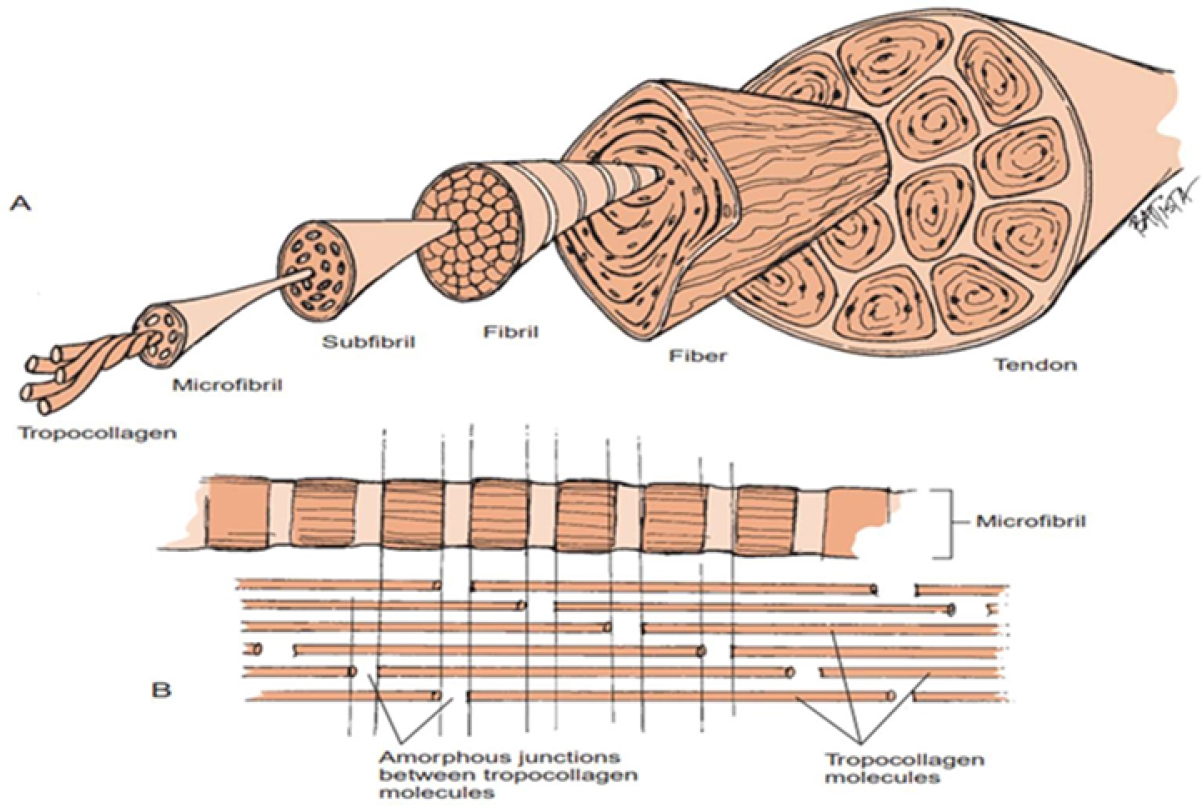
Physiotherapy plays a crucial role in rehabilitation, injury prevention and pain management, using evidence-based techniques to enhance mobility and overall well-being. This chapter explores modern advancements in physiotherapy, including manual therapy, electrotherapy, exercise therapy and innovative approaches such as virtual rehabilitation and AI-assisted techniques. Physiotherapists use patient-centered interventions to restore functional movement, reduce musculoskeletal discomfort and improve quality of life. The study highlights the importance of physiotherapy in managing chronic conditions such as arthritis, neurological disorders and post-operative recovery. By incorporating tailored rehabilitation programs, physiotherapy helps patients regain independence and functionality, ultimately reducing healthcare burdens. Additionally, the integration of technology in physiotherapy has significantly improved treatment outcomes by providing personalized rehabilitation programs. Wearable devices, motion-tracking sensors and AI-driven analytics enable precise assessment and real-time feedback, enhancing both clinical decision-making and patient adherence to treatment plans. Furthermore, this chapter emphasizes the significance of interdisciplinary collaboration in healthcare to optimize patient care. Physiotherapists work alongside physicians, occupational therapists and other healthcare professionals to develop holistic treatment strategies, ensuring comprehensive patient support. Through a review of recent literature and case studies, this study underlines the evolving role of physiotherapy in modern medicine. The findings suggest that continued research and technological integration will further enhance physiotherapeutic interventions, making them more accessible and effective in diverse clinical settings. As physiotherapy continues to evolve, its expanding scope and innovative methodologies promise to revolutionize rehabilitation and musculoskeletal healthcare, improving outcomes for a broad spectrum of patients worldwide. Tendons are essential components of the musculoskeletal system. Their main job is to transfer muscle forces to stiff bone levers, which in turn cause joint motion. Tendons can withstand 17 times body weight and are subject to significant compressive and tensile stresses, making them stronger than muscles. Their proprioceptive qualities aid in posture maintenance, and they serve as energy stores and shock absorbers (O’Brien 2005). A tendon acts as a “mechanical bridge”, transferring the forces generated by muscles to the bones and joints. Additionally, muscles use this strong, fibrous tissue to accomplish joint movements along a plane. The equivalent muscle reflects the morphology and purpose of the tendon. Tendon tissue is found not just at the terminals of muscles, but all the way down their length. The layers of connective tissue in muscles, known as the endomysium, perimysium and epimysium, combine to adhere to one or more fixed osseous sites. Contractile fibers can be found in tendon tissue near the muscle. Tendon activity is influenced by muscle, while muscle function is influenced by tendons (Bordoni et al. 2025). The purpose of the tendon is to facilitate the transmission of force that leads to joint motion while maintaining the ideal distance between the tendon and the joint. Tendons store and retrieve energy very efficiently because they function as springs. Conversely, ligaments serve to anchor bone to bone, which means they offer mechanical stability, enabling the joint to move within its normal range of motion under tensile loads and stop the joint from moving too much. The physiological characteristics of tendons and ligaments are similar, with a similar hierarchical structure and mechanical behavior, despite their different functions (Robi et al. 2013). Tendons are incredibly important for movement and mechanics. These anatomical structures promote movement and maintain proper posture by transmitting muscle forces to the skeletal levers. Tendons allow muscles to remain at the ideal distance from the joint they work on without requiring an excessive amount of muscle length to be present between the locations of origin and insertion. Tendons can bear heavy loads with little distortion, are more tensely strong than muscles and are stiffer than muscles. These characteristics reduce the amount of energy lost due to tendon strain and allow tendons to effectively transfer muscle forces to bones. Similar to other connective tissue structures, tendons are mostly formed of connective tissue, have only a small number of cells and a rich extracellular matrix, despite their often-complex structure (Kaya et al. 2019). Tendons are made of connective tissue. The body’s connective tissues act reflexively to join cells and organs together, giving the body support and shape. The three main categories of connective tissues are specialized connective tissues, supportive connective tissues and connective tissue proper. Cartilage and bone serve as supporting connective tissues. Adipose tissue and hematopoietic tissue are examples of specialized connective tissues. Dense or loose describes connective tissue proper. The “packing material” found internal and amid muscle sheaths, in supportive to epithelial tissue, and surrounding neurovascular bundles is a loose connective tissue (areolar connective tissue). Because it is so thin, the loose connective tissue is not very resilient to stress or pressure. The dense connective tissue is more resilient to stress and less flexible. Tendons are categorized as dense and regular connective tissue. In tendons and ligaments, fiber bundles are closely spaced, parallel to one another, and regularly subjected to forces. Their configuration especially suits them to withstand tensile or traction forces (Oatis 2009). Tendons, like all other thick connective tissues, are primarily made up of two components: an extracellular matrix and cells. When it comes to actively producing proteins, the fibrocyte (fibroblast) is the main cell type found in tendons. However, only approximately 20% of the bulk of the tissue is made up of cells. The components of the extracellular matrix, that of about the remaining 80%, are produced and secreted by fibroblasts. The ground substance and fibers (elastin and collagen) make up the extracellular matrix. The gelatinous substance that fills the voids left by cells and fibers is known as the ground substance. Water, proteoglycans (decorin, biglycan) and non-collagenous structural glycoproteins (fibronectin) make up its composition. Collagen makes up the majority of the fibrous component of tendons, which is why tendons seem white. A triple helix known as procollagen is formed when three polypeptide chains join together (Oatis 2009). The fibroblast secretes procollagen, which is an organic crystal, into the extracellular matrix (ECM). After the ends of the molecule are broken, the somewhat shorter molecule is now known as tropocollagen. In the extracellular space, tropocollagen molecules polymerize to form collagen microfibrils, which then group together to form fibrils, sub-fibrils and fibers. A far smaller percentage of the fibrous structure of tendons is made up of elastin fibers. Collagen makes up a far larger percentage of tendons than elastin does. An entire extracellular matrix consisting of water, proteoglycans and structural glycoproteins is known as the ground material. Structural glycoproteins are primarily composed of proteins with a minor amount of carbohydrates. These glycoproteins, which include undulin, fibronectin, thrombospondin and tenascin C, are crucial for a cell’s ability to adhere to fibers and the remaining ECM constituents. Proteoglycans are essential for tendon function, even though they make up less than 1% of a tendon’s dry weight. Large, intricate macromolecules called proteoglycans have a protein core that is covalently attached to one or many of the glycosaminoglycans (GAGs) enclosed. Glycosaminoglycans are linear molecules made up of repeated disaccharide units that have one end attached to the protein core and one end radiating outward in the shape of a “bottlebrush” (Oatis 2009). In tendons, the accumulation of GAGs is significantly lower. Proteoglycan molecules, on the other hand, are stiffly stretched due to their great charge density and charge-to-charge repulsion force (Oatis 2009), which helps tendons withstand tensile and compression pressures. These molecules’ polarity also draws and retains water in the connective tissues. This hydrophilic quality aids in the sustenance of tendon extensibility under tensile loads. For instance, a distraction force can cause a wet tendon to readily extend, whereas a dry tendon will lose compliance. Hydrophilic characteristics of proteoglycans mean they enable the quick diffusion of molecules that are soluble in water, and movement of cells inside the tendon’s extracellular matrix. Because proteoglycans give cellular and fibrous connective tissue components support and space, they also aid in controlling and maintaining the tissue’s structural organization (Oatis 2009). Figure 1.1. Ligament and tendon structural composition NOTES ON FIGURE 1.1.– A. From the tropocollagen molecule gross structure, the tendon and ligament are arranged hierarchically. Optical microscopy allows for the observation of fibers and their crimp pattern. Only electron microscopy delivers a visual representation of the fibril levels. B. Tropocollagen molecule end-to-end and lateral aggregation forming microfibrils. Tropocollagen molecules have micro-failures at their amorphous connections (Oatis 2009). GAGs include hyaluronic acid, chondroitin sulfate and dermatan sulfate; however, dermatan sulfate is typically found in tendons. Decorin and biglycan are two examples of frequent proteoglycans found in tendons. The three primary components of tendons are the bone insertion, the tendon itself and the muscle–tendon junction. They usually go through the joints and attach to the distal surface, thus improving the ability of muscle to work with the joints. The mechanical qualities of tendons differ according to the location of load bearing. Not all muscles have tendons, in actuality. Although certain tendons are directly engaged in joint movement, in other cases their function is to enhance the distance over which the muscle can move rather than the joint itself. The Achilles tendon, for instance, is a unique tendon that helps the body bear weight by concentrating the strength of a small number of muscles. Conversely, certain tendons, such as the posterior tibial tendon, function by dispersing the weight across multiple bones. While it is well known that the majority of tendons come from the muscle and attach to the bone, other tendons may serve as a muscle’s precursor or serve as a connection between two muscles (Buckwalter et al. 2000; Vesentini et al. 2013; Kaya et al. 2019). Classifying tendons based on their shapes is the simplest method, as it takes into account their anatomical structures, settlements and forms. They can have extremes in size and length – they can be both very little and quite long. Tendons can vary greatly in shape; they can be long, circular, rope-shaped (such as the Achilles tendon) or short. They can also exhibit flat tissue adhesion, as in the case of the bicipital aponeurosis. Basically, tendons have the ability to transform from a flat shape to a cylinder and from a fan to a ribbon shape. The two types of tendons in this basic classification are round and flat, which differ from one another as structural and functional. For instance, flat tendons such as rotator cuffs might respond micro-anatomically in the form of compression and shear forces because of longitudinal, oblique and transverse collagen sequences, whereas round tendons such as the flexor digitorum profundus respond similarly to tensile stresses with parallel collagen patterns. While, in circular tendons, the section area is rightly related to the muscle’s maximum isometric strength. Compared to spherical tendons that react to tensile evenly, flat tendons are resistant to compression and shear forces because of their parallel collagen sequences, which are flat, longitudinal and oblique (Buckwalter et al. 2000; Vesentini et al. 2013; Heybeli et al. 2016; Kaya et al. 2019). There are various classification schemes for tendons based on location; however, the most sensible scheme considers the tendon in relation to its perceived functions, which include extraarticular (Achilles tendon) and intra-articular (popliteus tendon, biceps long head). The majority of tendons are non-articular, but unlike intra-articular ligaments (such as an anterior cruciate ligament tear), intra-articular tendons are not able to heal following damage. Simultaneously, while the majority of tendons attach themselves to the bone, some help to connect two muscles (such as the omohyoid and digastric muscles) or serve as the origo point for muscles (the flexor digitorum profundus is where the lumbrical muscles begin). Furthermore, the gastrocnemius and soleus muscles may be the source of a significant portion of the tendon. For example, tendons in certain muscles enter the muscle joint and stick out at an angle. As a result, the muscle–tendon unit is stronger but has a smaller range of motion since a large percentage of muscle fibers are able to stick to the tendon (Kaya et al. 2019). The tendons can also be categorized based on their anatomy as unsealed or paratenon-coated (e.g. Achilles tendon) or sheathed or synovial-coated (e.g. the long flexor of the fingers). This suggests that these tendons, which are divided into intra-synovial and extra-synovial categories, have a higher slippage resistance when compared to the intra-synovial tendon structure when more closely examined. Additionally, the soft tissue protection and vascularity of these two tendons differ. Sheathed tendons and tendons wrapped with paratenon are the two different types of tendons. The vascular supply of each varies primarily. A mesotenon, or vincula, in sheathed tendons carries a vessel that supplies a single portion of the tendon. As a result, vascularity of a few tendons is limited; thus, diffusion becomes necessary. However, capillaries that pierce the tendon surface and establish a rich capillary system feed blood to tendons covered in paratenon. A variation in the vasculature is the reason why tendons covered in paratenon recover more quickly. Ligaments get their blood supply from insertion sites (Kaya et al. 2019). There is very finite vascularization in tendons. Their metabolic activity during healing and restoration is directly impacted by this. Only 1%–2% of their extracellular matrix (ECM) is made up of blood vessels. As a result, in contrast to the highly vascular red muscles from which they emerge, they seem white. Their anatomical location, morphology, history of injury and degree of physical activity are some of the features that affect their blood supply in addition to their restricted vascularity. Because of their shape and function, or because of their anatomical position and attachments, some tendons, for example, have been shown to be more vascular (e.g. peroneal tendon). Furthermore, evidence states that blood flow is enhanced in tendons and surrounding tissues after bouts of higher-intensity physical activity without showing signs of tissue ischemia, even under extreme loading conditions. The high-intensity physical activity following an injury that appears to initiate neovascularization and revascularization in previously avascular regions thus increases blood supply. Blood arteries in the paratenon or mesotenon, the periosteal insertion and the surrounding tissue immediately feed the tendon with blood. The term “vascular tendons” refers to tendons encircled by paratenon, while “avascular tendons” alludes to tendons encircled by tendon sheaths. This is an inaccurate description, as the mesotenon has blood arteries embedded in these “avascular” tendons. Vascular tendons have several peripheral entry sites, a longitudinal capillary system that anastomoses, and an endotenon that encircles the fiber bundles (fascicles). A sheathed tendon has two distinct blood supply mechanisms: it is submerged in synovial fluid within the sheaths. Mesotenons, which can be folded or elongated structures called vincula, are the conduits for blood arteries. Digital tendons contain the latter. There are regions of increased and decreased vascularity due to the regular connections made by the tendon sheath and tendon via vincula. Many researchers have proposed a dual channel for tendon nourishment as a result of the hypovascular regions: a vascular pathway and a synovial (diffusion) system exclusive to the hypovascular regions. Since there is a suggestion that healing and repair can occur for tendons contained in a sheath even in the absence of adhesions, the concept of diffusional feeding is of clinical importance. Tendon vascularity plays a critical role in the health and healing of tendons. Poor vascular supply can lead to tendon degeneration and delayed healing, particularly in tendon injuries. Artificial intelligence (AI) is increasingly being applied to study tendon vascularity, enabling more accurate, non-invasive and detailed assessments of blood flow within tendon structures. Below are the key ways AI is used to analyze tendon vascularity. Thus, AI is playing an increasingly important role in studying tendon vascularity, offering sophisticated, non-invasive methods to analyze blood flow, predict healing and optimize treatments. By leveraging advanced imaging techniques, machine learning and predictive modeling, AI enables clinicians to assess tendon health more accurately and intervene earlier, leading to improved outcomes in tendon rehabilitation and injury prevention. Two forms of tendinous junctions have to be recognized as tendons as they join skeletal muscles to bony structures, for example, musculotendinous tendon attachments to the muscle, and osteotendinous tendon attachments to the bone. At the osteotendinous junction, four different zones have been identified, with a slow transition between them (see Figure 1.2). With less PG decorin, the initial zone shares structural similarities with the tendon propter. The second zone is called fibrocartilage, and it contains primarily collagen types II and III with trace amounts of types I, IX and X. Moreover, PG decorin and aggrecan are less prevalent. Collagen type II predominates in the third zone of mineralized fibrocartilage, although aggrecan and collagen X are also present in significant amounts. Minerals and collagen type I make up the majority of the bone in the fourth zone (Miller 2008; Jung et al. 2009; Kaya 2020). Muscle cells are involuted and folded at the musculotendinous junction to provide the most surface area for fibrils to connect to. Because the quickly contracting muscles’ sarcomeres are shortened at the junction, the force intensity at that point may be lessened (Miller 2008; Robi et al. 2013). Figure 1.2. An osteotendinous junction. An osteotendinous junction; B – bone; MF – mineralized fibrocartilage; F – fibrocartilage; T – tendon (Miller 2008) The tendinous junction, where tendons connect to muscles and bones, plays a crucial role in force transmission and movement mechanics. The effective functioning of these junctions is essential for the proper functioning of musculoskeletal systems. AI is increasingly being applied to study the properties and behaviors of tendinous junctions, providing deeper insights into their mechanics, pathology and response to injury. The following sections explain how AI is used for studying the functions of tendinous junctions. AI thus offers valuable tools for studying the functions and mechanics of tendinous junctions. By enhancing imaging, providing predictive models and enabling real-time monitoring, AI facilitates a deeper understanding of tendon health and mechanics. Its application improves injury prevention, diagnosis, rehabilitation and recovery, ultimately advancing treatments for tendon and musculoskeletal injuries. Tendons possess sufficient strength to withstand the high tensile stresses generated by muscle contraction during joint motion. They also possess enough flexibility to flex around the surfaces of bones and deflect beneath retinacula, thus altering the direction of muscle pull (Nordin and Frankel 2001). Experimental studies of the bio-mechanical characteristics of tendons are often conducted in uniaxial tension, as their major purpose is to transmit tensile stresses. Furthermore, stress–strain curves are produced by uniaxial tensile tests. In order to achieve this, the tensile load is normalized by the structure’s cross-sectional area (i.e. stress), and the change in elongation in a specific tissue region is normalized by the original length (meaning strain). A uniform distribution of stress throughout the material is one of the test’s presumptions (Nordin and Frankel 2001). Some instrumentation can also be used to quantify in situ forces in people, for instance implantable transducers, magnetic resonance imaging, kinematic linkage measurements, the use of buckle transducers and instrumentation at insertion locations. The field of tissue biomechanics is benefiting greatly from advancements in other techniques including finite element modeling (FEM), ultrasound imaging and robotic/universal force-moment sensor (UFS) testing. In addition, load-elongation curves and stress–strain diagrams are obtained by analyzing the structural and mechanical properties of tendons and ligaments using the mentioned methods (Nordin and Frankel 2001). Both normal and excessive loading primarily apply tensile loads on tendon tissue. When damage occurs, both the amount and the rate of loading have an impact on how much damage is done. Mechanical characteristics of the tendon are gaged by applying uniaxial tensile loads to tissue preparations until they fail. Usually, tendon or bone–ligament–bone complexes make up tissue preparations. By graphing the externally applied force against the corresponding tissue elongation, the data from these tests are used to construct load-deformation curves. The structural characteristics of the tissue under test are represented by these load-deformation curves. The mechanical qualities are dependent upon the tissue’s composition, the collagen fibers’ orientation and the interplay between the ground substance’s constituents and collagen. However, the extraction of entire tissues is necessary using this method of assessing the mechanical properties of tendons. Studies that make use of these techniques thus use human tissue that has been extracted after surgery or tissues from a range of animal models. With the use of cutting-edge techniques such as ultrasound scanning techniques, scientists can now examine how different factors, such as aging and exercise, affect human tendon mechanical properties as they move (Oatis 2009). The load-elongation curve provides information about the tensile strength of a ligament or tendon after it has been loaded to failure. The slope of the curve between two limits of elongation in a load-elongation curve represents the structure’s stiffness (N/nm). It shows the maximum load and/or elongation that the structure can withstand before collapsing. The maximum load applied to the structure prior to failure is known as the ultimate load (N). The maximal elongation of the complex at failure is known as the ultimate elongation (mm). Lastly, the area under the whole curve, or the maximum energy retained by the complex, is the energy absorbed at failure (N/mm) (Nordin and Frankel 2001). Multiple zones on load-elongation curves reflect the tissue’s behavior. The “toe” zone is the initial area on the load-elongation curve. It is thought that a change in the relaxed collagen fibers’ crimp or wavy pattern is what causes the elongation seen in this area. The collagen fibers in this area become straight and lose their wavy look, the tissue stretches readily under mild pressures, and as the loading increases, sliding happens between the fibrils and fascicles (Nordin and Frankel 2001). As the stress rises, the tissue becomes more rigid, which alters the tissue’s elongation. The elastic or linear section of the curve is what is known as this area. It is characterized by a sharp rise in the curve’s slope that follows the toe area. Fiber bundle catastrophic failure happens in an unanticipated way when the linear region is exceeded. If there are irreversible alterations (failure), the curve may finish abruptly or slope downhill. When the maximum load is reached, which represents the specimen’s ultimate tensile strength, full failure happens quickly and the tendon’s capacity to support loads is significantly diminished. The resulting force, or load (N), is plotted after the tissue is stretched until it ruptures (Nordin and Frankel 2001). Figure 1.3. Load-elongation curve following loading a tendon structure to failure NOTES ON FIGURE 1.3.– The elongation the structure experiences due to loading, expressed in millimeters, is shown by the x axis. The tissue’s tensile load, expressed in newtons (N), is represented by the y axis (Nordin and Frankel 2001). Stress–strain curves are also produced in order to further test the tensile deformation of tendon and ligament specimens (Nordin and Frankel 2001). The load-elongation curve of a tendon is critical for understanding its mechanical properties, such as elasticity, stiffness and failure point. By analyzing how tendons elongate under different loading conditions, AI can provide valuable insights into tendon behavior, helping to predict potential injuries and improve treatment strategies. The following sections discuss how AI is used to study the load-elongation curve of tendons.
1
Advancements and Applications of Physiotherapy in Rehabilitation and Pain Management
1.1. Introduction
1.2. Composition of connective tissue
1.2.1. Composition of tendons
1.3. Tendon classification
1.4. Tendon vascularity
1.4.1. Artificial intelligence (AI) to study tendon vascularity
1.4.1.1. Imaging and artificial intelligence analysis
1.4.1.2. Artificial intelligence in vascularity quantification
1.4.1.3. Machine learning for tendon vascularity patterns
1.4.1.4. Predictive modeling for tendon healing
1.4.1.5. Real-time monitoring
1.4.1.6. Clinical decision support
1.4.1.7. Tendon disease diagnosis
1.4.1.8. Guiding interventions to improve vascularity
1.5. Tendinous junction
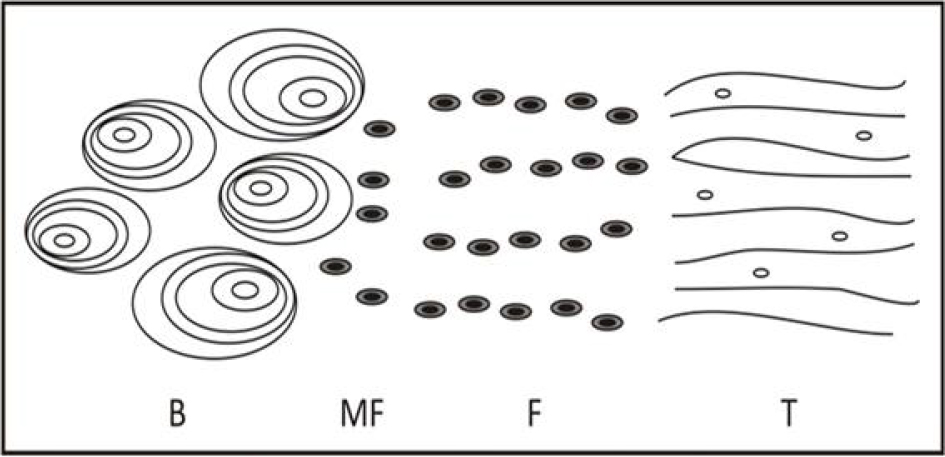
1.5.1. AI for studying functions of the tendinous junction
1.5.1.1. Imaging analysis and visualization
1.5.1.2. Force transmission assessment
1.5.1.3. Predicting tendon failure
1.5.1.4. Real-time monitoring and feedback
1.5.1.5. Automated detection of pathologies (Muir et al. 2025)
1.5.1.6. Simulation and virtual modeling
1.5.1.7. AI in rehabilitation planning
1.5.1.8. Understanding age-related changes
1.5.1.9. Muscle–tendon interactions
1.5.1.10. Clinical decision support
1.6. Mechanical properties
1.6.1. Load-elongation curve

1.6.2. Using AI to study load-elongation curves
1.6.2.1. Data analysis and pattern recognition
1.6.2.2. Predictive modeling
1.6.2.3. Optimization of tendon testing
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree