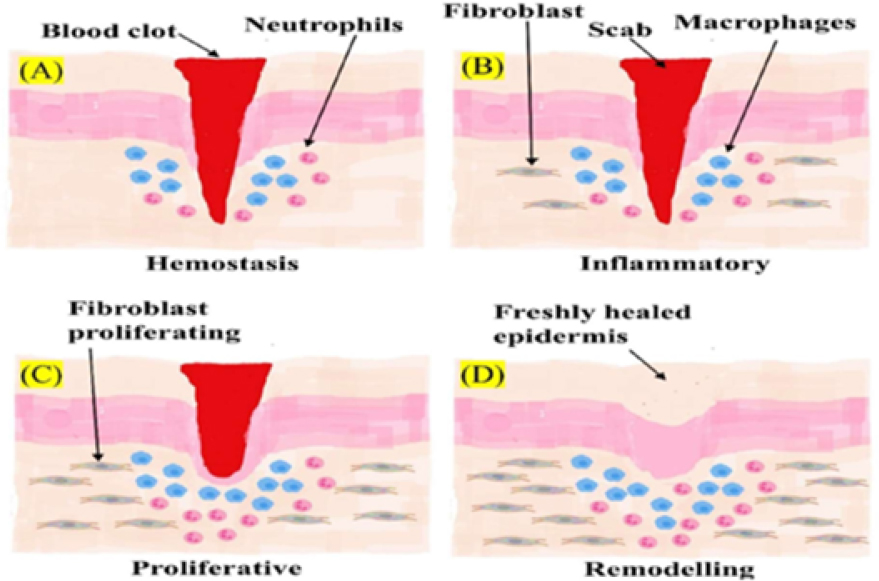
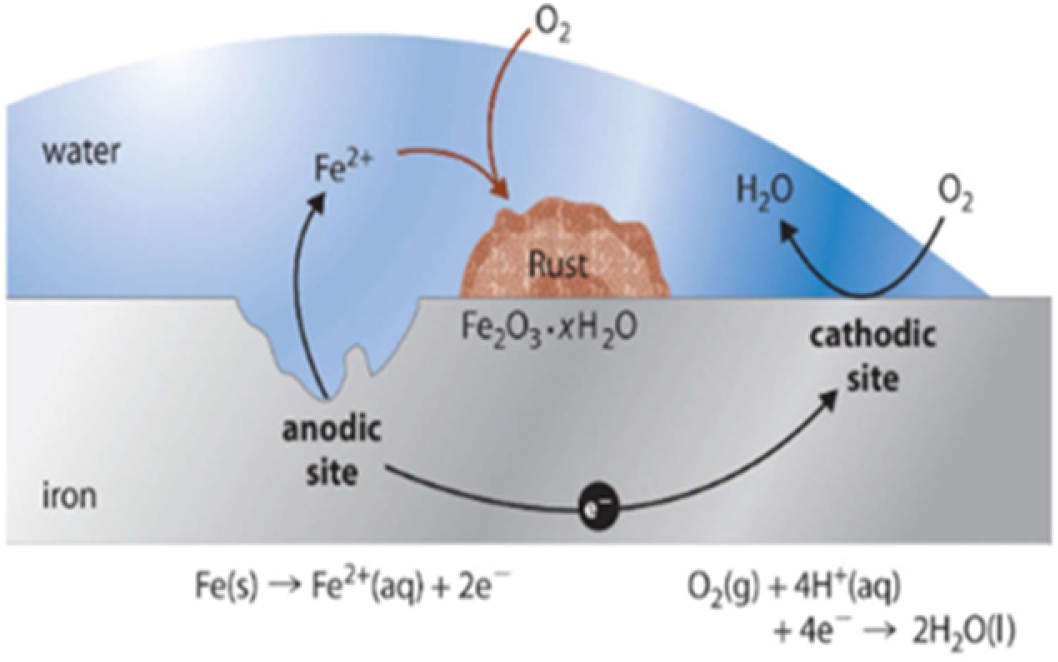
Biomaterials have revolutionized modern medicine and rehabilitation, especially within physiotherapy, by supporting tissue regeneration, replacing damaged structures and enhancing therapeutic outcomes. This chapter explores the intricate interactions between biomaterials and biological tissues, beginning with protein adsorption and the wound healing cascade, following implantation. It emphasizes the importance of biocompatibility and the tissue-specific integration of implants, highlighting the critical influence of surface properties and material composition. This chapter also discusses the significant role of physiotherapists in adapting rehabilitation protocols to account for implant-tissue dynamics, with special attention to movement, functionality and potential complications. Particular focus is placed on metallic implants, especially their susceptibility to corrosion and how degradation can affect physiotherapeutic strategies. Corrosion can alter mechanical stability, release toxic by-products and provoke inflammatory responses – necessitating cautious load-bearing exercises and close monitoring. This chapter further delves into biodegradable and resorbable materials, noting their advantages and challenges in dynamic healing environments. The integration of artificial intelligence (AI) into rehabilitation is presented as a transformative tool. AI algorithms aid in detecting implant degradation, analyzing sensor data, medical imaging and biomarkers to enable early interventions and personalized therapy planning. These advancements offer real-time monitoring and predictive analytics, ensuring safer, more adaptive rehabilitation. Ultimately, this chapter bridges the gap between biomedical engineering and clinical physiotherapy practice, underscoring the need for collaborative, personalized and technology-integrated approaches to optimize patient recovery and implant performance. The implantation of a biomaterial often creates a wound, and bleeding generally ensues. Blood thus typically makes a first contact with the implanted biomaterial. Proteins are building blocks of the supporting extracellular matrix (ECM) of many tissues. Changes in the levels of proteins or the structure of proteins can lead to altered function and are responsible for many diseases. This paved the way for blood screening for certain proteins indicating a diseased or cancer state. Thus, protein plays a major role in determining the final nature of the tissue–implant interface. Biomaterials can promote cell/tissue attachment and activity by allowing selective protein absorption or can inhibit tissue interactions by repelling protein. pH and ionic strength of the biomaterial can alter confirmation of nearby proteins and its functions. In fact, proteins also experience the structural alterations during interaction with the solid surface of biomaterials and lose some of their activity as the functions get altered (Enderle and Bronzino 2012). A special protein known as fibrinogen has greater affinity for the biomaterial surface. Disruption of the anatomic continuity of the tissue occurs after implantation, which creates a wound. There are four overlapping phases of wound healing: Figure 6.1 represents the stages (Palani et al. 2024). Figure 6.1. Wound healing response after biomaterial implantation Recent advances in wound dressing and the material and skin interaction are vital for in-depth knowledge about skin–material interaction. Metal nanoparticles and biomaterials have recently started playing a pivotal role in encouraging effective wound healing and averting secondary infections or any concern regarding immunogenicity. The tiny size of the healing materials, wound dressing or even the materials inserted topographically onto skin or in vitro have a high surface area, which facilitates enhanced biological interaction. Thus, the materials need to be specifically designed with respect to topography, size, material and biocompatibility. The material used for the therapy/assessment should have an optimal cell-to-cell interaction, fostering an effective purpose (Palani et al. 2024). Chapter 5 emphasized the type of biomaterial used in the current medical scenario. The targeted approach of this chapter is to emphasize the problems of interaction of the biomaterials commonly encountered in the medical field, especially in physiotherapy subjects. The biomaterials are specially designed to mimic the ECM and the tissue topographically and hence are biocompatible. The type of materials studied in recent evidence are nano-metallic particles and biodegradable materials. The metallic substances are generally known to have a major disadvantage; this is metallic corrosion. Tissue biomaterial interactions can significantly affect physiotherapy, influencing both the rehabilitation process and the outcomes of treatment. The way a biomaterial interacts with the surrounding tissue – whether it is bone, cartilage, muscle or skin – can determine the success of the implant and the effectiveness of physiotherapy interventions (Pramanik 2024). In summary, tissue biomaterial interactions play a significant role in determining how effective physiotherapy will be during rehabilitation. Physiotherapists must continuously assess and adapt their approaches based on the type of biomaterial used, the patient’s healing progress and the potential challenges presented by tissue–implant interactions. Several mechanisms can lead to metallic corrosion; thus, its property, which is corrosive resistance, is one of the important properties of metals used for implants. There are many types of corrosion (Manam et al. 2017): Figure 6.2. Metallic corrosions Corrosion resistance is a critical property of metals used for implants, as it directly impacts the longevity and functionality of the implant, which in turn affects the physiotherapy regimen. When metallic implants corrode, they can release harmful ions into the body, leading to tissue damage, inflammation, infection and sometimes implant failure. Several mechanisms contribute to metallic corrosion, such as electrochemical reactions, mechanical stress and the presence of body fluids, including sweat, blood and lymph. These reactions can weaken the mechanical properties of the implant over time, affecting its stability and performance (Reddy and Srinivasan 2018). The corrosion resistance of an implant is essential in ensuring the durability of the metal and maintaining its biocompatibility. If an implant is prone to corrosion, it may degrade over time, leading to complications such as implant loosening, infection and systemic toxicity. In these cases, the physiotherapy regimen must be adapted to take into account the potential for pain, swelling and compromised healing (Naeimirad et al. 2023). For example, if an implant shows signs of corrosion and becomes unstable, the physiotherapist may need to adjust the intensity and type of physical activity to prevent further damage or overloading of the compromised area. Corrosion can also affect the tissue–implant interface, potentially leading to implant migration or inflammation. As a result, physiotherapists may need to monitor the patient more closely and adjust their therapy plans to accommodate limited range of motion, muscle weakness or joint instability associated with implant degradation. Strengthening exercises may be altered to avoid putting excessive stress on the implant, and low-impact or protective techniques might be used to minimize the risk of injury or further degradation of the metal (Naeimirad et al. 2023). Overall, the corrosion resistance of metallic implants is vital for ensuring stable and safe rehabilitation. Implants with low corrosion resistance may require more frequent monitoring, adjustments to the physiotherapy plan and sometimes even earlier replacement, which can impact the patient’s rehabilitation timeline and recovery outcomes. Hence, physiotherapists must be aware of the material properties of the implant and adapt their approach accordingly to ensure that the recovery process is not hindered by potential complications from corrosion. Biomaterials can be permanent within the body or can be degradable. The degradation process may be chemically driven or accomplished by cells. The bioresorbable materials are known to degrade themselves in body over time and be replaced with natural tissue. The goal is to meet strength and cell support while the tissue or cell regenerates (Dewey and Harley 2022). Biomaterial degradation may lead to chronic non-healing wounds. This may happen if biomaterial degrades too quickly and releases particulate matter, which extends the inflammation stage. If the healing passes through to the fibrous capsule formation stage, there may still be complications (Dewey and Harley 2022). Figure 6.3. Biomaterial degradation and resorption Farraro et al. (2014) conducted an in vitro (goats as subjects of the experiment) study on the potential magnesium-based materials to understand its biodegradable ability and bio-absorbable feature. The authors used magnesium-based orthopedic devices such as interference screw to accelerate ACL graft healing and a ring to aid in the healing of an injured ACL. The authors studied their effect by using a robotic testing system and concluded that both devices could optimally restore the joint functions of the goat. Furthermore, the magnesium-based interference screw reported to non-toxic, even at the 12-week assessment (Farraro et al. 2014).
6
AI-driven Innovations in Biomaterial-based Physiotherapy and Oncology: Personalizing Patient Care Through Intelligent Monitoring and Predictive Rehabilitation
6.1. Interactions with blood and proteins
6.2. Wound healing response after biomaterial implantation
6.3. Effect of tissue biomaterial interaction on physiotherapy
6.4. Metallic corrosions
6.5. Effect of corrosion on physiotherapy regimen
6.6. Biomaterial degradation and resorption
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree