38 Cancer of the Pancreas
Epidemiology, Causal Factors, Genetics, and Cytogenetic Abnormalities
Pancreatic cancer is the fifth leading cause of cancer mortality among men and women of all ages; it strikes an estimated 37,680 Americans annually and caused 34,290 deaths in 2008.1 A National Cancer Institute (NCI) survey covering 1973 to 1988 showed less than 20% of affected patients survived 1 year and only 3% were alive 5 years after diagnosis.2,3 Even with the addition of adjuvant radiation and chemotherapy as well as recent decreases in surgical morbidity, only very modest improvements in survival have been realized. Worldwide, the reported incidence varies considerably. It is unclear whether this represents a true phenomenon or whether it is a reflection of more meticulous reporting patterns in more industrialized countries. In the United States, the incidence of pancreatic cancer is 9 in 100,000 in whites and 15.2 in 100,000 in blacks with a male-to-female ratio of 1.3 : 1. Diagnosis is rare before age 45 but rises sharply thereafter.2 Although the cause of pancreatic cancer is unknown, several environmental factors have been implicated. Smoking, in particular, raises the relative risk 1.5 times.4 Diets high in meat or fat also have been linked to increased risk, and a diet of fresh fruits and vegetables has been found to be protective.5 An increased incidence also is found with a prior history of surgery for peptic ulcer disease.6
Much controversy was generated in the early 1980s with a report linking pancreatic cancer with coffee consumption,7 but subsequent prospective studies have failed to confirm this.8 Both diabetes mellitus7,9 and chronic pancreatitis10 are associated with pancreatic cancer, but it is unclear whether this is a cause-and-effect relationship. Workers employed in manufacturing 2-naphthylamine, benzidine,11 and gasoline12 are reported to have a fivefold increased risk. More recently, the study of deoxyribonucleic acid content, oncogenes, and molecular biology has yielded intriguing data.13 Molecular histology data have been nicely summarized by Hruban.14 More and more evidence paints a picture of pancreatic cancer as a disease of acquired and inherited mutations. It was not until very recently that an international system of nomenclature was adopted that reflected the development of pancreatic ductal carcinoma as a continuum analogous to changes seen in the development of cervical carcinoma.15 Pancreatic intrepithelial neoplasia 1-3 is used to grade noninvasive early changes of pancreatic ductal neoplasia. Tumor suppressor genes regulate cell proliferation and their inactivation can play a profound role in the development of malignancies. There are four specific genes identified as crucial in the development of pancreatic cancer: p16, p53, DPC4, and BRCA2. An increased incidence of pancreatic cancer has been seen in families with the breast cancer-associated BRCA2 gene.16 Oncogenes become active by mutation or amputation. An example is the k-ras oncogene, which is often activated in human pancreatic duct carcinomas.17–21 Data from Almoguera and colleagues21 demonstrated that 95% of pancreatic adenocarcinomas contain k-ras oncogenes activated by a mutation at codon 12. This mutation is critical to oncogenesis and may mediate resistance to epidermal growth factor receptor inhibitors. Shibata and associates18 noted similar findings in 72% of patients undergoing fine needle aspiration. Kondo and colleagues22 also found specific mutations of the k-ras oncogene at codon 12 in 67% of pure pancreatic juice collected endoscopically in patients with proven pancreatic carcinoma. K-ras abnormalities were not detected in healthy controls, in patients with chronic pancreatitis, or in three patients with islet cell tumors.13
A summary of genetic abnormalities is provided in Table 38-1. The identification of consistent genetic abnormalities and the observation of higher cancer rates in some families were the genesis of the Johns Hopkins University Hospitals’ National Familial Pancreas Tumor Registry. Familial cases are defined as those in which two or more first-degree relatives are affected. In these individuals the risk extends to second-degree relatives who have a 3.7% chance of developing pancreatic cancer as opposed to 0.6% in second-degree relatives of sporadic cases.23
Table 38-1 Genetic Abnormalities in Pancreatic Cancer
| Inactivated/Suppressor Genes | Frequency Seen, % |
|---|---|
| K-ras | >90 |
| p53 | 50-70 |
| p16 | 95 |
| MKK4 | <10 |
| DPC4 | 50 |
| BRCA2 | 70 |
Anatomy
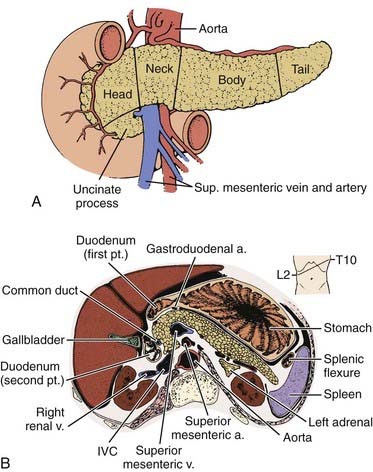
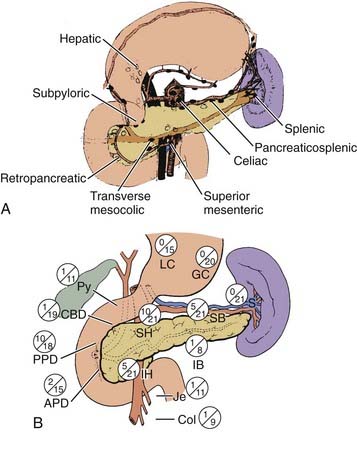
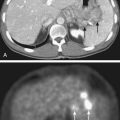
The pancreas is an elongated, coarsely lobulated gland lying transversely and retroperitoneally in the posterior abdomen at approximately the L1 to L2 level (Fig. 38-1A). The head lies in the duodenal flexure on the right and the tail extends to the spleen. The pancreas is divided into head, uncinate process (considered to be part of the head), neck, body, and tail. Tumors arising to the right of the superior mesenteric vein (SMV) are considered to be in the head, those arising to the left of that vein and left border of the aorta are part of the body, and those arising from the tail are located between the left border of the aorta and the hilum of the spleen. The pancreas is in close contact with surrounding organs including the spleen, stomach, duodenum, jejunum, and kidneys, all of which may be involved with tumor early on (see Fig. 38-1B). A rich lymphatic network surrounds the pancreas, including the celiac, superior and inferior pancreaticoduodenal, superior mesenteric, porta hepatis, and pancreaticosplenic nodes (Fig. 38-2A). With posterior extension of tumor, the lateral aortic nodes are also at risk. The main venous drainage of the pancreas is via the portal system to the liver. Tumors of the head of the pancreas often cause jaundice caused by invasion or compression of the common bile duct.
Pathologic Conditions
The most common type of pancreatic cancer is of ductal origin, comprising from 75% to 90% of patients.24 It is twice as common in the head as in the body or tail. Perineural invasion occurs in approximately 90% of patients, and in more than 85% of patients tumor has extended beyond the organ at the time of diagnosis. The most common extralymphatic sites of involvement are the liver and peritoneum; the lung is the most commonly involved extra-abdominal organ.
Less frequently occurring exocrine tumors, such as cystadenocarcinoma or intraductal carcinoma, are more common in women and may run a much more indolent course,25,26 remaining localized for many years and having up to a 50% five-year survival rate.27 Solid and cystic papillary neoplasms, also known as Hamoudi tumors, occur in women in their third decade of life, only rarely metastasize, and have a good prognosis. Rare acinar cell cancers are associated with fat necrosis and high lipase production and have a poor prognosis; they may be associated with a clinical picture that includes rash, eosinophilia, and polyarthralgia. Giant cell tumors, which account for only a small percentage of pancreatic cancers, are very large and aggressive and have a very poor survival rate.28 Clinical presentation of these less aggressive histologies, however, is indistinguishable from the more aggressive pathologic subtypes.25 Cancer metastatic to the pancreas is a relatively uncommon finding. In a series of 2587 consecutive autopsies at the Memorial Sloan-Kettering Cancer Center (MSKCC), 10% of patients were found to have metastases in the pancreas with the most frequent primary sites being breast, lung, or melanoma.24 The remaining 5% of pancreatic cancers are of endocrine origin and are discussed separately.
Clinical Presentation
More than 80% of patients present with pain, jaundice, or both29,30; these symptoms, along with weight loss, constitute a classic triad. Tumors in the body and tail may not cause jaundice or pain until far advanced; thus their prognosis is particularly poor,31 whereas periampullary cancers have a significantly better prognosis32 because of earlier clinical presentation. Tumors of the endocrine pancreas may present in a similar fashion but are often recognized because of various clinical syndromes produced by circulating polypeptides elaborated by the tumor.33 Rarely, presenting symptoms may be diabetes mellitus9 or acute pancreatitis.10 Infrequently, patients may present with migratory thrombophlebitis (Trousseau sign) or with a palpable gallbladder (Courvoisier sign). Abnormal laboratory tests, such as alteration in glucose, amylase, lipase, bilirubin, alkaline phosphatase, lactate dehydrogenase, or aspartate aminotransferase, may all be present but also are all nonspecific. The search for screening tests to allow for earlier diagnosis and, potentially, to effect cure has generally been elusive with the exception of the Cancer of the Pancreas 2 Screening Project. High-risk but asymptomatic individuals were assessed using endoscopic ultrasound (EUS), and the detection of premalignant and preinvasive lesions suggests that EUS may play a role in screening.34
Routes of Spread
Most pancreatic cancers are unresectable at presentation and thus there is sparse literature systematically analyzing patterns of failure. Cubilla and associates35 reported on the incidence of nodal involvement in completely resected early cancers of the head of the pancreas (see Fig. 38-2B) and showed that carcinomas in the head tended to spread to multiple nodal groups. In addition to initial spread to regional lymph nodes, early peritoneal seeding may be exceedingly common. A series from Massachusetts General Hospital (MGH) reports a 40% incidence of peritoneal studding in patients clinically considered to be resectable and who first underwent laparoscopy before definitive resection.36 In a patient who has undergone curative therapy, once the therapy fails, local recurrence again dominates the clinical picture.37,38 In a series of Griffen and associates,38 only 17% of patients developed disease purely outside the abdominal cavity (Fig. 38-3).
Diagnostic and Staging Studies
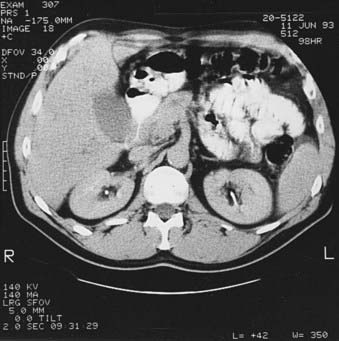
Preoperative staging of pancreatic cancer to detect resectability is extremely important to avoid unnecessary surgery. Table 38-2 reflects the staging system as revised in 2010, with the T classification distinguishing between potentially resectable (T3) and unresectable (T4) primary tumors. The stage grouping allows stage III to signify unresectable, locally advanced pancreatic cancer; stage IV is reserved for patients with metastatic disease. Pancreatic cancer is considered resectable if there is no portal vein or celiac axis involvement and no metastasis evident. Borderline resectability may include unilateral or bilateral SMV/portal vein impingement, less than 180° tumor abuttment, or encasement of the hepatic artery or SMV if the surgeon determines it is reconstructible.39 Magnetic resonance imaging (MRI), ultrasound, and computed tomography (CT) (Fig. 38-4) can detect pancreatic cancers as small as 1 cm. All three modalities easily detect dilatation of the pancreatic and bile ducts as well as liver metastases.40 Ultrasound can help distinguish between obstructive and nonobstructive jaundice, but CT is more helpful in providing definition of the tumor and surrounding structures. A clinical suspicion of pancreatic cancer or evidence of a dilated pancreatic duct warrants a pancreatic CT scan obtained via a defined pancreas protocol such as a helical or spiral triphasic crosectional CT with thin slices. The quality of modern imaging is so good as to predict resectabilty in 80% to 85% of cases when appropriate criteria are met.41,42 If cancer is fairly certain, the scan should include the chest to rule out metastatic lung disease. Positron emission tomography (PET) is considered a complementary study, as is EUS, which is of particular usefulness in the assessment of vascular invasion and thus resectability.43,44 Specifically, PET scan is helpful in characterizing equivocal lesions on CT and MRI, but is not helpful as a staging tool.45 Advances in technology have improved both the sensitivity and predictive value of cross-sectional imaging to such an extent that abdominal ultrasound, cholangiography, and angiography are no longer required for accurate staging. Percutaneous ultrasound or CT-guided biopsy can safely and easily provide pathologic diagnosis in approximately 90% of cases,46 but in the case of adenocarcinoma it may cause increased risk of local failure secondary to rapid seeding of tumor.47 A review from MGH48 showed a fourfold increased risk of positive peritoneal cytology in patients who had prior percutaneous fine needle aspirates. Some institutions thus advocate deferring preoperative biopsy in apparently resectable cases because histologic diagnosis is not required before surgery, although it is clearly required before embarking on neoadjuvant therapy or declaring a patient unresectable or having metastatic disease. Older literature suggested a benefit from treating patients already biopsied with low-dose (5 to 10 Gy) preoperative radiation in an attempt to decrease dissemination of tumor.49 This is now very rarely done and would not be considered standard. Endoscopic retrograde cholangiopancreatography (ERCP) is extremely useful in the differential diagnosis of tumors of the pancreaticobiliary junction (Fig. 38-5).50 Ampullary and duodenal cancers can be visualized and biopsied during ERCP; pancreatic duct stricture longer than 10 mm, especially if irregular, is more consistent with carcinoma than pancreatitis.51 Supportive serologic evidence of pancreatic cancer includes elevated levels of cancer antigen (CA) 19-9,52 carcinoembryonic antigen, pancreatic oncofetal antigen, tissue polypeptide antigen, and CA-125. A decrease in serial CA 19-9 levels has been found to correllate with survival; however, caution is warranted because of false elevations in the presence of benign biliary disease or obstruction or false negativity in individuals who are Lewis-a antigen negative. Unfortunately, no currently available marker is diagnostic or sufficiently sensitive or specific for screening purposes.53 Following serial levels of these markers, however, is very useful, and baseline and post-treatment levels of CA 19-9 do correlate with tumor response.54
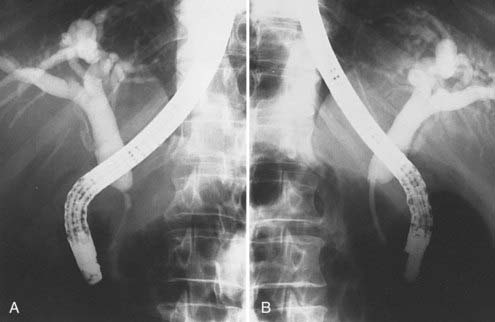
FIGURE 38-5 • Endoscopic retrograde cholangiopancreatography of the same patient shown in Fig. 38-4. A, Note thin ragged contour of the pancreatic duct—stenosis of the length (see text) is usually indicative of neoplasm. B, A stent has successfully been placed in the pancreatic duct to relieve obstructive jaundice.
Following this, in patients without prior abdominal surgery, laparoscopy may be used to rule out small hepatic or peritoneal metastasis. Laparoscopic ultrasound in conjunction with peritoneal cytologic findings significantly decreases the nontherapeutic laparotomy rate as positive peritoneal cytologic findings indicate advanced disease. Investigators at MGH prospectively evaluated 88 patients referred for curative resection using MRI and CT, angiography, and laparoscopy for staging. They preoperatively identified 89% of patients who could not have curative resection. These patients are best spared an operation unless duodenal obstruction is present.48 Excellent palliation for jaundice can be achieved with endoscopic placement of large stents.55 Diagnostic workup and tumor-node-metastasis (TNM) classification are outlined in Table 38-2 and Table 38-3. It should be noted that the TNM staging criteria include data obtained both preoperatively and postoperatively and that the American Joint Committee on Cancer also recommends that the surgeon assess the completeness of resection as R0 for complete resection with negative margins, R1 for incomplete resection with microscopically positive margins, and R2 for incomplete tumor resction with gross residual tumor.56 For practical purposes and to better understand the historical literature on the treatment of pancreatic cancer, much of it predating high-resolution imaging modalities, many of the clinical series have reported the conceptual classifications for pancreatic cancer patients as resectable, borderline resectable, locally advanced, and metastatic. This leads to confusion and difficulty in comparing results across institutions and trials.
Table 38-3 Evaluation of Pancreatic Mass
CA, Cancer antigen; ERCP, endoscopic retrograde cholangiopancreatography; GI, gastrointestinal; MRI, magnetic resonance imaging; PET, positron emission tomography.
Therapeutic Approaches
Options for treating any malignancy include surgery, radiation, and chemotherapy, either singly, sequentially, or in combination. The overall disappointing outcomes in the treatment of pancreatic cancer have involved all modalities in every order and combination possible. The standard therapeutic approach to pancreatic carcinoma has been surgical, and the standard operation for cancers in the pancreatic head has been the pancreaticoduodenectomy or Whipple procedure. Complete surgical resection offers the only possibility for cure, yet more than 80% of patients presenting with pancreatic cancer cannot be cured with resection.57 Interestingly, there are no universally accepted criteria defining surgical resectability, although the driving consideration for the multidisciplinary team evaluating a potential surgical candidate is the likelihood of obtaining a complete resection with negative microscopic margins.58,59 Tumors of the body and tail of the pancreas generally present late and thus usually are not resectable. Although the Whipple is the standard procedure, some controversy still surrounds the debate of the optimal surgerical technique. One controversy is the role of preoperative biliary drainage, with a 1999 MSKCC series60 showing increased morbidity, including mortality, and a similar MD Anderson Cancer Center series61 showing no increase in sepsis or death. Thus most institutions selectively decompress the biliary tract in symptomatic patients or in those for whom surgery will be delayed. In most instances, this can be done by ERCP. CA-19-9 levels should be drawn after biliary decompression. Other surgical questions under investigation include the analysis of the optimal pancreatic anastamosis to minimize leaks and whether pylorus preservation offers a better quality of life and nutritional status. Total pancreatectomy has been advocated by some as a better operation because it removes more potentially involved tissue and because it avoids a pancreaticojejunal anastomosis, which is the source of significant surgical morbidity and mortality. However, it does result in exocrine insufficiency and brittle diabetes that is difficult to manage,62 and it confers no survival advantage. Fortner63 advocated a regional pancreatectomy, which is an en bloc resection of the pancreas and surrounding tissue along with a segment of the mesenteric-portal vein and occasionally major arterial segments. This operation is less frequently performed in the United States because of high operative mortality (8% in Fortner’s series) and no clear evidence of survival benefit. Some groups in Japan combine the Whipple procedure with extensive lymph node dissection, claiming better results with no apparent increase in operative mortality.64 The opposite approach has been taken by several groups in the United States65,66 who believe there is no compromise of survival when preserving the pylorus in an attempt to avoid postgastrectomy symptoms. In the 1960s and 1970s, the pancreaticoduodenectomy was associated with a 20% to 25% in-hospital mortality rate.67 By the 1980s and 1990s, surgical techniques and supportive care improved to the point at which this operation now carries only a 1% to 3% mortality rate in major centers.68 With certainty, the pylorus-preserving pancreaticoduodenectomy improves gastrointestinal function and does not appear to compromise further management. The dramatic improvements in surgical techniques have made it possible to conduct an ongoing prospective randomized trial with intraoperative randomization between a standard operation and one with extended retroperitoneal lymph node dissection. What is clear from the surgical literature is that, if tumor is knowingly left behind, survival after resection is not much better than after bypass alone.53 This has led to the question of appropriate surgical management of patients with positive lymph nodes at frozen section. Yeo and colleagues68 have shown that long-term survival out to 5 years is possible in both lymph node–positive and lymph node–negative tumors as long as the patient is otherwise technically suitable for resection. An additional important question answered recently involves determination of the best management of the elderly patient. Sohn and colleagues,69 Magistrelli and colleagues,70 and Balcom and colleagues71 clearly showed that it is appropriate to perform aggressive surgery in appropriately selected patients into their seventh and eighth decades of life. Several other important points from the surgical literature are worth noting. These involve the importance of both a multidisciplinary approach as well as the importance of an experienced team. The importance of an experienced team is further underscored by series from Robinson et al.72 and from Sohn et al.73 at two different institutions, showing the benefit of reoperation at an experienced center: 67% of patients, many deemed unresectable elsewhere, were in fact successfully resected without compromise of their overall outcome. Other series have clearly shown the benefit of decreased in-hospital morbidity and mortality rates as well as survival in those centers with a high volume of cases.74,75
Multiple large series have shown that only 5% to 30% of patients have resectable tumors.64,76–78 Remaining patients require some form of palliation for pain, duodenal obstruction, or jaundice. Obstructive jaundice can be relieved percutaneously, albeit with complications in up to 35% in some series,53 or with stents that have more than an 85% success rate and procedure-related mortality of only 1% to 2%.79 This represents a significant improvement over surgical morbidity. Two randomized trials80,81 have shown equal survival with both surgical bypass and endoscopic stent placement. In addition to analgesics, percutaneous neurolysis of the celiac ganglion and radiation therapy offer good palliation of pain.
Another strategy to treat in patients with resectable pancreatic adenocarcinomas is preoperative treatment with gemcitabine and radiotherapy. The rationale for this approach is as follows: (1) early systemic treatment with gemcitabine addresses risk of micrometastatic disease when tumor burden is lowest, (2) gemcitabine is a potent radiosensitizer, and (3) restaging studies performed after neoadjuvant therapy selects out those patients who are not good surgical candidates because of rapid disease progression. In a phase II study of 86 patients, this approach resulted in a resection rate of 74% having a median survival rate of 34 months. The median survival rate in the entire group of 86 was 22.7 months. The 5-year survival rate in those patients able to undergo successful surgical resection was 36% versus 0% in those who did not undergo surgery.82
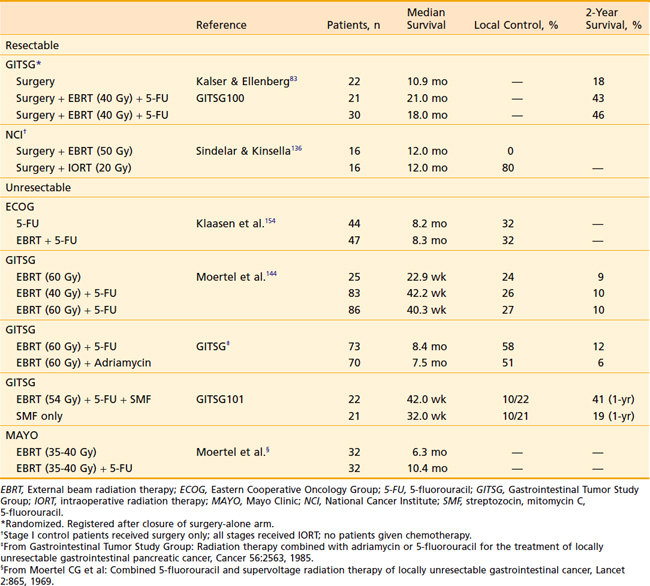
For patients with resectable disease, the role of adjuvant chemoradiotherapy was established by the Gastrointestinal Tumor Study Group (GITSG) in 1985, when it showed that patients receiving adjuvant chemoradiation experienced a doubling of survival time.83 Small series using neoadjuvant treatment were reported in the 1980s, but that approach did not attract general interest until improved outcomes in surgical series were reported.84,85 Historically 5-fluorouracil (5-FU) was the chemotherapeutic agent of choice (Table 38-4) until Burris et al.86 showed an improvement in survival with gemcitabine over 5-FU as first-line therapy for patients with advanced pancreatic cancer. Subsequently, gemcitabine and 5-FU in varying doses and dose rates have been combined with and without radiation with varying results.87–89
Gemcitabine, now the most widely used chemotherapy in pancreatic cancer, has shown significant promise; but combination studies with other chemotherapies and targeted agents have been largely dissappointing. Gemcitabine has been combined with radiation therapy with modest success. However, because of the significant radiosensitizing properties of gemcitabine, reduced radiation dose and field size is necessary if full-dose gemcitabine is administered. Investigators from the University of Michigan90 reported a median overall survival (OS) of 11.2 months in patients (n = 74) with locally advanced pancreatic cancer treated with full-dose gemcitabine (1000 mg/m2, weekly) and concurrent radiotherapy (36 Gy in 2.4-Gy fractions). Using three-dimensional (3-D) conformal techniques, they treated the primary tumor with a 1-cm margin. Despite not treating the regional lymph nodes, only 5% of patients failed regionally, suggesting that treatment of the regional lymph nodes may be omitted. Similarly, Chang et al.91 reported that 0 of 77 patients developed an isolated nodal failure after stereotactic radiotherapy targeting only the primary tumor. Overall, there were nine nodal recurrences, but all were in the context of progressive disease at other sites.
Other strategies for improving the effectiveness of radiotherapy for pancreatic cancer include intensity-modulated radiotherapy (IMRT),92 image-guided radiotherapy (IGRT),93 and stereotactic radiotherapy.94,95
The major difficulty in treatment of pancreatic cancer is not the radioresistance of the tumor, but radiation dose limitations imposed by surrounding normal organs (see Fig. 38-1B) and the frequent presence of occult distant metastases. Attempts to overcome this problem have included the use of intraoperative electrons or implantation of the tumor either directly or percutaneously. Obviously, the advantage offered by IORT is precise field localization and the abilities to shield sensitive normal structures and to deliver a relatively high dose quickly. High-dose-rate brachytherapy has the advantage of reduced radiation exposure to medical personnel and the potential to overcome limitations of attempting to control tumors with a relatively fast doubling time with a low-dose-rate source.
Discouraging survival statistics, a 50% to 76%37,38 locoregional failure even after “curative” resection, and the proven survival benefit of adjuvant treatment83,96 make postoperative radiation with concurrent chemotherapy justifiable in virtually all patients who have had a potentially curative resection of pancreatic cancer. Patients with localized but unresectable cancer can expect doubling of survival with EBRT and concurrent 5-FU.97–99 They should be offered treatment unless poor clinical status precludes treatment.
Simulation
With the use of multiple fields and high-energy photons, doses of 60 to 65 Gy can be delivered safely.100 The addition of concurrent chemotherapy typically requires dose reduction to avoid excessive toxicity. With the widespread development and availability of IMRT techniques, increased dose intensity to the tumor is possible while minimizing radiation dose to adjacent normal tissues. Furthermore, reduced tumor margins also are possible with the application of IGRT techniques.