Anorexia/Weight Loss
Toby C. Campbell
Jamie H. von Roenn
Cancer cachexia is a complex metabolic process, due to both host and tumor factors, which results in excess catabolism as well as aberrant fat and carbohydrate metabolism. The syndrome is most commonly seen in patients with advanced cancer and characterized clinically by unintentional weight loss, muscle wasting, anorexia, fatigue, impaired performance status, anemia, and edema (1, 2, 3, 4). The incidence of weight loss varies with both the stage and primary tumor site. Tumors of the upper gastrointestinal tract and lung are most frequently associated with weight loss, while it is an uncommon problem for patients with advanced breast cancer (2). Treatment-related symptoms contribute to decreased oral intake but do not cause cachexia. Cachexia is distinguished from starvation (decreased energy intake) by the disproportionate loss of skeletal muscle over adipose tissue and its recognized lack of response to nutritional supplementation (1). The diagnosis of cachexia should be entertained in any patient with involuntary weight loss and muscle wasting.
Multiple clinical investigations have demonstrated the impact of weight loss on prognosis. Involuntary weight loss is associated with decreased overall survival (2, 5, 6, 7, and decreased response to, and symptomatic benefit from, chemotherapy (8, 9). Even in patients with a good performance status, as little as 5% loss in body weight adversely affects prognosis. For many patients, decreasing weight correlates with declines in performance status. The relationship between muscle wasting and mortality has been well documented. Loss of approximately 30% of total body weight or loss of 20% of lean body mass is incompatible with survival (1). In one autopsy series, up to 20% of patients with cancer had no discernible immediate cause of death other than “cachexia” (10).
In this chapter, we will explore the current knowledge of the pathophysiology of the cachexia/anorexia syndrome, discuss its evaluation, and outline pharmacologic and nonpharmacologic treatment options.
Pathophysiology
Inflammation
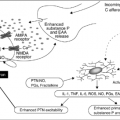
The mechanisms underlying cancer-related weight loss are incompletely understood but reflect a complex interplay between symptoms that impact nutritional intake, tumor-related factors, metabolic derangements, and hormone dysregulation. The negative nitrogen balance in patients with cancer cachexia results from an imbalance between protein synthesis (anabolism) and protein degradation (catabolism). The disproportional loss of muscle mass is mediated, at least in part, by proinflammatory cytokines. Cancer cachexia is associated with a chronic inflammatory response characterized by an increase in acute phase proteins and proinflammatory cytokines (11, 12, 13). Elevated levels of proinflammatory cytokines, including interleukin-6 (IL-6), interleukin-1 (IL-1), interferon-γ (IFN-γ), and tumor necrosis factor α (TNF-α) are present in many patients with cancer and their levels seem to correlate with tumor progression. IL-6 levels in newly diagnosed patients with lung cancer are related to the degree of weight loss (14). In patients with pancreatic cancer who have been losing weight, high concentrations of TNF-α and IL-6 in peripheral blood mononuclear cells, increased plasma concentrations of a soluble TNF receptor, and other adhesion molecules have been observed (15). The wasting effect of these proinflammatory cytokines seems to be targeted against the myosin heavy chain of skeletal muscle (16). Similarly, in patients with advanced colon cancer, elevated levels of TNF-α and IL-6 have been associated with anorexia (17). In patients with advanced pancreatic cancer, the presence of an acute phase response is associated with accelerated weight loss, hypermetabolism, anorexia, and shortened survival (15). It has been hypothesized that the increased demand for amino acids for the synthesis of acute phase proteins is met at the expense of skeletal muscle (18). Amidst this cytokine “storm,” patients may experience nonspecific signs and symptoms, including depression, anxiety, cognitive impairment, fatigue, and pain (19).
The ubiquitin-proteosome pathway, a regulatory gateway for protein synthesis and degradation in skeletal muscle, may be the final common pathway for cancer-related wasting (20). This pathway, regulated at least in part by proinflammatory cytokines, is downregulated by anticytokine treatment in animal models. The ubiquitin-proteosome proteolytic pathway is responsible for the bulk of basal proteolysis in skeletal muscle and as much as 80% of the muscle degradation in cancer cachexia (21).
Tumor Factors
The excessive catabolism which characterizes cancer cachexia is mediated not only by host factors but also by tumor-derived factors. Proteolysis-inducing factor (PIF) is a tumor-derived gly-coprotein which induces protein degradation in skeletal muscle by upregulation of the ubiquitin-proteosome proteolytic system (22). Preliminary data suggests that PIF can be detected in the urine of 80% of patients with unresectable pancreatic cancer (23). The identification of urinary PIF is associated with
a greater degree of weight loss compared with those without urinary PIF, 12.5 kg versus 4.5 kg, respectively. Elevated PIF has not been observed in all tumor types in the presence of wasting. The underlying mechanisms of wasting likely vary by tumor type and potentially by other, not yet defined, factors.
a greater degree of weight loss compared with those without urinary PIF, 12.5 kg versus 4.5 kg, respectively. Elevated PIF has not been observed in all tumor types in the presence of wasting. The underlying mechanisms of wasting likely vary by tumor type and potentially by other, not yet defined, factors.
Energy Dysregulation
Elevated resting energy expenditure (REE) may contribute to wasting in some patients with cancer. REE has been measured in patients with a variety of cancers with variable results; cancers of the lung and pancreas are most frequently associated with an increase in REE (24). Wasting in the setting of other tumor types is not predictably associated with alterations in REE (24). Hypermetabolism, when present, may occur secondary to tumor growth, increase in the Cori cycle, increase in acute phase protein response, gluconeogenesis, and increased protein turnover.
Dysregulation of the control of energy intake also contributes to the anorexia-cachexia syndrome. There are four neurotransmitters active in the appetite center of the hypothalamus. Neuropeptide Y and Agouti-related peptide (AGRP) are appetite-stimulating neurotransmitters, while opio-melanocortin and the cocaine-amphetamine-related factor (CART) inhibit appetite (25). These neuropeptides regulate the balance between ghrelin, a hormone produced in the stomach that stimulates appetite, and leptin, an anorexigenic hormone produced in fatty tissue (26). Proinflammatory cytokines may stimulate pathologic alterations in the central control of energy intake. Neuropeptide Y–induced feeding is antagonized by IL-1 in rats (27). In addition, IL-1 upregulates central corticotropin-releasing factor, a potent anorexigenic stimulus (28). Proinflammatory cytokines stimulate the expression of leptin and/or mimic the negative feedback from leptin, resulting in long-term inhibition of energy intake (25).
Our understanding of the intracellular mechanisms involved in muscle homeostasis and the impact of host- and tumor-derived factors on cancer cachexia continues to expand. There remain, however, numerous unanswered questions. Although our therapeutic approach once focused entirely on the treatment of nutrition impact symptoms and anorexia, we now recognize the need to consider anabolic as well as anticatabolic interventions.
Evaluation
The initial evaluation of cancer-associated weight loss includes a detailed history and physical examination to determine the rate and extent of weight loss and to identify treatable, easily reversible causes of weight loss. The evaluation includes a precise measurement of body weight, height, and documentation of premorbid body weight, rate of weight loss, assessment of appetite, review of current medications, and presence of nutrition impact symptoms (dysphagia, nausea, constipation, depression, pain, infection, or altered taste). Both the amount of weight loss and its rate define severity. Weight loss of 2% in 1 week is as significant clinically as weight loss of 5% over 1 month (Table 11.1).
Table 11.1 Definition of Significant Weight Loss | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Physical examination is directed at identifying signs of nutrition impact symptoms (e.g., mucositis), skeletal muscle wasting, strength, and mobility. In addition to standard measurements of weight, evaluation of body composition is ideal, as lean body mass is the metabolically active component of body mass. Anthropometric assessments, including midarm circumference using a tape measure and triceps skinfolds using calipers, are useful indicators of change in arm muscle mass. Although anthropomorphic measurements are easy, readily completed, and a potentially useful measure of change, there is variation in results when measurements are taken by different clinicians.
Bioelectric impedance analysis (BIA) is another simple bedside technique to monitor changes in body composition. BIA is based on the differential resistance to a low-intensity electrical current by the fat and lean tissue body compartments. Measured values of reactance and resistance obtained by BIA can be used in regression equations to estimate fat and lean tissue and total body water. The BIA equipment is relatively inexpensive and easy to use. A measurement takes only approximately 10 minutes and involves no discomfort for the patient. Accurate measurements of weight and height and consistent positioning of the electrodes are essential to obtain accurate body composition data with BIA. The BIA technique is most useful for monitoring change.
More accurate measurement techniques for the assessment of body composition include underwater weighing, whole body counting of potassium, nitrogen, and other elements, dual energy x-ray absorptiometry, and estimation of total body water and extracellular water by dilution techniques. These methods are only reasonable considerations in the research setting.
Standardized, validated nutrition assessment tools are available but not routinely utilized. The Patient-Generated Subjective Global Assessment (PG-SGA) of Nutritional Status is a screening tool that can be utilized to identify patients in need of intervention. This tool is accepted as the standard for the nutritional assessment of patients with cancer by the American Dietetic Association. The assessment includes a patient report of weight and weight change, food intake, symptoms, activities, function, and a physician evaluation of the disease and its related nutritional requirements.
Body weight is objective and simple to measure, but it is only one, late manifestation of the cancer cachexia syndrome. Early in the trajectory of disease and/or for research, more objective tools for analysis, including measurement of lean body mass, strength, energy expenditure, and biochemical assays of inflammatory mediators, are important concerns. At the bedside, nutritional interventions must fit a particular patient’s goals of care. Dahele and Fearon advocate focus on patient-centered outcomes, such as quality of life, performance status, and activity level (1). Although a patient with end-stage cancer may not gain weight with an appetite stimulant, it may effectively relieve anorexia and allow the social pleasure of eating dinner with family and friends. On the other hand, a patient with a new diagnosis of cancer and wasting might be best served by a combined anticatabolic and anabolic approach. Recommendations for treatment of weight loss and anorexia must be anchored by a discussion of the goals of care.
Treatment
The first step in the management of cancer-related weight loss is identification and treatment of nutrition impact symptoms.
Table 11.2 lists common, potentially treatable, nutrition impact symptoms. Unfortunately, for most of the patients with involuntary weight loss and muscle wasting, treatment of these symptoms does not reverse the progressive cachexia. The impact of aggressive symptom control on oral intake and, more specifically, wasting may improve sense of well-being but is unlikely to adequately treat the wasting.
Table 11.2 lists common, potentially treatable, nutrition impact symptoms. Unfortunately, for most of the patients with involuntary weight loss and muscle wasting, treatment of these symptoms does not reverse the progressive cachexia. The impact of aggressive symptom control on oral intake and, more specifically, wasting may improve sense of well-being but is unlikely to adequately treat the wasting.
Table 11.2 Nutrition Impact Symptoms | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Role of Caloric Supplementation
Decreased energy intake and anorexia contribute to weight loss. Intense caloric repletion, at first glance, seems a rational intervention for weight-losing patients with cancer, and many patients and families focus their energies on increasing intake. However, in advanced patients with cancer, the striking difference between starvation, an absolute lack of calories, and cachexia highlights the inadequacy of caloric supplementation as a primary treatment for cachexia (see Table 11.3). In starvation, adaptations lead to decreased energy expenditure, relative preservation of muscle mass, and decreased proportional loss of muscle and visceral mass, with continued caloric restriction. In the setting of cancer cachexia, energy expenditure may remain elevated in spite of inadequate energy intake and continued loss of skeletal muscle mass. The lack of benefit from caloric supplementation for advanced wasting is true whether the calories are delivered enterally or parenterally.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree