Fig. 5.1.
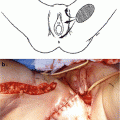
(a) Complete hydatidiform mole. All chorionic villi are enlarged and have abnormal shapes. There is marked hyperplasia of the trophoblast. Circumferential proliferation of the trophoblast around the villi is also noted, best seen in the upper left corner. Hematoxylin and eosin, original magnification 20×. (b) Partial hydatidiform mole. A few markedly enlarged chorionic villi containing central fluid-filled spaces (cisterns) are interspersed with smaller villi, creating a dimorphic population of villi. There is hyperplasia of the trophoblast, best seen in the upper right corner. Hematoxylin and eosin, original magnification 20×.
PSTT from intermediate trophoblast.
Hydatidiform moles: Varying degrees of trophoblastic proliferation.
Complete moles: Absence of fetus or embryo.
Trophoblast is hyperplastic.
10–17 % of hydatidiform moles become invasive mole: Myometrial invasion of hydatidiform mole via direct extension through tissue or venous channels [7].
15 % of invasive moles metastasize: Lung and vagina most common.
Clinically diagnosed with persistently elevated hCG levels.
CCA: Abnormal trophoblastic hyperplasia and anaplasia, absence of chorionic villi, hemorrhage, and necrosis [4] (Fig. 5.2).

Fig. 5.2.
Choriocarcinoma. The tumor is highly hemorrhagic. Necrosis is seen in the lower left corner. The tumor is composed of neoplastic cytotrophoblast (clear cells) and syncytiotrophoblast (purple-staining cells with multiple nuclei). Hematoxylin and eosin, original magnification 100×.
PSTT: From placental implantation site with mononuclear intermediate trophoblasts without chorionic villi.
More lymphatic metastasis.
Diffuse cytokeratin and human placental lactogen (HPL) staining [9] (Fig. 5.3).

Fig. 5.3.
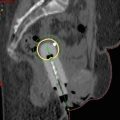
Placental site trophoblastic tumor. The tumor is composed of masses of atypical intermediate trophoblast (pale-staining cells) infiltrating between the bundles of myometrium (staining bright pink). Hematoxylin and eosin, original magnification 100×.
Matrix metalloproteinases (MMPs): Involved in metabolism of extracellular matrix, needed for invasion of maternal tissues [10].
Genetics
Partial moles typically are triploid in nature (69 XXX or 69 XXY) (Table 5.1) [8].
Feature
Partial mole
Complete mole
Karyotype
Triploid (90 %) 69XXX or 69XXY
Diploid; 46XX (90 %) or 46XY (10 %)
Usually fertilization of normal ovum by 2 sperm
Usually ovum without maternal chromosomes or inactive chromosomes and fertilized by 1 sperm that duplicates
Pathology
–
–
Fetus
Often present
Absent
Amnion, fetal red blood cells
Usually present
Absent
Villous edema
Variable, focal
Diffuse
Trophoblastic proliferation
Focal, slight to moderate
Diffuse, slight to severe
Clinical presentation
–
–
Diagnosis
Missed abortion
Molar gestation
Uterine size
Small for gestational age
50 % larger for gestational age
Theca lutein cysts
Rare
15–25 %
Medical complications
Rare
Less than 25 %
Postmolar malignant sequelae
<5 %
6–32 %
Normal ovum with two spermatozoa [13].
Complete moles are diploid with all chromosomes from paternal origin (46 XX or XY).
Recurrent molar pregnancies have biparental rather than androgenic complete moles [16].
Associated with a strong family history, with imprinting abnormalities noted that resemble fully paternal chromosomes [17].
Affected chromosome noted in familial cases: 19q13.3–13.4 with mutations in NLRP7 [18].
NLRP7 is a cytoplasmic protein, with members of the NLRP family associated with inflammatory processes and apoptosis [15].
Diagnosis
Symptoms
Complete hydatidiform moles:
Partial moles:
Present with incomplete or missed abortion 90 % of the time, followed by vaginal bleeding (75 %) [20].
Imaging
Ultrasound has replaced any other diagnostic tool to diagnose complete or partial moles [21].
Complete moles are characterized by a heterogenous mass (snowstorm pattern) without fetus.
Ultrasound can demonstrate cystic spaces in the placenta and vesicular pattern of multiple echoes [22].
Only 40–60 % of complete moles are detected as molar on ultrasound.
American Congress of Obstetrics and Gynecology (ACOG) recommends pathologic evaluation of tissue after spontaneous and therapeutic abortions to confirm diagnosis [23].
Presentation of gestational trophoblastic neoplasia is dependent on the antecedent pregnancy event, extent of disease, and histopathology [19].
Postmolar GTN presents with enlarged irregular uterus, persistent ovarian enlargement, and irregular bleeding [19].
GTN can be diagnosed after metastases are found, and these metastases can bleed.
HCG Levels in GTD
hCG is composed of an α- subunit, and β-subunit; the β-subunit is specific to the placenta.
hCG is hyperglycosylated in the first trimester of normal pregnancies; in GTD there are other subunits, such as β core, free β-hCG, nicked free β, or c-terminal peptide [24].
Hydatidiform moles have markedly elevated hCG levels above normal pregnancy.
Complete moles pre-evacuation have hCG levels >100,000 [25].
Less than 10 % of partial moles have pre-evacuation hCG levels >100,000.
Postmolar GTN is diagnosed with rising or plateauing hCG levels after evacuation. CCA is diagnosed with elevated hCG levels after other pregnancy event, likely associated with metastatic disease. PSTT only associated with slight elevation of hCG, but elevated human placental lactogen.
Postmolar GTN diagnosed by one of the following [26].
hCG level plateau of 4 values plus or minus 10 % recorded over a 3-week period (days 1, 7, 14, and 21).
hCG level increase of more than 20 % of 3 values recorded over a 2-week period (days 1, 7, and 10).
Persistence of detectable hCG for more than 6 months after molar evacuation.
Histopathologic diagnosis of CCA.
Presence of metastatic disease.
New pregnancy must be excluded prior to diagnosis.
Important to use an assay that can detect intact hCG molecules, but also H-hCG and H-freeβ, as well as degradation products.
Different assays can have up to 58-fold variation of hCG results [27].
Diagnosis of GTD confirmed by cervical dilation and suction curettage; [28]. GOG 242 (results discussed below) illustrated utility of second D&C as a curative measure.
Persistently Low Levels of hCG
Multiple conditions can cause low levels of hCG.
Pregnancy, GTD, false positive or phantom hCG, other malignancies, pituitary hCG.
High proportion of women with persistent hCG, without findings of pregnancy or GTD, can go on to unnecessary chemotherapy or hysterectomy [29].
Guidelines recommended by the USA hCG reference service for persistent hCG values include ruling out pregnancy, determining if hCG values are real, and determining if active GTN, PSTT, or non-trophoblastic malignancy is present.
Serum should be sent to special laboratories to check for H-hCG and free βhCG [29].
Phantom or false hCG values can be present in 3–4 % of healthy women [30].
Caused by cross-reactivity with heterophilic antibodies in the serum.
Product of “sandwich” assays that measure mixtures of hCG.
Not present in urine hCG tests; and thus negative urine hCG can be used to confirm false positive [31].
Serum can be diluted serially and run through assays to check for false positives (dilution does not affect false titers), or sent for additional tests (see above) [32].
No treatment is required for false positive hCG results [32].
Diagnostic Evaluation
When GTN diagnosed or greatly suspected, patient should undergo thorough evaluation for assessment of extent of disease prior to initiation of therapy.
Blood work: Hepatic function, renal function, baseline serum hCG, blood count (CBC), thyroid function tests, blood type.
Pelvic imaging: By ultrasound or CT to assess for residual disease within uterus, evidence of pelvic spread.
Chest imaging: Can be performed via Chest X-ray or CT scan. While there is a higher sensitivity for detection of pulmonary metastasis with Chest CT (as high as 40 % in patients with negative Chest X-ray) compared to Chest X-ray, mandatory Chest CT is not necessary if detection of occult pulmonary metastasis does not change treatment protocol [33].
Patients who are asymptomatic with normal pelvic and chest imaging do not require any additional radiologic imaging [23].
In all patients with choriocarcinoma or in those patients with vaginal or lung metastasis, Brain MRI should be obtained.
If lesion in vagina suggestive of GTN present, biopsy is not recommended due to severe bleeding risk [34].
Staging
FIGO Committee on Gynecologic Oncology in 2000 set forth a staging and classification system for Gestational Trophoblastic Neoplasia [35].
The staging system is used in combination with the World Health Organization risk-factor scoring system for GTN [36] (see Tables 5.2 and 5.3).
FIGO stage
Stage I
Disease confined to the uterus
Stage II
GTN extending outside of the uterus but limited to genital structures (adnexa, vagina, broad ligament)
Stage III
GTN extending to the lungs with or without genital tract involvement
Stage IV
GTN involving all other metastatic sites
Score
Risk factor
0
1
2
4
Age
<40
≥40
Antecedent pregnancy
Mole
Abortion
Term
Pregnancy event to treatment interval (months)
< 4
4–6
7–12
>12
Pretreatment hCG (mIU/mL)
< 103
103–<104
104–<105
≥105
Largest tumor size (including uterus)
–
3–<5 cm
≥5 cm
–
Site of metastases
Lung
Spleen, kidney
Gastrointestinal
Liver, brain
Number of metastases
–
1–4
5–8
≥8
Previous failed chemotherapy
–
–
Single drug
2 or more drugs
The FIGO staging system is anatomically based.
Diagnosis should be presented with Stage (denoted by roman numerals I–IV), followed by the sum of actual risk factors (in Arabic numerals) separated by a colon. For example, II: 4 [35].
Non metastatic and low risk metastatic GTN (Stage I and Stages II–III with scores <7) can be treated with single agent chemotherapy (see Sect. 6) [4].
Treatment
Pretreatment workup includes: CBC including platelet count, clotting studies, renal and liver function tests, blood type with antibody screen, hCG level, and chest X-ray (pre-evacuation) [23].
Prior to suction evacuation and curettage, include possible cross match of 2 units.
Suction evacuation and curettage is standard of care for those who desire fertility; hysterectomy is appropriate for those who do not [37].
The largest cannula that can be introduced through the cervix should be utilized to facilitate rapid evacuation [23].
Intraoperative ultrasound can be used to decrease risk of uterine perforation.
Oxytocin IV should be used at the start of evacuation to increase uterine contractility and minimize blood loss [23].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree