Abstract
As the field of cancer rehabilitation grows, the need for skilled management of patients with brain tumors is needed. New chemotherapeutic agents, new surgical techniques, and updated protocols for radiation therapy delivery have increased survivability and decreased morbidity. Interventions by multidisciplinary rehabilitation professionals have been shown to improve functional outcomes and quality of life. The purpose of this chapter is to discuss medical and therapeutic interventions in the treatment of patients with benign and malignant intracranial tumors.
Keywords
Brain, Function, Management, Pain, Rehabilitation, Tumor
Epidemiology
There are 700,000 people living in the United States with a primary brain tumor. Approximately 80,000 new cases of primary brain tumors are expected to be diagnosed in 2017, with 26,000 of these expected to be malignant. There are an expected 16,700 deaths in 2017 from primary brain tumors accounting for 2.8% of all cancer deaths. The median age for diagnosis of primary brain tumors is 59 years old; however, these are the most common cancer in those aged 0–14 years.
Tumor Types
As of 2016, the World Health Organization (WHO) updated the brain tumor staging recommendations to include not only microscopic histology but also by incorporating molecular parameters into the classification of central nervous system (CNS) tumors. Tumors are staged from I to IV, with stage I tumors classified the most benign and stage IV being the most malignant ( Table 5.1 ). The most common type of brain tumor is a meningioma, which represents nearly 37% (27,110 in 2017) of all primary brain tumors. Gliomas account for 24.7% of all brain tumors and 74.6% of all malignant tumors. Tumors included within the glioma category are astrocytomas (which include glioblastomas), oligodendrogliomas, and ependymomas. Gliomas carry the highest mortality rate among brain tumors, with a median survival rate of approximately 12–15 months despite many advances in treatment. Glioblastomas account for 14.9% of all primary brain tumors and 55.4% of all gliomas, with an estimated 12,390 new diagnoses in 2017. Pituitary tumors a typically benign and there are an anticipated 14,230 new tumors in 2017 (16% of all primary brain tumors). Lymphomas and oligodendrogliomas account for 2% of primary brain tumors, whereas medulloblastomas represent 1%. Within an inpatient rehabilitation population, 20%–30% of brain tumor admissions are glioblastomas and approximately 20% are meningiomas.
| Diffuse astrocytic and oligodendroglial tumors | Desmoplastic infantile astrocytoma and ganglioglioma | I | |
| Diffuse astrocytoma, IDH-mutant | II | Papillary glioneuronal tumor | I |
| Anaplastic astrocytoma, IDH-mutant | III | Rosette-forming glioneuronal tumor | I |
| Glioblastoma, IDH-wild type | IV | Central neurocytoma | II |
| Glioblastoma, IDH-mutant | IV | Extraventricular neurocytoma | II |
| Diffuse midline glioma, H3 K27M-mutant | IV | Cerebellar liponeurocytoma | II |
| Oligodendroglioma, IDH-mutant and 1p/19q codeleted | II | Tumors of the pineal region | |
| Anaplastic oligodendroglioma, IDH-mutant and 1p/19q-codeleted | III | Pineocytoma | I |
| Other astrocytic tumors | Pineal parenchymal tumor of intermediate differentiation | II or III | |
| Pilocytic astrocytoma | I | Pineoblastoma | IV |
| Subependymal giant cell astrocytoma | I | Papillary tumor of the pineal region | II or III |
| Pleomorphic xanthoastrocytoma | II | Embryonal tumors | |
| Anaplastic pleomorphic xanthoastrocytoma | III | Medulloblastoma (all subtypes) | IV |
| Ependymal tumors | Embryonal tumor with multilayered rosettes. C 19MC-altered | IV | |
| Subependymoma | I | Medulloepithelioma | IV |
| Myxopapillary ependymoma | I | CNS embryonal tumor. NOS | IV |
| Ependymoma | II | Atypical teratoid/rhabdoid tumor | IV |
| Ependymoma. RELA fusion-positive | II or III | CNS embryonal tumor with rhabdoid features | IV |
| Anaplastic ependymoma | III | Tumors of the cranial and paraspinal nerves | |
| Other gliomas | Schwannoma | I | |
| Angiocentric glioma | I | Neurofibroma | I |
| Chordoid glioma of third ventricle | II | Perineurioma | I |
| Choroid plexus tumors | Malignant peripheral nerve sheath tumor (MPNST) | II, III, or IV | |
| Choroid plexus papilloma | I | Meningiomas | |
| Atypical choroid plexus papilloma | II | Meningioma | I |
| Choroid plexus carcinoma | III | Atypical meningioma | II |
| Neuronal and mixed neuronal-glial tumors | Anaplastic (malignant) meningioma | III | |
| Dysembryoplastic neuroepithelial tumor | I | Mesenchymal, nonmeningothelial tumors | |
| Gangliocytoma | I | Solitary fibrous tumor / hemangiopericytoma | I, II, or III |
| Ganglioglioma | I | Hemangioblastoma | I |
| Anaplastic ganglioglioma | III | Tumors of the sellar region | |
| Dysplastic gangliocytoma of cerebellum (Lhermitte-Duclos) | I | Craniopharyngioma | I |
| Granular cell tumor | I | ||
| Pituicytoma | I | ||
| Spindle cell oncocytoma | I | ||
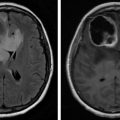
The exact incidence of metastatic brain tumors is not known; however, metastatic disease to the brain is diagnosed in between 200,000 and 300,000 patients per year. These are diagnosed with computed tomography (CT) or magnetic resonance imaging (MRI) when neurologic symptoms manifest. Lung, breast, melanoma, colon, and renal carcinomas are the most common primary sites for metastatic spread to the brain. The majority (approximately 80%) of metastatic brain lesions present as multiple tumors, whereas only 15%–20% present as a single lesion. A total of 85% of metastatic lesions are located within the cerebrum, whereas 15% present within the cerebellum. The probability of cerebellar metastases is highest in lung and breast cancers; however, the majority of brain metastases are located within the watershed areas of the brain such as the gray-white matter junction. The location of the lesion corresponds to the symptoms that manifest within the patient ( Fig. 5.1 ). A total of 25%–30% of brain tumors admitted to inpatient rehabilitation facilities are due to metastatic disease.

Diagnosis
Presenting Symptoms
Patients with brain tumors may present with a variety of symptoms that may be nonspecific or are more closely correlated to intracranial pathology. The most common presenting symptom of a tumor is a headache, with 40%–80% being tension type and migraine in approximately 10%. Epileptic seizures may also be a functionally limiting complication of patients with metastatic and primary brain malignancy. Approximately 50% of patients with an intracranial malignancy will have an epileptic episode during their disease. As previously stated, focal neurologic deficits as presenting symptoms of malignancy are dependent on the location of the tumor. Physical examination, including fundoscopic examination which could signal increase in intracerebral pressure, is key to finding a diagnosis.
Imaging
Structural MRI remains the standard of care for diagnosis and in management of intracranial neoplasms. The primary role of structural MRI is to determine the location of the tumor, extent of involvement, and the effect of compression due to edema on the brain parenchyma, vessels, or other structures. Standardized protocols exist and the recommended imaging sequence involves a three-dimensional (3D) T1, axial fluid-attenuated inversion recovery (FLAIR), axial diffusion-weighted imaging (DWI), axial gadolinium contrast–enhanced T2, and 3D gadolinium contrast–enhanced T1 obtained on a 1.5 T MRI system. Two-dimensional sequences can be substituted if time is limited or if there are other contraindications. Structural sequences, which include T2-weighted, FLAIR, and T1-weighted images with and without contrast, are the basis for MRI examinations. For presurgical examinations, 3D T2-weighted images and 3D T1-weighted images may be useful in planning.
MRI offers superior views of the soft tissues of the brain as opposed to other neuroimaging techniques. Intravenous contrast shows enhancement of areas that have had disruption of the blood-brain barrier (BBB). Degree of contrast enhancement is highly correlated with tumor grade; however, some lower grade gliomas, i.e., WHO grade I tumors such as pilocytic astrocytoma and ganglioglioma, have intense enhancement signals. Hyperintense signals on T2 and FLAIR imaging is identified as peritumoral edema and can be vasogenic or infiltrative edema. Vasogenic edema is due to increase in extracellular fluid and plasma due to poorly formed capillaries feeding into the tumor. It can also be seen in metastatic disease or noninfiltrating tumors such as meningiomas. Infiltrative edema associated with gliomas is due to a combination of vasogenic edema and tumor cells that penetrate white matter tracts within the brain. This enhancement may be difficult to differentiate from the actual primary mass lesion. Contrast-enhanced MRI is more sensitive that nonenhanced MRI or CT in detecting intracranial lesions, either primary or metastases. However, CT may be used in patients that have a contraindication to MRI.
There has been increased interest in functional MRI (fMRI) and its utilization in diagnosing and prognostic value in intracranial neoplasms. fMRI measures activity of neurons by giving a ratio of the deoxyhemoglobin to oxyhemoglobin in the brain and is reported as the blood oxygen level dependent (BOLD) signal. The interest lies in the fact that fMRI can be used for mapping of sensorimotor, language, and memory, which can have significant impact on surgical planning and intraoperative evaluation. Task-based fMRI involves asking the patient to oscillate between a resting and active state while performing a motor or language task. This technique can also be used to characterize tumors. A decrease in the BOLD signal in the affected brain tissue shows alterations in blood flow, which can be used to distinguish high- v. low-grade tumors. Resting state fMRI (rs-fMRI) does not require the patient to perform any tasks and can be performed while the patient is receiving anesthesia. At times there are small fluctuations in BOLD signals known as resting state networks (RSNs). When compared with task-based fMRI, rs-MRI can map the brain quickly using RSN in a shorter amount of time. Although promising, rs-fMRI needs further investigation before it has more clinical utility.
As previously stated, MRI is the gold standard for imaging in regard to diagnosis of gliomas; however, MRI is not without its limitations. MRI alone oftentimes gives limited information when grading a tumor or delineation of the tumor. There is also difficulty in distinguishing between necrotic tissue and recurrent tumor. A rapidly enlarging, contrast-enhanced tumor is usually considered a progressive lesion, even in the absence of worsening neurologic dysfunction, as opposed to a treatment side effect such as radiation necrosis.
Recent studies have shown that positron emission tomography (PET) scanning has utility in the diagnosis of gliomas. Owing to improved ability to radioactively label amino acids, PET scans can be used to aid in therapy planning and monitoring in glioblastomas. PET scanning can also be used in noninvasive grading of gliomas. There is also better delineation of tumor invasion, more precision with surgical or radiation planning, and improved posttreatment monitoring and prognostic evaluations. 18 F-2-fluoro-2-deoxy- d -glucose ( 18 F-FDG) is the most well-studied and validated radiotracer available in nuclear medicine literature. It has also been proven to be cost effective. In lower grade gliomas, 18 F-FDG is used to monitor for anaplastic transformation and can be used in prognosis. This technique is useful because it can be used to assess biologic activity in living organisms. This technique is not without its limitations though. High uptake of 18 F-FDG in normal, healthy brain tissue limits its use in cerebral brain tissue.
Biopsy
Biopsy is used in cases when the diagnosis with imaging is inconclusive or needs to be confirmed. Intraoperative stereotactic biopsy (STB) showed correlation with the actual diagnosis 90% of the time, and a 96% sensitivity in obtaining a diagnostic specimen. More recent studies have been examining other techniques to obtain biopsies when needed and in a more cost-effective and in a time conscious manner. When compared with CT-guided STB, ultrasound-guided biopsy (USGB) showed diagnostic utility of 93%–91% with a procedure time of 149–94 min. Three hematomas were seen the USGB group which were managed conservatively.
Initial Management
Initial medical management of patients presenting with brain tumors is imperative to minimize risk of complications and irreversible brain damage associated with delay. The following section discusses some of these early interventions.
Steroids
Nearly all patients with an intracranial neoplasm receive steroids during their disease. However, there is not a standardized method of dosing or weaning. Steroids help minimize symptom burden due to associated vasogenic edema. Dexamethasone has been the steroid of choice due to its low mineralocorticoid activity, but methylprednisolone and prednisone have also been used. Dexamethasone has a half-life of 36–54 hours which provides extended coverage after dosing. Standard starting dose is 16 mg divided into four daily doses. The goal of steroid management is to taper to the minimum effective dose once the patient is symptomatically stable. This dose will minimize symptom burden and maximize functional benefit while minimizing risk of the long-term side effects associated with prolonged steroid use. One study showed 29% of patients were able to be weaned off steroid therapy within 3 months of completion of radiation ; however, 55%–58% of patients had to have steroid dosage increased during radiation therapy (RT). Steroid-induced myopathy is a potential complication of long-term steroid usage and manifests as proximal muscle weakness most notably in the lower extremities, but in more severe cases, it can be seen in the neck extensor muscles and respiratory muscles. Other more common side effects include anxiety, insomnia, psychosis, and delirium. Hiccups may also be seen and can be treated with baclofen among other medications. Despite a lack of definitive evidence showing gastrointestinal (GI) bleeding in patients on corticosteroids are often prescribed a proton pump inhibitor or an H 2 receptor antagonist. Associated endocrine side effects including Cushing’s syndrome and steroid-induced hyperglycemia usually resolve upon discontinuation of therapy. Corticosteroids also have detrimental effects on bone. Fractures of the spine and hip can also occur, with impaired calcium absorption being the proposed mechanism. Supplementation of calcium and vitamin D should be considered. Avascular necrosis of the hip can also occur. There are also other potential benefits of dexamethasone in brain tumor patients. A recent case report showed potential antineoplastic effects in glioblastoma multiforme (GBM) with dexamethasone and levetiracetam; however, this would need to be further proven by randomized control trials. Affronti et al. also showed that dexamethasone in combination with palonosetron was effective in preventing chemotherapy-induced nausea and vomiting (CINV) in patients receiving treatment for gliomas while maintaining quality of life (QoL). Palonosetron, dexamethasone, and aprepitant were also effective in improving CINV in patients receiving temozolomide.
Seizure Management
Seizures are among the most common presenting symptoms in patients with brain tumors. Surgical resection of the malignancy has been shown to effectively control the seizure. Location of the tumor is an important determinant in the likelihood of having seizures during the illness. Seizures are more common in oligodendrogliomas that are located in the cortex and in lesions of the insula and temporal lobe. White matter and posterior fossa tumors do not often cause seizures, although multifocal deep tumors can be the cause of epileptic episodes. Metastatic disease causes seizures in 20%–40% of patients when they are hemorrhagic, multiple, and/or involve the temporal lobe. There is conflicting evidence about the use of chemoradiation therapy in brain tumor treatment and its effectiveness in managing seizure activity, and further randomized control trials are recommended.
Administration of antiepileptic drugs (AEDs) is standard of care in patients with intracranial malignancy who present with seizure activity, and a 1996 survey found that 55% of practitioners administered prophylactic AEDs to brain tumor patients. Although the use of prophylactic AEDs seems to becoming more of a routine practice, this has not been proven to statistically significantly reduce the risk of seizures. Further clinical trials are recommended. The use of AEDs is often limited by side effect profiles and drug interactions; however, newer AEDs such as levetiracetam, lamotrigine, lacosamide, and pregabalin are better tolerated. Levetiracetam has no known drug interactions and has become a favorite among clinicians as a prophylactic AED. Lacosamide has been proven to be an effective second-line therapy in conjunction with other AEDs or as monotherapy in reducing seizures associated with brain tumors. Valproic acid has also been shown to be effective; however, as it is a CYP450 inhibitor, it may increase toxicity associated with certain chemotherapeutic medications.
Venous Thromboembolism Risk Management
Patients with brain tumors are at high risk for developing deep venous thrombosis (DVT) or pulmonary embolism (PE). DVT or PE has been shown to occur in glioma patients 3%–20% of the time and in up to 60% of GBM patients. This alone should give providers a low threshold for investigation of potential VTE if needed; however, treatment of these is contingent on whether the patient has a hemorrhagic lesion or if there is an increased risk for hemorrhage. Metastatic disease from lung and breast cancer has a lower incidence of hemorrhage when compared with patients with melanoma or renal cell carcinoma. If there are contraindications to chemoprophylaxis in patients with a known DVT, consideration can be made for placement of an inferior vena cava filter. These should be restricted to patients with absolute contraindications to chemoprophylaxis. Low-molecular weight heparin products, such as enoxaparin, can be used for prophylactic and treatment dosing in patients with VTE. Unfractionated heparin products are generally used in patients with renal insufficiency or at higher risk for bleeding due to its ability to be reversed. More recently, direct Xa inhibitors, such as rivaroxaban, have been used in treating VTE in oncology patients. Rivaroxaban received approval in 2012 after the EINSTEIN trials for treatment of VTE; however, these data could not be reliably extrapolated to an oncology population as only 5% of the population studied had active cancer and the study did not account for complications that arise during cancer treatment such as drug interactions, bleeding, and thrombocytopenia. Mantha et al. explored this in a 2017 study in patients who had a cancer-related VTE with similar rates of bleeding risk and recurrent VTE as other published studies. However, this study did not include patients with a primary brain tumor, only those with metastatic disease. A recent case report showed that severe postoperative bleeding was found in a patient with a DVT started on rivaroxaban after thoracic surgery for lung cancer indicating that caution should be used when initiating any anticoagulant.
Treatment
Chemotherapy
The treatment of a brain tumor involves a combination of surgery, chemotherapy, and RT. The first-line chemotherapy for GBM is temozolomide, which is an oral alkylating agent. The effectiveness of this chemotherapy is determined by the methylation status of the O 6 -methylguanine methyltransferase (MGMT) genes. MGMT functions to displace alkyl groups from compounds and works within the DNA direct reversal repair pathway. Loss or silencing of this gene by methylation may increase the likelihood of developing malignancy with exposure to other alkylating agents. High levels of MGMT within malignant cells create temozolomide resistance by blocking the effectiveness of the medicine. Therefore, methylation of MGMT increases the efficacy of temozolomide. Temozolomide has also been shown to improve long-term survival in patients with anaplastic gliomas with leptomeningeal spread. More recent studies suggest that the addition of the vascular endothelial growth factor (VEGF) receptor inhibitor cediranib to the standard GBM treatment of radiation and temozolomide may improve overall survival. Neutropenia and myelosuppression are rare with temozolomide alone; however, the risk doubles with the addition of bevacizumab. If thrombocytopenia occurs, platelet transfusions are recommended for platelet counts less than 10,000 or less than 20,000 if the patient is febrile, has an active hemorrhage, or is demonstrating signs of sepsis.
Bevacizumab is a human-derived monoclonal antibody against VEGF that has shown improvement in 6-month survival in previous trials. It is commonly used in treatment of recurrent GBM. It is known that high-grade gliomas produce large quantities of VEGF which promotes endothelial proliferation, and overexpression of this growth factor is directly proportional to blood vessel density, grade of tumor, and patient outcome. VEGF promotes angiogenesis; however, these vessels are often disorganized, which causes an increase in leakiness of the BBB. Bevacizumab functions to help the vessels function more normally, which in turn improves oxygenation and drug delivery. An intact BBB decreases vasogenic edema, which can improve symptoms by reducing mass effect. Although the effectiveness of bevacizumab is apparent, negative side effects can occur. It has been shown to increase the risk of cancer-associated hypercoagulability while also increasing the risk of hemorrhage. Other less common side effects include posterior reversible encephalopathy syndrome, GI perforation, and impaired wound healing.
Chemotherapy intervention for metastatic disease depends on the type of cancer. A recent case report showed 25 months of disease-free progression in a patient with intracranial non–small cell lung cancer (NSCLC) with crizotinib then ceritinib. Targeted therapy against the ALK oncogene has been proven effective in patients with NSCLC.
Radiation Therapy
Whole brain radiation therapy (WBRT) is becoming less favorable in managing metastatic disease due to the concern of neurocognitive toxicity. In patients that are candidates for WBRT with extensive intracranial metastatic disease, the standard doses are 3000 cGy in 10 daily fractions or 2000 cGy in 5 daily fractions. The addition of radiosensitizers does not improve overall survival when compared with WBRT alone. In a randomized control trial of patients with metastatic NSCLC, patients who received WBRT and optimum supportive care, defined as dexamethasone and a proton pump inhibitor for GI prophylaxis, had no statistically significant improvement in quality-adjusted life years or median survival when compared with those who received optimum supportive care alone. Stereotactic radiosurgery (SRS) alone is the treatment of choice in patients with one to three brain metastases. In younger patients with limited extracranial disease and four or more intracranial disease, there have also been positive results. SRS has been shown to have local control rates at 1 year of approximately 90% with minimal toxicity in patients with prior surgery or with SRS alone. Radionecrosis rates are approximately 20% after initial SRS. In 2014 the American Society for Radiation Oncology (ASTRO) released the following statement to help clinicians choose appropriate radiation techniques in patients with limited metastatic disease:
Don’t routinely add adjuvant whole brain radiation therapy to stereotactic radiosurgery for limited brain metastases. Randomized studies have demonstrated no overall survival benefit from the addition of adjuvant whole brain radiation therapy (WBRT) to stereotactic radiosurgery (SRS) in the management of selected patients with good performance status and brain metastases from solid tumors. The addition of WBRT to SRS is associated with diminished cognitive function and worse patient-reported fatigue and QoL. These results are consistent with the worsened, self-reported cognitive function and diminished verbal skills observed in randomized studies of prophylactic cranial irradiation for small cell or non–small cell lung cancer. Patients treated with radiosurgery for brain metastases can develop metastases elsewhere in the brain. Careful surveillance and the judicious use of salvage therapy at the time of brain relapse allow appropriate patients to enjoy the highest QoL without a detriment in overall survival. Patients should discuss these options with their radiation oncologist. Tsao.
Discussions between healthcare providers and the patients regarding prognosis and goals of therapy should be a routine part of the communication prior to initiating any intervention.
Surgical Resection
Surgical resection of brain tumors may also be considered for patients that have significant disease burden and to minimize symptoms. Several randomized trials have recommended the use of surgery for single brain metastatic disease to increase survival. Gamma knife radiosurgery (GKRS) is a good treatment option for small meningiomas, and a study shows that 97.9% showed 5-year tumor control. A recent study showed that the 5-year recurrence rate of grade I meningiomas to be 10% after complete resection and as high as 45% with subtotal resection. RT and GKRS are good for preventing recurrence in lower and higher grade meningiomas. However, further surgery could not be reliably recommended due to high rates of recurrence and complication rates. In a study of patients with stage II and III ependymomas, adult patients with a gross total resection saw a statistically significant increase in progression-free survival as compared with those with subtotal resection. This finding was independent of those who received postoperative adjuvant RT.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree