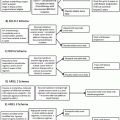
Panici trial
ASTEC trial
#Patients
514
1408
Eligibility
Clinical Stage I, >50 % myometrial invasion (excluded Stage IB grade 1), age <75, good performance status, no prior RT or chemotherapy
Disease clinically confined to corpus
Randomization
Standard treatmenta with systematic pelvic lymphadenectomy versus Standard treatment and no lymphadenectomy (control)
Hysterectomy, BSO, pelvic node dissection, palpation of para aortic nodes, resection of suspicious nodes at surgeons discretion versus
Hysterectomy, BSO, palpation of nodes with removal of suspicious nodes at surgeons discretion (control)
Aortic node dissection required
No
No
Median follow-up
49 months
37 months
Standardized adjuvant therapy
None
Yes-randomized specific cohort of intermediate and high risk, early stage disease defined as Stage IA or B grade 3, pap serous or clear cell and IC or IIA, also included those with positive pelvic nodes. Cohort randomized to external beam RT versus no external beamb
Both of these trials failed to demonstrate either an overall survival or recurrence free survival benefit for pelvic lymphadenectomy.
Although the investigators should be commended on their efforts to complete a randomized trial assessing the benefits of lymphadenectomy, many questions remain regarding the outcomes and methodologies for both of these trials.
Both included a preponderance of low risk patients, which makes identifying a small difference in survival challenging.
In the Panici study, there were two significant concerns:
Specifically the lack of para aortic node assessment and the lack of standardization for adjuvant therapy.
Approximately 22 % of the patients in the no lymphadenectomy arm actually had node sampling or lymphadenectomy.
In fact, 7 % had more than 20 nodes excised which was considered the “standard” in the lymphadenectomy arm as an acceptable per protocol node count.
With respect to the ASTEC trial, Creasman et al. appropriately summarized some of the confounding factors that “muddy” the investigators conclusions [31].
These include inequitable distribution of high risk factors in the lymphadenectomy arm including 3 % more grade 3 lesions, higher rate of lymphovascular space invasion and more deeply invasive disease.
In this trial 5 % of those randomized to the no lymphadenectomy group had nodes removed of which 30 % were positive.
Also 8 % of those randomized to lymphadenectomy had no nodes removed and at least 30 % had less than 9 nodes excised which per GOG standards is considered inadequate.
More concerning was the second randomization to external beam RT or no RT for those with intermediate or high-risk disease including those with positive nodes. Per the randomization, the cohort of patients that might benefit from lymphadenectomy and adjuvant RT, i.e., those with positive nodes, could be excluded from adjuvant RT at time of second randomization. From a clinical perspective this raises many ethical questions. Although randomized, multi-institutional trials, the flaws in both of these trials make it challenging to modify what has been standard therapy for endometrial cancer.
Although it may be impossible to design the “perfect” trial to assess the efficacy or therapeutic benefit of lymphadenectomy, it is important to recognize the continued prognostic significance of lymphadenectomy in planning adjuvant therapy.
The GOG is currently developing a randomized trial to address the questions that still exist with respect to indications and potential benefits from lymphadenectomy.
Risk Assessment
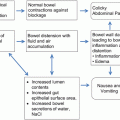
Following surgery, stage and other significant pathologic risk factors are utilized to determine risk for persistent disease or recurrence. This risk assessment is often utilized to define adjuvant therapy.
Low Risk: This is typically defined as endometrioid cancers confined to the endometrium, a subset of Stage IA, and grade 1 or 2.
Typically these patients are managed with close surveillance alone following surgery.
Intermediate Risk: This is typically defined as disease confined to the uterus, including the cervix (Stage II) with myometrial invasion (Stage IA or IB).
Other prognostic factors such as outer myometrial invasion, grade 2 or 3 and the presence of lymphovascular invasion can further subdivide this group into low or high intermediate risk.
The Gynecologic Oncology Group defines high intermediate risk based on a combination of age and pathologic factors including deep myometrial invasion, grade 2 or 3 and the presence of lymphovascular invasion [32].
High intermediate risk is thus defined by the following:
Any age with three risk factors.
Age 50–69 with two risk factors.
70 or older with one risk factor.
In contrast, the PORTEC (Post-operative Radiation Therapy in Endometrial Cancer) investigators define high intermediate risk as:
Age >60 with two risk factors including outer half myometrial invasion and
Grade 3 histology [33].
Management for intermediate risk category, specifically high intermediate risk remains controversial and will be discussed.
High Risk: This group consists of advanced stage disease, Stage III and uterine serous and clear cell carcinomas of any stage.
This category is associated with a high rate of recurrence and death from endometrial cancer. As such, adjuvant chemotherapy is often utilized post-operatively.
The role of RT is unclear but is currently being explored in a randomized trial thru the GOG (GOG 258).
In GOG 258 eligible patients include surgical Stage III and IVA endometrial cancer as well as early Stage (I and II) serous and clear cell carcinoma.
Patients are randomized to cisplatin on days 1 and 29 and volume directed radiation therapy followed then by 4 cycles of carboplatin and paclitaxel versus 6 cycles of carboplatin and paclitaxel.
Adjuvant Therapy
Intermediate “Low and High” Risk
Two randomized clinical trials would support observation rather than adjuvant radiation therapy for this group.
The Gynecologic Oncology Group study (GOG 99) was a phase III trial that randomly assigned 448 women with “intermediate-risk” endometrial cancer (defined as Stage IB, IC and occult II, excluding papillary serous and clear cell histologies) following comprehensive surgical staging to whole pelvic radiation versus observation [32].
The cumulative incidence of recurrence was higher in the no additional treatment group than the RT (radiation therapy) cohort 12 and 2 %, respectively.
The most common site of recurrence in the no RT group was the vagina.
There was; however, no statistically significant difference in overall survival between the two groups.
The estimated 4-year survival for no additional therapy versus RT was 86 and 92 %, respectively.
Importantly, the investigators identified a subset of patients with “high intermediate risk” that represented 1/3 of the entire cohort but two-thirds of all recurrences. In this population there was a significant benefit from radiation with respect to local recurrence.
This trial suggested redefining the intermediate risk group, specifically into a low and high risk and reserving radiation therapy for the high intermediate risk patients.
In comparison, PORTEC I assessed a relatively similar population of patients; however comprehensive surgical staging was not completed [33].
Similarly these investigators demonstrated a reduction in pelvic recurrences but no difference in overall survival.
Actuarial 5-year loco-regional recurrence rates were 4 % in the radiotherapy group and 14 % for those randomized to observation alone.
The overall survival rates were 81 and 85 %, respectively. As in GOG 99, the PORTEC investigators also identified a cohort of patients, based on age and grade 3 histology that accounted for the majority of recurrences.
This cohort, like the high intermediate risk subgroup in GOG 99, may actually benefit from adjuvant RT.
Table 4.2 summarizes both of these critical trials [34].
Trial
Eligibility
# Patients
Surgery
Randomization
Locoregional recurrence
Survival
PORTEC 1
Stage IB, grade 2–3
714
TAH-BSO
No therapy versus pelvic RT
14 % versus 4 % at 5 years (p < 0.001)
85 versus 81 % p = 0.31
Stage IC, grade 1–2
GOG 99
Stage IB and IC
392
TAH-BSO and comprehensive lymphadenectomy (pelvic and para-aortic
No therapy versus pelvic RT
12 % versus 2 % at 2 years (p < 0.01)
86 versus 92% p = 0.56
Occult Stage II
Many have hypothesized that the reason both GOG 99 and PORTEC I failed to demonstrate an improved survival with radiation was due to the preponderance of low risk patients that comprised these cohorts.
Although both trials failed to demonstrate a survival advantage for external beam radiation, each clearly identified a population of patients defined as “high intermediate risk” that had the most significant reduction in locoregional recurrence after external beam RT.
In summary, since the recurrence rates for low intermediate risk endometrial cancer is so low, and prognosis excellent, observation alone may be the most appropriate adjuvant “therapy.”
In comparison, for those with high-intermediate risk, recurrence rates range from 5 to 30 % with or without RT. As such, consideration for adjuvant radiation therapy is warranted.
Lastly the ASTEC and EN.5 pooled trial demonstrated similar findings in a relatively analogous population of patients [35].
This pooled trial raised another important question regarding the role of pelvic radiation in this population but more importantly the type of radiation, specifically vaginal brachytherapy.
These initially independent trials sought to assess the benefit of postoperative adjuvant radiation in early staged endometrial cancer with intermediate and high risk of recurrence.
Eligible patients include Stage IA and IB grade 3, all IC and all stages of papillary serous and clear cell carcinomas. A lymphadenectomy was not required. Eligible women were randomly assigned to observation or to radiation therapy.
Interestingly brachytherapy could be utilized in either arm. In fact 51 % of those randomized to observation alone, received brachytherapy.
Overall there was no difference in 5-year survival between the two groups. The rate of locoregional recurrence was 6.1 % in observation arm compared to 3.2 % in RT group.
This finding may certainly be due to the extensive use of vaginal brachytherapy in the observation arm.
This trial again demonstrated a lack of data to support the use of adjuvant external beam radiation therapy in these intermediate risk patients and high-risk patients with endometrial cancer. As with GOG 99 and PORTEC 1 this trial clearly demonstrated that radiation therapy has no overall effect on distant disease or overall survival. Interestingly, one might conclude from this trial that possibly brachytherapy alone could be used to reduce the risk of local recurrence and in fact may be the preferred strategy.
Lastly the role of adjuvant therapy for high intermediate risk patients recently assessed by a large randomized trial, GOG 249, which recently closed enrollment.
GOG 249 is a randomized trial of pelvic radiation versus vaginal cuff brachytherapy followed by paclitaxel/carboplatin in high risk, early stage endometrial cancer. Eligible patients include Stage I with high intermediate risk factors and Stage II as well as Stage I–II serous or clear cell carcinoma with negative cytology. As a review, high intermediate risk categories included patients with:
Age ≥70 years with one risk factor.
Age ≥50 with 2 risk factors.
Age ≥18 years with 3 risk factors.
Risk factors were defined as: Grade 2 or 3 tumors, (+) lymphovascular space invasion, and outer 1⁄2 myometrial invasion. Patients with these risk criteria could be enrolled with either positive or negative cytology.
Preliminary results presented at the 2014 SGO Annual Meeting indicated no difference in recurrence free survival at 24 months between the pelvic radiation arm and brachytherapy + chemotherapy arm.
OS data was not yet mature.
Vaginal Brachytherapy
The use of vaginal brachytherapy was addressed indirectly in the ASTEC/EN.5 trial and multiple retrospective studies.
In light of these data, the PORTEC investigators initiated a trial in high intermediate risk patients to assess whether vaginal brachytherapy was as effective as external beam [36].
The primary endpoint for this trial was vaginal recurrence.
This non-inferiority randomized trial was undertaken at 19 Dutch centers and enrolled 427 patients with high intermediate risk (age >60 and Stage IC grade 1 or 2, Stage IB, grade 3, Stage IIA, any age.
Patients were randomized to vaginal brachytherapy (VBT) and external beam RT (EBRT).
At median follow-up of 45 months, there were 3 recurrences in VBT group compared to 4 in the EBRT.
The estimated 5 year vaginal recurrence rates were 1.8 % for VBT and 1.6 % for external beam. Other important outcome measures when comparing VBT to external beam included:
Similar rate of distant metastasis, 8.3 % versus 5.7 %.
No difference in overall survival 84.8 % versus 79.6 %.
No difference in disease-free survival 82.7 versus 78.1 %.
Rates of GI toxicity lower in VBT compared to external beam 12.6 % versus 53 %.
Consequently, the PORTEC 2 investigators concluded that vaginal brachytherapy should be recommended for adjuvant therapy in those high-intermediate risk patients since it demonstrated good vaginal control with significantly less GI toxicities than external beam RT.
High Risk Disease
Women with high-risk endometrial cancer typically have a poor prognosis with surgery alone. As such, adjuvant therapy is typically recommended.
As mentioned previously, this group typically consists of advanced stage disease, Stage III, and any stage of papillary serous and clear cell carcinoma.
Currently there is not a “standard” approach for high-risk disease. Often adjuvant therapy is dictated by surgical and pathologic factors, i.e., uterine or extrauterine disease.
Since multiple questions remain, typically enrollment on a clinical trial may be the most appropriate option for patients in this risk category especially those patients with early stage serous or clear cell carcinoma.
Chemotherapy
For advanced stage endometrioid cancer, chemotherapy rather than radiation is now typically recommended.
One of the sentinel trials to support the use of adjuvant chemotherapy as compared to radiation therapy was GOG 122.
In this trial patients with Stage III or IV and <2 cm residual disease were randomized to whole abdominal RT versus chemotherapy (doxorubicin plus cisplatin) q 3 weeks for a total of 7 cycles.
Overall there was an improvement in PFS and OS in the chemotherapy arm.
Five-year survival was 53 and 42 %, respectively.
The chemotherapy arm did have a higher pelvic recurrence rate (18 vs. 13 %) and a higher proportion of GI, neurotoxicity and grade 3–4 hematologic toxicity of any type (88 % vs. 14 %) [37].
In a relatively similar Japanese trial, 385 patients with Stage I–III endometrial cancer were randomized to pelvic RT versus 3 cycles of cyclophosphamide, doxorubicin and cisplatin (CAP).
In this trial there was no difference in overall survival or progression free survival. Grade 3–4 toxicity was reported in 4.7 % those receiving chemotherapy versus 1.6 % in the radiation cohort [38].
The choice of chemotherapy has evolved over the past few decades. Many of the initial phase II and III trials in advanced and recurrent populations demonstrated that taxanes, anthracyclines and platinum compounds are the most active chemotherapeutic agents with response rates ranging from 20 to 35 %.
Table 4.3 summarizes the Gynecologic Oncology Group’s efforts to study chemotherapy in the advanced and recurrent setting.
Trial
Population
Regimens
RR
PFS
OS
“Winner”
GOG 107 [63]
Stage III/IV or recurrent
Dox versus Dox + Cis
25 % versus 42 %a
3.8 versus 5.7 monthsa
9.2 versus 9.0 months
Dox + Cis
GOG 163 [64]
Stage III/IV or recurrent
Dox + Cis versus Dox + Tax
40 % versus 43 %
7.2 versus 6.0 months
12.6 versus 13.6 months
Dox + Tax
Not superior
GOG 177 [65]
Stage III/IV or recurrent
Dox + Cis versus Dox + Cis + Tax (TAP)
34 % versus 57 %a
5.3 versus 8.3 monthsa
12.3 versus 15.3 monthsa
TAP but significant peripheral neuropathy
GOG 209
Stage III/IV or recurrent
TAP versus Carbo/Taxol
Awaiting results
The GOG has clearly studied in a progressive and rational fashion the role of chemotherapy for advanced and recurrent disease.
Although the triplet of paclitaxel, cisplatin and doxorubicin (TAP) demonstrated an improved PFS and overall survival, widespread acceptance was tempered by the significant peripheral neuropathy and treatment related deaths observed in this trial.
Recently, preliminary results from GOG 209 have been presented at the Annual Meeting of the Society of Gynecologic Oncology.
At time of presentation it would appear that the carboplatin and paclitaxel arm was not inferior to TAP.
As reported, there was a similar overall response rate (51 %), similar PFS (13 months for each arm) and overall survival.
The limited toxicity, familiarity, and preliminary findings from GOG 209 has led to an increasing use of this doublet for advanced or recurrent disease.
With respect to second line therapy, there remains a relative paucity of options. The GOG has conducted multiple phase II trials of single agent therapy with a limited number of objective responses.
Some of the agents studied include ifosfamide, topotecan, oxaliplatin, docetaxel, ixabepilone, bevacizumab, and pegylated liposomal doxorubicin [39].
Combined Therapy: Chemotherapy and Radiation
The role for combined therapy, radiation and chemotherapy, has yet to be clearly defined.
It has been hypothesized that an improved overall survival would be the theoretic benefit of combined therapy by improving both local and distal control. As such, this is currently being explored in three large randomized collaborative group trials:
GOG 249, PORTEC 3, and GOG 258. Although the populations being studied in these three trials are similar, there are some essential inclusion criteria differences meant to address different clinical questions.
GOG 249 has previously been discussed (with preliminary findings showing no difference in recurrence free survival between arms); however, the population of interest in this trial included women with Stage I disease with high intermediate risk characteristics and early stage papillary serous and clear cell carcinomas with negative cytology.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree