility
•Secondary malignancy
•Must be administered with Mesna (at least 60 % of ifosfamide dosage) to prevent hemorrhagic cystitis
•Adequate hydration, dose fractionation may be used to decrease hemorrhagic cystitis
•Neurotoxicity increased in patients with hypoalbuminemia, renal dysfunction, and history ifosfamide induced encephalopathy
•Neurotoxicity may be treated with methylene blue
•Dose adjust for renal impairment
•Drug interactions
1.CYP2A6, CYP2C19, and CYP3A4 substrates
2.CYP2C9 inducer
3.Conivaptan
4.St. John’s wort
5.Telithromycin
Alkylating agent; Cross-links strands of DNA inhibiting DNA and RNA synthesis
•Myelosuppression
•Nausea/vomiting (minimal risk)
•Secondary malignancy (risk as high as 11 %)
•Administer oral formulation on empty stomach
•Dose adjust for renal impairment
Folate antimetabolite, inhibits folate dependent enzymes involved in DNA and RNA function and synthesis, thus inhibiting cell function and replication
•Fatigue
•Nausea/vomiting
•Myelosuppression
•Do not give when CrCl <45 mL/min
•Must give with vitamin B12 1,000 mcg subQ every 9 weeks and folic acid 400–1,000 mcg PO daily started 1 week prior to initial dose
•Give dexamethasone 4 mg PO BID for 3 days starting 24 h prior to each dose
•Drug interactions
1.NSAIDs
Exact mechanism unknown, suggested to have alkylating effect as well as antimetabolite activity
•Nausea/vomiting (High risk)
•Diarrhea
•Flu-like syndrome
•Alopecia
•Photosensitivity
•Rash
•Vascular irritant
•Dose adjust for renal impairment
•Drug interactions
1.CYP1A2 and CYP2E1 substrates
Prodrug which is nonenzymaticaly converted to alkylating agent. Bonds to DNA leading to double strand breaks and cell death
•Nausea/vomiting (moderate risk)
•Myelosuppression
•Fatigue
•Alopecia
•Constipation
•Capsules should be taken with a full glass of water
•Food decreases absorption
•Do not crush, break or chew capsule
•Administer on empty stomach at bedtime to avoid nausea/vomiting
Tyrosine kinase inhibitor, decreases activity of vascular endothelial growth factors (VEGF), platelet-derived growth factor receptors, cytokine receptor, interleukin-2 receptor inducible T-cell kinase, leukocyte-specific protein tyrosine kinase, and transmembrane glycoprotein receptor tyrosine kinase
•Hypertension
•Fatigue, insomnia, hemiparesis
•Hair color change
•Hand-foot skin reaction
•Rash/skin depigmentation
•Diarrhea
•Nausea/vomiting (minimal risk)
•Myelosuppression
•Hepatotoxicity
•Administer 1 h before or 2 h after a meal
•Do not crush or chew tablet
•If receiving steroids monitor for Pneumocystis Jiroveci and consider prophylaxis
•Dose adjust for hepatic impairment
•Drug interactions
1.CYP3A4 and P-glycoprotein substrates
2.CYP3A4 strong inhibitors—consider decreasing dose by at least 50 %
3.CYP3A4 strong inducers – do not use
4.Avoid grapefruit juice
Topoisomerase II inhibitor, activity results in DNA strand breaks. S-phase specific
•Nausea/vomiting (low risk)
•Myelosuppression
•Alopecia
•Secondary malignancy
•Infusion reaction/hypersensitivity
•Vascular irritant
•Do not crush, open or chew capsules
•Dose adjust for renal impairment
•Drug interactions
1.CYP3A4 and P-glycoprotein substrates
Antitumor antibiotic, binds to DNA leading single and double strand breaks and decreased DNA, RNA, and protein synthesis
•Vascular irritant
•Hypersensitivity reaction
•Pulmonary dysfunction
•Hyperpigmentation
•Mucositis
•Acute febrile reaction
•Complete pulmonary function tests prior to initiation and consider monitoring every 2 cycles or as clinically indicated
•Risk factors for pulmonary toxicity include smoking, prior radiation, and concurrent oxygen administration
•Dose adjust for renal impairment
Treatment Regimens
Regimens used for the treatment of gynecologic cancers including the drugs, dosage, and frequency are detailed in Table 6.2.
Cervical cancer: locally advanced | ||
|---|---|---|
Chemotherapy regimen | Chemotherapy regimens details | References |
Cisplatin + RT | Cisplatin 40 mg/m2 IV day 1 Q1 week × 6 weeks Concurrent radiotherapy 55–75 Gy | |
Cisplatin + 5-FU + RT | Cisplatin 70 mg/m2 IV day 1 5-FU 1,000 mg/m2/day continuous IV days 1–4 Q3w × 2 cycles Concurrent radiotherapy 49.3 Gy Followed by cisplatin 70 mg/m2 IV day 1 5-FU 1,000 mg/m2/day continuous IV days 1–4 Q3w × 2 more cycles | [35] |
Cervical cancer: recurrent or metastatic first line | ||
Cisplatin + paclitaxel | Cisplatin 50 mg/m2 IV day 1 Paclitaxel 135 mg/m2 IV day 1 Q3w | |
Carboplatin + paclitaxel | Carboplatin AUC = 6 IV day 1 Paclitaxel 175 mg/m2 IV day 1 Q3w | [38] |
Cisplatin + topotecan | Cisplatin 50 mg/m2 IV day 1 Topotecan 0.75 mg/m2/day IV days 1–3 Q3w × 6 cycles | |
Cisplatin +paclitaxel +bevacizumab | Cisplatin 50 mg/m2 IV day 1 Paclitaxel 135–175 mg/m2 IV day1 Bevacizumab 15 mg/kg IV day 1 Q3w | [40] |
Cisplatin + gemcitabine | Cisplatin 50 mg/m2 IV day 1 Gemcitabine 1,000 mg/m2/day IV days 1, 8 Q3w | [37] |
Cisplatin | Cisplatin 50 mg/m2 IV day 1 Q3w | [39] |
Carboplatin | Carboplatin AUC = 5–7.5 IV day 1 Q3w | [41] |
Paclitaxel | Paclitaxel 135–175 mg/m2 IV day 1 Q3w | [42] |
Cervical cancer: recurrent or metastatic second line | ||
Bevacizumab | Bevacizumab 15 mg/kg IV day 1 Q3w | [43] |
Docetaxel | Docetaxel 75 mg/m2 IV day 1 Q3w | [44] |
5-FU | 5-FU 370 mg/m2/day IV push days 1–5 Leucovorin 200 mg/m2 IV push days 1–5 Q4w OR 5-FU 1,000 mg/m2/day continuous IV infusion days 1–5 Q4w | [45] |
Gemcitabine | Gemcitabine 800 mg/m2 IV days 1, 8, 15 Q4w | [46] |
Ifosfamide | 1.5 g/m2/day IV days 1-5 Mesna 300 mg/m2 IV 0 h, 4 h, 8 h, d1–5 Q4w | |
Irinotecan | Irinotecan 125 mg/m2 IV days 1, 8, 15 & 22 Q6w | [49] |
Mitomycin | Mitomycin 10–15 mg/m2 IV push day 1 Q4-6w | [50] |
Topotecan | Topotecan 1.5 mg/m2/day IV days 1–5 Q3–4w or Topotecan 3–4 mg/m2/d IV days 1, 8, 15 Q4w | |
Endometrial cancer: hormonal therapy for recurrent, metastatic, or high-risk endometrial cancer | ||
Hormonal regimen | Hormonal regimen details | References |
Medroxyprogesterone | Medroxyprogesterone 200–1,000 mg po daily days 1–14 Q28d | [53] |
Tamoxifen | Tamoxifen 20 mg po bid | [54] |
Megestrol acetate | Megestrol 80 mg po bid OR Megestrol 800 mg po daily in divided doses | |
Megestrol/tamoxifen | Megestrol 80 mg po bid × 3 weeks alternative with tamoxifen 20 mg po bid | [57] |
Letrozole | Letrozole 2.5 mg po daily | [58] |
Anastrozole | Anastrozole 1 mg po daily | [59] |
Endometrial cancer: adjuvant for recurrent, metastatic, or high-risk endometrial cancer | ||
Cisplatin + doxorubicin | Doxorubicin 60 mg/m2 IV day 1 Cisplatin 50 mg/m2 IV day 1 Q3w | |
Cisplatin +doxorubicin +paclitaxel | Doxorubicin 45 mg/m2 IV day 1 Cisplatin 50 mg/m2 IV day 1 Paclitaxel 160 mg/m2 IV day 2 Q3w | [62] |
Carboplatin +paclitaxel | Paclitaxel 175 mg/m2 IV day 1 Carboplatin AUC 5-7 IV day 1 Q4w | |
Weekly paclitaxel +carboplatin | Paclitaxel 80 mg/m2 IV days 1, 8, 15 Carboplatin AUC = 2 IV days 1, 8, 15 Q3w | [66] |
Carboplatin +docetaxel | Carboplatin ACU = 6 IV day 1 Docetaxel 60 mg/m2 IV day 1 Q3w Carboplatin AUC = 6 IV day 1 Docetaxel 75 mg IV day 1 followed by radiation Q3w | |
Cisplatin | Cisplatin 50–60 mg/m2 IV day 1 Q3w | |
Carboplatin | Carboplatin 360–400 mg/m2 IV Q3-4w | |
Doxorubicin | Doxorubicin 45–60 mg/m2 IV day 1 Q3w | [73] |
Liposomal doxorubicin | Liposomal doxorubicin 40–50 mg/m2 IV day 1 Q3w | [74] |
Paclitaxel | Paclitaxel 175–250 mg/m2 IV day 1 Q3w | |
Weekly docetaxel | Docetaxel 36 mg/m2 IV days 1, 8, 15 Q3w | [77] |
Bevacizumab | Bevacizumab 15 mg/kg IV day 1 Q3w | [78] |
Cisplatin + ifosfamide (for carcinosarcoma) | Cisplatin 20 mg/m2/day IV days 1–4 Ifosfamide 1.5 g/m2/day IV days 1–4 Mesna 120 mg/m2 IV loading dose followed by 1.5 g/m2/day IV days 1–4 Q3w | |
Ifosfamide + paclitaxel (for carcinosarcoma) | Ifosfamide 1.6 g/m2/day (1.2 g/m2/day if patients received prior radiation) IV days 1–3 Paclitaxel 135 mg/m2 IV over 3 h day 1 Mesna 2 g IV over 12 h days 1–3 Q3w × 8 cycles | [81] |
Ifosfamide (for carcinosarcoma) | Ifosfamide 2.0 g/m2/day (1.2 g/m2/day if patients received prior radiation) IV days 1–3 Mesna 2 g IV over 12 h days 1–3 Q3w × 8 cycles | [81] |
Uterine sarcoma: hormonal therapy for endometrial stromal sarcoma | ||
Hormonal regimen | Hormonal regimen details | References |
Medroxyprogesterone | Medroxyprogesterone 500–1,000 mg po daily | [82] |
Megestrol acetate | Megestrol 160 mg po daily | |
Letrozole | Letrozole 2.5 mg po daily | [85] |
Anastrozole | Anastrozole 1 mg po daily | [85] |
GnRH analog | Optimal dose unknown | |
Uterine sarcoma: chemotherapy | ||
Gemcitabine + docetaxel | Gemcitabine 900 mg/m2 IV over 90 min days 1, 8 Docetaxel 75–100 mg/m2 IV day 8 Q3w Dose reduced by 25 % for patients with prior history of pelvic radiation | |
Doxorubicin + ifosfamide | Doxorubicin 50–75 mg/m2 IV day 1 Ifosfamide 5 g/m2 IV over 24 h day 1 Mesna 6 g/m2 IV over 36 h day 1 Q3w Dose reduced by 25 % for patients with prior history of pelvic radiation | |
Doxorubicin + dacarbazine | Doxorubicin 60 mg/m2 IV day 1 Dacarbazine 250 mg/m2 IV days 1–5 Q3w Dose reduced by 25 % for patients with prior history of pelvic radiation | [92] |
Gemcitabine + dacarbazine | Gemcitabine 1,800 mg/m2 IV over 180 min day 1 Dacarbazine 500 mg/m2 IV Q2w | |
Gemcitabine + vinorelbine | Vinorelbine 25 mg/m2 IV day 1 Gemcitabine 800 mg/m2 IV over 90 min days 1, 8 Q3w | [95] |
Doxorubicin | Doxorubicin 60 mg/m2 IV day 1 Q3w | [92] |
Epirubicin | Epirubicin 75 mg/m2 IV day 1 Q3w | [96] |
Gemcitabine | Gemcitabine 1,000 mg/m2 IV days 1, 8, 15 Q4w | [97] |
Ifosfamide | Ifosfamide 1.5 g/m2/day IV days 1–5 Q3w | [98] |
Liposomal doxorubicin | Liposomal doxorubicin 50 mg/m2 IV day 1 Q4w | [99] |
Paclitaxel | Paclitaxel 175 mg/m2 IV day 1 Q3w | |
Temozolomide | Temozolomide 50–75 mg/m2 po daily week 1–6 Q8w | [102] |
Dacarbazine | Dacarbazine 1,200 mg/m2 IV day 1 Q3w | [94] |
Vinorelbine | Vinorelbine 30 mg/m2 IV day 1 Q2w | [103] |
Pazopanib | Pazopanib 800 mg po daily | [104] |
Ovarian, fallopian tube, or primary peritoneal carcinoma: primary chemotherapy for Stage II–IV | ||
Paclitaxel +carboplatin | Paclitaxel 175 mg/m2 IV over 3 h day 1 Carboplatin AUC = 5–7.5 IV day 1 Q3w × 6 cycles | [105] |
Docetaxel +carboplatin | Docetaxel 60–75 mg/m2 IV day 1 Carboplatin AUC = 5–6 IV day1 Q3w × 6 cycles | [106] |
Paclitaxel +cisplatin | Paclitaxel 135 mg/m2 IV over 24 h day 1 Cisplatin 75–100 mg/m2 IV day 2 Q3w × 6 cycles | [107] |
Paclitaxel IV +cisplatin IP +paclitaxel IP | Paclitaxel 135 mg IV over 3 h or 24 h day 1 Cisplatin 75–100 mg/m2 IP day 2 Paclitaxel 60 mg/m2 IP day 8 Q3w × 6 cycles | [108] |
Paclitaxel D1,8, 15 +carboplatin | Paclitaxel 80 mg/m2 IV over 1 h days 1, 8, 15 Carboplatin AUC = 6 IV day 1 Q3w × 6 cycles | [109] |
Paclitaxel +carboplatin +bevacizumab | Paclitaxel 175 mg/m2 IV day 1 Carboplatin AUC = 5–7.5 IV day 1 Q3w × 6 cycles + Bevacizumab 15 mg/kg IV day 1 (C2–22) | [110] |
Paclitaxel +carboplatin +bevacizumab | Paclitaxel 175 mg/m2 IV day 1 Carboplatin AUC = 5–7.5 IV day 1 Q3w × 6 cycles + Bevacizumab 15 mg/kg IV day 1 (C2-18) | [111] |
Ovarian, fallopian tube, or primary peritoneal carcinoma: platinum-sensitive, first-relapse | ||
Paclitaxel + carboplatin | Paclitaxel 175 mg/m2 IV day 1 Carboplatin AUC = 5–6 IV day 1 Q3w | |
Gemcitabine + carboplatin | Gemcitabine 1,000 mg/m2 IV days 1, 8 Carboplatin AUC = 4 IV day 1 Q3w | [114] |
Gemcitabine + carboplatin + bevacizumab | Gemcitabine 1,000 mg/m2 IV days 1, 8 Carboplatin AUC = 4 IV day 1 Bevacizumab 15 mg/kg IV day 1 Q3w | [115] |
Gemcitabine + cisplatin | Gemcitabine 600–750 mg/m2 IV days 1, 8 Cisplatin 30 mg/m2 IV days 1, 8 Q3w | |
Docetaxel + carboplatin | Docetaxel 75 mg/m2 IV day 1 Carboplatin AUC = 5 IV day 1 Q3w | [118] |
Paclitaxel D1,8, 15 + carboplatin | Paclitaxel 80 mg/m2 IV over 1 h days 1, 8, 15 Carboplatin AUC = 6 IV day 1 Q3w | [109] |
Docetaxel D1,8,15 + carboplatin | Docetaxel 35 mg/m2 IV days 1, 8, 15 Carboplatin AUC = 2 IV days 1, 8, 15 Q4w | [119] |
Liposomal doxorubicin + carboplatin | Liposomal doxorubicin 30 mg/m2 IV day 1 Carboplatin AUC = 5 IV day 1 Q4w | [120] |
Carboplatin | Carboplatin AUC = 5–6 IV day 1 Q3w | |
Bevacizumab | Bevacizumab 15 mg/kg IV day 1 Q3w | [122] |
Ovarian, fallopian tube, or primary peritoneal carcinoma: platinum-resistant or subsequent recurrence | ||
Docetaxel | Docetaxel 75–100 mg/m2 IV day 1 Q3w Or Docetaxel 30 mg/m2 IV days 1, 8, 15 Q4w | |
Weekly paclitaxel | Paclitaxel 80 mg/m2 IV days 1, 8, 15 Q4w | [125] |
Gemcitabine | Gemcitabine 600–1,000 mg/m2 IV days 1,8, 15 Q4w | |
Etoposide (oral) | Etoposide 50 mg/m2 po days 1–21 Q4w | [129] |
Altretamine | Altretamine 260 mg/m2 po days 1–14 Q4w | [130] |
Bevacizumab | Bevacizumab 15 mg/kg IV day 1 Q3w | |
Nab-paclitaxel | Nab-paclitaxel 260 mg/m2 IV day 1 Q3w | [133] |
Oxaliplatin | Oxaliplatin 130 mg/m2 IV day 1 Q3w | [134] |
Liposomal doxorubicin | Pegylated liposomal doxorubicin 40 mg/m2 IV day 1 Q4w | |
Topotecan | Topotecan 1.25 mg/m2 IV days 1–5 Q3–4w Or Topotecan 3–4 mg/m2 IV days 1, 8, 15 W3-4w | |
Vinorelbine | Vinorelbine 30 mg/m2 IV days 1, 8 Q3w | [141] |
Pemetrexed | Pemetrexed 900 mg/m2 IV day 1 Q3w | [142] |
Ifosfamide | Ifosfamide 1,000–1,200 mg/m2/day IV days 1–5 Mesna 200 mg/m2/day IV 0 h, 4 h, 8 h, days 1–5 Q4w | [143] |
Capecitabine | Capecitabine 2,000 mg/m2/day po in two divided doses, days 1–14 Q3w | [144] |
Irinotecan | Irinotecan 250–300 mg/m2 IV day 1 Q3w | [145] |
Tamoxifen | Tamoxifen 20 mg po bid | [146] |
Anastrozole | Anastrozole 1 mg po daily | [147] |
Letrozole | Letrozole 2.5 mg po daily | |
Leuprolide acetate | Leuprolide 3.75 mg im Q4w | [151] |
Megestrol acetate | Megestrol 800 mg po daily × 28 days followed by 400 mg po daily | |
Chemotherapy-Induced Nausea and Vomiting (CINV)
Background [154–157]
Nausea /Vomiting (N/V) are two of the most feared adverse effects of chemotherapy. 70–90 % of patients will experience some form of N/V during their treatment. Since the advent of 5-Hydroxytryptamine-3 (5HT3) antagonists the incidence of vomiting has been decreased to 30 %. However, nausea still remains a significant adverse effect that can have a major impact on the treatment of gynecologic cancers.
Definitions [155, 158]
Nausea—a feeling of sickness in the stomach characterized by an urge to vomit.
Vomiting—an expulsion of gastrointestinal contents through the mouth.
Acute emesis—occurs in the first 24 h after chemotherapy.
Delayed emesis—takes place 24 h or more after chemotherapy administration.
Anticipatory emesis—result of a learned response to chemotherapy.
Breakthrough emesis occurs despite prophylactic antiemetics.
Refractory emesis failure to respond to prevention or intervention in the previous cycle.
Alternative Etiologies [159]
Medications (opioids, antimicrobials).
Surgery/radiation.
Electrolyte imbalances/dehydration.
Gastrointestinal: obstruction, gastroparesis, constipation.
Psychological (anxiety, anticipatory).
Brain metastasis.
Complications [157, 158]
Metabolic imbalances.
Decreased performance status.
Nutrient depletion/Anorexia.
Wound dehiscence.
Esophageal tears.
Noncompliance with treatment.
Aspiration.
Decreased quality of life.
Risk Factors [154, 157, 158, 160, 161]
Patient Related
Age—increased risk in younger patients.
Gender—increased risk for females.
History of alcohol use—decreased with prior use.
History of CINV.
History of vertigo/motion sickness.
Non-chemotherapy related etiologies.
History of nausea/vomiting with pregnancy.
Chemotherapy Related
>90 % = High risk.
31–90 % = Moderate risk.
10–30 % = Low risk.
<10 % = Minimal risk.
See Table 6.3 for CINV risk for agents used to treat gynecologic cancers.
Table 6.3.
Chemotherapy induced nausea/vomiting (CINV) risk by agent.
Risk category | Agents |
|---|---|
High risk (>90 %) | Cisplatin Dacarbazine Doxorubicin >60 mg/m2 Ifosfamide ≥2 g/m2/dose |
Moderate risk (31–90 %) | Carboplatin Dactinomycin Doxorubicin <60 mg/m2 Epirubicin ≤90 mg/m2 Ifosfamide <2 g/m2/dose Irinotecan Melphalan Methotrexate ≥250 mg/m2 Oxaliplatin Temozolomide |
Low risk (10–30 %) | Docetaxel Liposomal doxorubicin Etoposide Fluorouracil Gemcitabine Mitomycin Paclitaxel Pemetrexed Topotecan |
Minimal risk (<10 %) | Bevacizumab Bleomycin Vinblastine Vincristine Vinorelbine |
Therapeutic Options [2, 160, 168]
High Therapeutic Index Agents
Used primarily for prevention first line breakthrough. Dosing outlined in Table 6.4.
Table 6.4.
High therapeutic index antiemetic common dosing.
Agent | Pre-chemotherapy | Post-chemotherapy |
|---|---|---|
Ondansetron | 8–16 mg IV/PO 30 min prior to | 8 mg PO TID × 3 days |
Dolasetron | 100 mg IV/PO 30 min prior to | 100 mg PO daily × 3 days |
Granisetron | 1 mg IV/PO 30 min prior to; 34.3 mg transdermal patch applied 24–48 h prior to | 1–2 mg PO BID × 3 days |
Palonosetron | 0.25 mg IV 30 min prior to | |
Dexamethasone w/aprepitant 125 mg PO | 12 mg IV/PO 30 min prior to | 8 mg PO daily × 3 days |
Dexamethasone w/fosaprepitant 150 mg IV | 12 mg IV/PO 30 min prior to | 8 mg day 2, 8 mg PO BID days 3 and 4 |
Dexamethasone w/o aprepitant | 8-20 mg IV/PO 30 min prior to | 8 mg PO BID × 3 days |
Aprepitant | 125 mg PO 1 h prior to | 80 mg PO × 2 days |
Fosaprepitant | 150 mg IV 30 min prior to |
5HT3 receptor antagonists:
Agents: Ondansetron, Palonosetron, Granisetron, Dolasetron.
Adverse effects: headache, constipation, QT prolongation.
Corticosteroids.
Agents: Dexamethasone, Prednisone, Methylprednisolone.
Adverse effects: hyperglycemia, insomnia, hypertension, immunosuppression.
Neurokinin-1 receptor antagonists.
Agents: Aprepitant, Fosaprepitant.
Adverse effects: headache, hiccups, fatigue.
Moderate inhibitor and inducer of CYP3A4, weak inducer of CYP2C9.
Low Therapeutic Index Agents
Used primarily for breakthrough N/V. Dosing outlined in Table 6.5.
Table 6.5.
Low therapeutic index antiemetic common dosing.
Agent | Dosing |
|---|---|
Promethazine | 6.25–25 mg IV/PO q6h prn 25 mg PR q6h prn |
Prochlorperazine | 5–10 mg IV/PO q6h prn 25 mg PR q6h prn |
Metoclopramide | 0.5–2 mg/kg IV q4h prn (must give w/diphenhydramine 25 mg IV q6h to prevent extrapyramidal side effects) 10–40 mg PO q6h prn |
Olanzapine | 2.5–5 mg PO qHS |
Alprazolam | 0.5–2 mg PO prior to chemotherapy |
Lorazepam | 1–2 mg IV/PO prior to chemotherapy 0.5–2 mg PO q4h prn N/V |
Haloperidol | 1 mg IV q4h PRN |
Dronabinol | 5–10 mg PO q3h prn |
Nabilone | 1–2 mg PO q12h prn |
Phenothiazines.
Agents: Prochlorperazine, Promethazine.
Adverse effects: sedation, anticholinergic effects, extrapyramidal side effects.
Metoclopramide.
Adverse effects: sedation, extrapyramidal side effects, diarrhea.
Olanzapine.
Adverse effects: sedation, weight gain.
Benzodiazepines.
Drug of choice for anticipatory N/V.
Agents: Lorazepam, Alprazolam.
Adverse effects: sedation, amnesia.
Butyrophenones.
Agents: Haloperidol
Adverse effects: sedation, constipation, arrhythmias, extrapyramidal side effects.
Cannabinoids.
Agents: Dronabinol, Nabilone.
Adverse effects: sedation, abnormal thinking, palpitations, tachycardia, euphoria.
General Principles of Treatment [155, 160, 161, 165]
Primary goal is prevention of CINV.
Agents are chosen based upon chemotherapy regimen.
Consider toxicity of antiemetics used.
Always provide “rescue” medication for breakthrough CINV.
Treatment Recommendations [160, 161, 165, 169, 170]
High Risk Chemotherapy
Acute Emesis Prevention.
5HT3 antagonist + Dexamethasone + Neurokinin 1 antagonist +/− lorazepam +/− H2 blocker or proton pump inhibitor.
Delayed Emesis Prevention.
If fosaprepitant 150 mg: dexamethasone 8 mg PO day 2 then 8 mg PO BID days 3–4.
If aprepitant day 2–3: dexamethasone 8 mg PO days 2–4.
Moderate Risk Chemotherapy
Acute Emesis Prevention.
5HT3 antagonist + dexamethasone +/− Neurokinin 1 antagonist +/− lorazepam +/− H2 blocker or proton pump inhibitor.
Delayed Emesis Prevention.
5HT3 antagonist monotherapy for 2–3 days, OR.
Dexamethasone monotherapy for 2–3 days, OR.
Neurokinin 1 antagonist (if used day 1) + dexamethasone.
Low Risk
Prior to chemotherapy.
Dexamethasone PO/IV, OR.
Metoclopramide PO/IV, OR.
Prochlorperazine PO/IV, OR.
+/− Lorazepam and/or H2 blocker or proton pump inhibitor.
Minimal Risk
No prophylaxis recommended.
Multiday Chemotherapy Regimens
Consider emetogenic potential of each day.
5-HT3 antagonist should be administered daily for moderately or highly emetogenic chemotherapy.
Dexamethasone should be given daily prior for moderately or highly emetogenic chemotherapy.
Prevent delayed emesis with 2–3 days of prophylaxis following chemotherapy.
Palonosetron or transdermal granisetron may be used in lieu of daily 5HT3 dosing.
Dosing of Aprepitant beyond 3 days has been shown to be safe and effective in phase II trials.
Repeat doses of palonosetron have been studied and shown to reduce CINV.
Breakthrough Emesis
Add agent from different class.
PO administration often unfeasible due to emesis.
Routine administration of “rescue” medication should be considered.
Multiple concurrent agents in alternating schedules.
Reevaluate for alternative etiologies.
Change regimen for next cycle.
Chemotherapy-Induced Diarrhea
Introduction [171–173]
Many chemotherapy agents can cause damage to the intestinal mucosa ultimately resulting in diarrhea. If not managed properly chemotherapy-induced diarrhea can result in treatment delays, dose reductions, and serious complications that may be fatal. Most agents for the primary treatment of gynecologic oncology do not commonly cause diarrhea but a number of agents used for recurrence or rare tumor types are known to cause diarrhea.
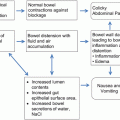
Pathogenesis [171–178]
Direct damage to intestinal mucosa (fluorouracil, capecitabine, Irinotecan late-onset, doxorubicin, gemcitabine, dacarbazine).
Cholinergic stimulation (Irinotecan acute-onset).
Inhibition of vascular endothelial growth factor (pazopanib).
Dihydropyrimidine dehydrogenase (DPD) deficiency and thymidylate synthetase gene (TYMS) polymorphism can increase severity of diarrhea with fluorouracil and capecitabine.
Irinotecan is metabolized by the enzyme uridine diphospho-glucuronosyltransferase 1A1 (UGT1A1).
Patients that are heterozygous or homozygous for UGT1A1*28 may be at increased risk for diarrhea.
Signs and Symptoms [171–173, 179]
Increase in number of stools or ostomy output.
Dehydration.
Renal insufficiency.
Electrolyte abnormalities (hypokalemia, metabolic acidosis, hyponatremia, or hypernatremia).
Fatigue.
Decreased quality of life.
Noncompliance with treatment.
Evaluation [171–173, 179]
Determine onset and duration.
Assess for alternative etiologies (infection, medication, radiation, diet, colitis, etc.).
Consider testing for DPD deficiency, TYMS variants, or UGT1A1 polymorphism.
Determine severity (Table 6.6).
Table 6.6.
Severity/grade of chemotherapy-induced diarrhea.
Grade 1
Grade 2
Grade 3
Grade 4
Grade 5
Diarrhea
Increase of <4 stools per day over baseline; mild increase in ostomy output compared to baseline
Increase of 4–6 stools per day over baseline; moderate increase in ostomy output compared to baseline
Increase of >6 stools per day over baseline; incontinence; severe increase in ostomy output compared to baseline; limiting self-care ADL
Life-threatening consequences; urgent intervention indicated
Death
Identify causative agent.
Therapeutic Options [2, 162–164, 167, 171–173, 179, 180]
General Principles
Treat any alternative/underlying etiologies.
Provide supportive care in the form of hydration and electrolyte repletion.
Severe cases may require delays or dose reduction of chemotherapy.
Scheduled doses of antidiarrheal are usually necessary to control symptoms.
Once controlled medications can be titrated to maintain control.
Nonpharmacologic Treatment
Avoid diarrhea inducing foods (dairy, spicy foods, alcohol, caffeine, high fiber).
Discontinue all laxative, stool softeners, or promotility agents.
Aggressive oral hydration (8–10 glasses per day).
Small frequent meals.
Pharmacologic Treatment
Loperamide.
Rapid onset of action.